Alexandrium tamarense
Topic: Biology
 From HandWiki - Reading time: 23 min
From HandWiki - Reading time: 23 min
| Alexandrium tamarense | |
|---|---|
| Scientific classification | |
| Domain: | Eukaryota |
| Clade: | Diaphoretickes |
| Clade: | SAR |
| Clade: | Alveolata |
| Phylum: | Myzozoa |
| Superclass: | Dinoflagellata |
| Class: | Dinophyceae |
| Order: | Gonyaulacales |
| Family: | Ostreopsidaceae |
| Genus: | Alexandrium |
| Species: | A. tamarense
|
| Binomial name | |
| Alexandrium tamarense (Lebour) Balech
| |
Alexandrium tamarense is a species of dinoflagellates known to produce saxitoxin, a neurotoxin which causes paralytic shellfish poisoning (PSP)[1]. Historically, it has been treated as part of the Alexandrium tamarense species complex alongside A. fundyense and A. catenella, but molecular studies showed that the three original morphospecies do not form distinct monophyletic groups [2][3].
Like other cyst forming Alexandrium species, its lifecycle includes motile cells, sexual stages, resistant resting cysts, and its blooms are shaped by environmental conditions such as temperature, nutrients, salinity, light, oxygen, and water movement [4][5][6][7][8].
The species is important because its blooms impact marine food webs, human health, and coastal economies, and because tracking and identifying it requires a combination of morphology-based and bloom-monitoring methods[4][6][8][9][10][11][12][13][14].
In the post-2014 taxonomy, the name Alexandrium tamarense refers exclusively to the former Group III of the historical species complex, so older reports using the name must be interpreted carefully[15].
History of Knowledge
The species now known as Alexandrium tamarense was first described by Lebour in 1925 from the Tamar River in southern England under the basionym Gonyaulax tamarensis Lebour[16][17][15]. During the 20th century, the taxon moved through several taxonomic placements including Gonyaulax tamarensis, Protogonyaulax tamarensis, and Gesserium tamarensis, before being classified under the name Alexandrium tamarense (Lebour) Balech in 1995[18][19][15] .
Before genetic analysis was standardized, classification within the genus relied heavily on morphology. Three classical morphospecies, A. tamarense, A. cantanella, and A. fundyense, were defined based on cell shape, chain forming ability, and the presence of a ventral pore[20]. This framework shaped most of the older literature on bloom dynamics, distribution, and shellfish toxicity[2][19]. Within the historical A. tamarense complex, these morphological characters proved more variable than once assumed, and intermediate forms were observed in both feild material and cultured isolates.[20][15].
A major turning point came when molecular studies in the 1990s and 2000s found that the historical morphospecies didn't correspond to distinct evolutionary lineages[3][15][20]. This re-evaluation led to the recognition of five groups within the historical Alexandrium tamarense species complex, and eventually to the formal taxonomic revision in 2014[15][20]. In that revision, the name Alexandrium tamarense was restricted to former Group III, tying the modern species concept to the Tamar estuary type locality[15] . This change is important because much of the older literature used the name Alexandrium tamarense actually refers to the broader historical species complex rather than the narrow modern species concept[15].
Later work also expanded knowledge of the species beyond taxonomy by examining its life cycle, especially the role of resting cysts in bloom formation and recurrence[4][5][21].
Studies on bloom ecology then showed that salinity, temperature, nutrient availability, freshwater input, stratification, and meterological conditions also help shape bloom development [4][5][6][7][8].
Research also developed methods for tracking blooms through thecal plate staining, repeated cell counts, and long-term monitoring linked to environmental data[6][7][8][21].
At the same time, work on saxitoxin, paralytic shellfish poisoning, and bloom impacts showed that Alexandrium tamarense has major ecological, public-health and economic significance[1][9][10][11][12].
Morphology
Cell structure
Alexandrium tamarense is a thecate dinoflagellate, and species in Alexandrium share a broadly similar Kofoidean plate pattern [22][19]. Like many species in the genus, the cells are relatively featureless in light microscopy unless the plates are stained, dissected, or examined using electron microscopy[19]. Historically, identification of A. tamarense depended on subtle characters such as cell size/shape, chain formation, the shape of the apical pore complex, and the details of sulcal and apical plates[19] . Calcofluor White staining became an important practical method for visualizing these defining thecal features in dinoflagellates[21][22] .
Detailed published descriptions specific to the modern Alexandrium tamarense taxon are limited, so morphological descriptions often draw on older literature or cultures identified within the historical complex. Cells isolated in Scottish waters, identified morphologically as A. tamarense, Brown et al. described the cells as ranging from slightly rounded to more hexagonal to slightly oval and measuring 14.5-39.2 μm in diameter and 24.5-44.2 μm in length[22]. Brown et al. also noted that the cells occurred singly or in short chains of two cells, with occasional chains of 3 or 4 cells[22]. Those cultures showed a deeply excavated cinglulum: the apical pore complex was attached to the first apical plate, an obvious ventral pore was present in the first apical plate, and the posterior sulcal attachment pore varied between and within cultures. As those descriptions were based on morphology alone [23].
Phylogenetics
Ribosomal markers, especially LSU, SSU, and ITS regions, played a central role in taxonomic reinterpretation of the historical Alexandrium tamarense complex because they showed that the traditional morphospecies did not form discrete monophyletic groups.[3][15][20]. Molecular studies in the 1990s identified five phylogenic ribotypes (North American, Western European, Temperate Asian, Tasmanian, Tropical Asian) that disagreed with the species boundaries proposed by the historical morphospecies [2][19]. LSU rDNA data found that the three historical morphospecies did not satisfy phylogenetic, biological, or morphological species concepts and suggested that the historical morphospecies names within the concept should be discontinued[24]. Instead, the study recognized five well supported groups (Groups I-V) and developed a numbered scheme because the older geographically named ribotypes had become misleading[24]. SSU based work then showed strong intragenomic polymorphism in the small-subunit rDNA of some strains, and that ignoring this variation may inflate estimates of diversity in bloom samples[3]. This SSU based analysis divided the historical morphospecies into two distinct clades and linked those clades to the five groups recognized by Lilly and colleagues[3][20]. Uwe and colleagues then formalized the revision in 2014 by assigning Group I to A. fundyense, Group II to A. mediterraneum, Group III to A. fundyense, Group IV to A. pacificum, and Group V to A. australiense [15].
Following the 2014 taxonomic revision, the name Alexandrium tamarense applies specifically to former Group III[15]. Uwe et al. linked that assignment to the Tamar estuary type locality, and reported that Group III isolates known at that time were non-toxic [15]. Klemm and colleagues emphasized how important this revision was for interpreting the record from northern Europe, because many older light-microscopy reports of A. tamarense likely included what is not recognized as A. catenella or other members of the complex[23].
Gene level analysis provided a functional perspective on interpretation of the complex [25][19]. Stüken and colleagues showed that saxitoxin related genes such as sxtA are encoded in the nuclear genome rather than only in associated bacteria[25]. They also found strong overall agreement between the presence of genomic sxtA and saxitoxin production, although they noted a few strains identified at the time as A. tamarense in which sxtA was detected but toxin was not[25][19]. Those strains predate and therefore do not necessarily follow the 2014 revision, they should be interpreted cautiously when discussing the modern concept of A. tamarense [15][23].
Lifecycle

There are nine stages in the lifecycle of Alexandrium tamarense with transitions throughout the lifecycle between asexual and sexual reproduction [26]. The first stage is the replication of vegetative motile cells through binary fission[26]. Next, if environmental conditions are unfavorable, cells may enter a resting state called a temporary or pellicle cyst [26]. This resting stage allows the cells to withstand short term unfavorable environmental conditions[26]. When environmental conditions become favorable again cells will exit this resting stage and become vegetative and motile and ready to enter the sexual reproduction stages of the lifecycle[26]. Formation of female and male gametes terminates asexual reproduction and facilitates sexual reproduction[26]. The male and female gametes then fuse together to produce a swimming zygote also known as a planozygote[26]. After formation of the planozygote the planozygote becomes a hypnozygote which is a dormant cyst[26]. In the dormancy stage the hypnozygote is found in the sediment and serves as an inoculum population for bloom formation whereas when cells are vegetative and motile they inhabit the water column[4][5]. Transforming into a dormant cyst allows Alexandrium tamarense to endure harsh environmental conditions that would negatively impact vegetative growth[5]. During the dormancy or hypnozygote stage A. tamarense has low metabolic activity and germination is not possible[26][5]. The dormancy stage is mandatory for Alexandrium cysts to mature in order to germinate, initiate a bloom, and then enter the final stage of the lifecycle which is asexual reproduction using binary fission to produce more vegetative cells[26].
Bloom initiation is dependent on exiting of the dormancy or hypnozygote stage to the germination stage, which is impacted by many factors including endogenous and environmental[26][5]. Exposure time to low temperatures can impact the germination ability of A. tamarense[5]. The environmental factors of temperature, salinity, oxygen, and light impact germination rates[26][5]. In Masan Bay, Korea seasonal blooms of A. tamarense are connected to seasonal variations in salinity and dissolved oxygen[5]. The dormancy period can also be controlled by an endogenous annual clock and not external environmental conditions[26][5]. Genovesi et al., 2009 suggest that resuspension of resting cysts from the sediments into the water column could be a prerequisite for germination[5]. In addition, strong wind sequences that facilitate resuspension of dormant cysts from sediments followed by several weeks of favorable temperatures has promoted bloom formation and exiting of the dormancy stage[5]. Therefore, there are many factors impacting the success rate of bloom formation of A. tamarense and in different environmental systems the steps involved in exiting dormancy differ.
Ecology
The most prominent grazers of A. tamarense are small heterotrophs including microzooplankton like the tintinnid ciliate Favella taraikaensis [27], other heterotrophic dinoflagellates such as Oxyrrhis marina, Gyrodinium dominans, Polykrikos kofoidii, and Strombidinopsis spp. [28], as well as larger zooplankton, such as copepods (Calanus helgolandicus, Acartia clausii, and Oithona similis) [29]. In the open ocean, these grazers play the important role of regulating the growth of A. tamarense populations, preventing overgrowth and eutrophication as well as potential toxin blooms. However, it has been found that many Alexandrium species respond to grazing cues with elevated toxin production of their own, in an apparent defense mechanism. These cues and responses, however, are species-specific and cannot be generalized for the grazer community as a whole [30]. For A. tamarense, this has led to something of an evolutionary arms-race, wherein some copepod grazers appear themselves to have developed an immunity to saxitoxin to continue their grazing without consequence [29].
Another group of A. tamarense consumers which are perhaps the most notorious are the filter-feeding bivalves which give name to the saxitoxin-induced Paralytic Shellfish Poisoning. Due to the mechanism of filter feeding, bivalves like mussels, scallops, and clams are nondiscriminate consumers, with the main factor regulating their diet is the size of filtered particles. Because of this, high levels of toxin can accumulate in bivalve tissues, which ultimately leads to transmission of saxitoxin to marine food webs and beyond. Among intertidal organisms, the BC Centre for Disease Control specifically outlines clams, mussels, whelks, moon-shells, dogwinkles, oysters, whole scallops, crabs and lobster as some of the most dangerous species to avoid eating during bloom periods[31]. The effect of saxitoxin varies significantly between species, with some shellfish having very rapid detoxification times while others can retain dangerous levels of saxitoxin for months to years[9].
Ecologically, the implications of an A. tamarense bloom and the injection of saxitoxin into the foundation of ecosystems can have drastic effects on food webs in general. In zooplanktivore populations of fish, filter feeders, and even whales, as well as their predators, even low levels of toxicity in primary consumers can accumulate and have serious, even lethal consequences. In July of 1976, a massive kill of herring was determined to have been caused by A. tamarense toxicity, with repeat incidents identified with markedly fast mortality from toxins accumulated through zooplankton grazing [10]. In the St Lawrence Estuary, a 2008 bloom of A. tamarense was linked to mass mortality of beluga, seals, porpoises, birds, and fish [11]. The potential for loss of predator populations has the potentially to completely reshape marine systems and food web dynamics.
Geography
Several studies have recorded Alexandrium tamarense in multiple locations around the world, with higher populations favouring temperate waters. This species is not restricted to any specific ocean basin or sea region. In North America, it has been documented in several Canadian and American waters, including the Strait of Georgia (British Columbia, Canada), the Gulf of St. Lawrence (Quebec, Canada), and the Gulf of Maine (Maine, USA) [6][32]. Outside North America, strains have also been recorded in coastal Japan, with recurrent algal blooms in Hiroshima Bay (Japan)[33]. Populations have also been found off the coast of Europe, and within the Southern Hemisphere, with populations existing off the coast of Argentina, New Zealand, and Australia[34].
Environmental Conditions
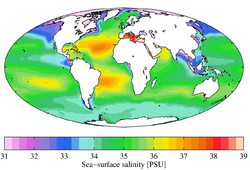
Salinity
Alexandrium tamarense concentrations have been found in temperate brackish waters around coastal environments. The dinoflagellate has been linked to thriving in regions with freshwater input, increased stratification, and increased nutrients [6][7]. Research done within the St. Lawrence estuary suggests that there is a strong correlation between low salinity, high freshwater input, and strong bloom development [7]. Weise et al., 2002 suggest the same outcome accounting for average cell growth over a 10-year period, stating that bloom development is often associated with conditions where river water input lowers surface salinity and increases the stratification of the water column[6]. Stratification allows for stable conditions within a water column, allowing for favourable conditions to remain long enough for bloom growth[6]. Weise et al., 2002 suggest that a salinity percentage of 20-26% correlated with the highest A. tamarense cell concentrations[6].
Temperature

Temperature has additionally been found to be a control for Alexandrium tamarense blooms. Peak A. tamarense cell concentrations were found in warmer waters > 12°C [6]. Temperate areas with strong seasonal warming often provide the ideal conditions for bloom growth within the summer season [6][7]. Locations such as the Strait of Georgia, the Gulf of St. Lawrence, and Hiroshima Bay all experience strong seasonal warming patterns that are able to support bloom growth [6][33][7].
Meteorological Conditions
Specific meteorological conditions have been shown to play a role in bloom intensity [6]. Studies have shown a correlation between periods of high river runoff, increased precipitation, and A. tamarense cell concentration growth [6][7]. It is not determined whether the impacts freshwater has on salinity and stratification or the increase in available nutrients is the cause for this increase in cell concentration [6]. Studies have also shown that low wind conditions are ideal for cell growth, as the reduction of turbidity allows for ideal conditions to remain for cell growth [6]. In the St. Lawrence estuary, cell growth was seen to occur with wind speeds around 3.3 ± 1.8 m/s with disruption occurring at wind speeds > 7 m/s [6]. These strong winds likely disturb stratification, causing the dispersal of cell concentrations and nutrients needed for cell growth. This can also have an impact on salinity and temperature, causing issues with vertical migration patterns [6][8]. Periods of high precipitation on a day-to-day scale was correlated to high A. tamarense cell counts as well [6]. The initiation of bloom development in the St. Lawrence estuary is characteristically prior to heavy rainfall events occurring at the beginning of spring and fall [6].
Nutrient Variability
Studies have shown a correlation between nutrient availability and Alexandrium tamarense cell growth. In the St. Lawrence estuary, a link was established between A. tamarense growth and nitrogen, phosphorus, and ammonium concentrations, suggesting that these nutrients are the most vital to support A. tamarense development [7]. Fauchot et al., 2005 state that a nitrogen-phosphorus ratio below the Redfield Ratio (N:P=16) was the most favourable for increased cell abundance, and a non-linear relationship was established between phosphate concentrations and A. tamarense growth rates. Results from the St. Lawrence estuary suggest that phosphate-controlled growth rates, while nitrogen controlled the number of divisions achieved by A. tamarense [7].
Humic substances may influence bloom growth, suggesting that terrestrial coastal waters can create conditions favourable for growth [6]. Weise et al., 2002 suggest that the nutrients, nitrogen, phosphorus, and ammonium are easily replenished within these areas due to high freshwater runoff and high nutrient input[6]. Fauchot et al., 2005 suggest that salinity has a greater impact on Alexandrium tamarense cell abundance than nutrient availability[4].
Nutrient availability can impact PSP toxicity [8]. A relationship between toxin concentration and nitrogen availability was found by MacIntyre et al.,1997 where toxin concentrations were found to increase with greater nitrogen-stratification. Minimal nitrogen concentration near-surface and higher deep nitrogen concentration levels resulted in this toxin increase, proving that A. tamarense uses diel vertical migration to obtain nutrients [8].
Methods to Measure Blooms
To track Alexandrium tamarense blooms, methods were focused primarily on tracking changes in cell abundance and cell growth rates. Repeated cell counts were conducted across different sites, seasons, and years to determine bloom rates and distribution [7]. Cell counts found over prolonged periods performed by Weise et al., 2002 were used to compare hydrological and meteorological conditions to determine environmental controls on phytoplankton blooms[6]. Lilly et al., 2007 measure bloom development to find A. tamarense species boundaries[20]. To find nutrient dependencies, MacIntyre et al., 1997 used a controlled laboratory environment and changes in nutrient availability to measure the impacts on A. tamarense toxin concentration and growth rates[8]. Long-term monitoring, cell count monitoring, and species distribution are common techniques for analysis.
Thau lagoon, Southern France Bloom Dynamics
An environment that experiences consistent blooms of A. tamarense is Thau lagoon, France[5]. To determine the optimal environmental conditions that facilitate A. tamarense bloom formation Genovesi et al., 2009 used laboratory produced cysts and compared germination patterns and germling cell viability to natural cysts from Thau lagoon, France[5]. It was found that during the dormancy stage cysts survive due to living in sediments during the winter and that there is no requirement for vernalization[5]. Then resuspension of cysts into the photic zone away from anoxic sediment conditions promotes bloom seeding[5]. The resuspension is thought to be triggered by windy periods that also restore dissolved oxygen saturation in the water column during spring and summer[5]. Hadjadji et al., 2014 found that there is a connection between inorganic phosphorus limitation and germination of A. tamarense cells to initiate blooms in Thau Lagoon, France[35].
St. Lawrence Estuary Bloom Dynamics
The St. Lawrence estuary in eastern Canada experiences recurrent A. tamarense blooms[4]. One of the important relationships that contribute to bloom formation are cyst germination and estuarine circulation[4]. Fauchot et al., 2008 found that A. tamarense blooms that develop near the north shore of the estuary get transported to the south shore via the Manicouagan and Aux-Outardes plume and that the plume remaining along the north shore for several days could be a pre-requisite for development of blooms[4].
Chukchi Sea shelf Bloom Dynamics: Impact of Global Warming
In the Chukchi Sea Shelf it was discovered using in vitro cyst germination experiments that cell growth and cyst germination increased with warmer water temperatures and that winter formed water of the Chukchi Sea Shelf inhibits effective germination[36]. This finding led Natsuike et al., 2017 to infer that A. tamarense blooms could occur with future warming, which can have impacts on animal taxa in the Chukchi Sea Shelf[36].
Saxitoxin
Paralytic Shellfish Poisoning
Saxitoxin is a potent neuro toxin produced by A. tamarense, marine dinoflagellates, freshwater or brackish water cyanobacteria and is the causative agent of paralytic shellfish poisoning[1]. It is a part of the paralytic shellfish toxins group and has a tricyclic structure with a biguanide group[1]. Its mechanism of action is blocking sodium ion influx into voltage gated sodium channels which prevents nerve impulse transmission, and can cause serious physiological problems in the nervous, respiratory, cardiovascular and digestive systems[1]. The neurotoxin has a minimum lethal dose of 1-4mg/kg and a median lethal dose of 10ug/kg[1].
Symptoms of paralytic shellfish poisoning according to the BC CDC are[37]:
- Tingling
- Numbness spreading from lips and mouth to face, neck and extremities
- Dizziness
- Arm and leg weakness
- Paralysis
- Headache
- Nausea
- Vomiting
- Respiratory failure and in severe cases death
Paralytic shellfish poisoning can occur from eating at risk shellfish, and or contaminated seafood[37]. The median time between ingestion and onset of symptoms is 1 hour[37]. There are no known available treatments or drugs to treat paralytic shellfish poisoning[37].
Production of saxitoxin
A. tamarense toxin profile
There are 57 known types of saxitoxin (called analogs) that vary slightly based on their level of toxicity and R- group structure. Broadly, the categories of toxin found in A. tamarense are grouped as: Non-sulfated (STX, neoSTX), mono-sulfated gonyautotoxins (GTX 1-6), decarbamylated (dcneoSTX), and di-sulfated (C1,C2) in order of toxicity[38][39]. The saxitoxin profile of A. tamarense (and all Alexandrium spp.) can vary significantly, but is mainly dominated by C2 and GTX4 types [40]. Both the composition and amount of saxitoxin in each cell are variable, with concentrations ranging from 40-120 fmol/cell [40][39].
Regulation of saxitoxin synthesis
Production of saxitoxin in A. tamarense is mainly regulated at the post-transcriptional level[41]. This is thought to be the case for most marine dinoflagellate genes, due to their lack of detectable histone proteins (which regulate transcription during protein synthesis)[41]. There are several levels of saxitoxin regulation which have been described, although the complete process of toxin biosynthesis in Alexandrium has yet to be fully unveiled. Some of the genes which have been identified in the saxitoxin synthesis/release pathways are described in the table below.
| Core synthesis | These genes and enzymes with which they are associated are responsible for building the base structure of non-substituted saxitoxin. | Genes: sxtA, stxB, stxD, stxG, stxH, stxT, stxF, stxU [42] |
| Tailoring | These genes and enzymes carry out the substitution of functional groups that transform STX into various analog toxins through processes like decarbamoylation, N-sulfotransfer, and N1-hydroxylation. The specific proteins involved at each step and for all substitutions are yet to be identified. | Genes: sxtL, stxN, stxX [42] |
| Regulation | Specific genes that regulate saxitoxin production through in A. tamarense have not yet been identified. | Genes: unknown |
| Transportation | These genes and enzymes are responsible for controlling the release of saxitoxin from A. tamarense cells during a toxic bloom event. | Genes: sxtF, stxM, stxP [42] |
Note: The sxtA4 gene is of particular interest, due to its role in initiating saxitoxin biosynthesis. In A. minutium and A. ostenfeldii, the number of stxA4 copies in the genome has been found to correspond with the amount of toxin produced in the cell [42].
Indirect metabolic associations with saxitoxin production
Apart from these specific genes that are directly associated with the biosynthesis pathway, elevated levels of cellular toxins have been found in association with genes and proteins involved in primary metabolic functions. This suggests that for A. tamarense, it is not unlikely that the production of saxitoxin is concurrent with and supported by the process of digestion and photosynthesis, but the specific factors causing this linkage remain undescribed[41].
Proteins associated with amino acid metabolism upregulated during high toxin phase of A. tamarense CI01[41]
| Protein name | Role in primary metabolism | Role in saxitoxin production |
| Argininosuccinate synthase | Catalyses the formation of argininosuccinate during the synthesis of arginine, and is involved in the urea cycle to maintain high NO3- levels [43]. | Arginine is a precursor in the biosynthesis of saxitoxin, and saxitoxin synthesis requires high levels of bioavailable N [44][45]. |
| Methyluridine methyltransferase cystine ABC transporter | Involved in cysteine and proline metabolism, and facilitates the uptake of sulfur [46]. | Sulfated saxitoxin analogs depend on A. tamarense's uptake of sulfur to the cell to complete the transformation of base molecules [47]. |
| Phosphoserine phosphatase |
|
Phosphate availability and general nutrient status is strongly linked to the production of saxitoxin (as described below). |
Proteins associated with photosynthesis, upregulated during high toxin phase of A. tamarense CI01 [41]
| Protein name | Role in photosynthesis |
| Phosphoenolpyruvate carboxylase | Involved in C4-type photosynthesis, and converts phosphoenol pyruvate (3C) to malate (4C) to cope with CO2-limited environments to prevent oxygenation of RubisCO [49]. |
| Chloroplast phosphoglycerate kinase | Involved in carbon assimilation during the Calvin cycle [50]. |
| Peridinin-chlorophyll α-binding protein | A dinoflagellate-specific light-harvesting complex that is specialized to blue-green (470–550 nm) light. This transfers to chlorophyll-A to maximize the efficiency of light capturing and photosynthesis [51]. |
| Mg2+ transporter protein | Magnesium is required for the synthesis and activation of chlorophyll-A, RubisCO, and other important photosynthetic structures [52]. |
Impact of nutrients on saxitoxin production
Based on the association of saxitoxin production with the nutritional status of A. tamarense, it's thought that nutrient availability plays a significant role in the limitation and amplification of saxitoxin production as well as the growth and development of A. tamarense blooms. A 2015 study demonstrated that for a culture of the A. tamarense strain CI01, collected from the South China Sea, population growth rates were limited for samples grown in both nitrate and phosphate - deplete media. However, they found that the cellular concentration of saxitoxin was much higher in the phosphate-deplete test group compared to both the nitrate-limited and high-nutrient samples, while the low-nitrate cultures generated significantly less toxin overall [42]. This means that in A. tamarense, nitrogen is a much more important nutrient to saxitoxin production than phosphate; likely because nitrogen can account for 17% - 34% of the molecular mass of saxitoxin [42], whereas phosphate is generally not a component of the molecular structure. Under phosphate-limiting conditions, growth slows as DNA synthesis and cell division are halted (due to the high P-demand of DNA's phosphate backbone and the phospholipid bilayer) [53]. However, this causes intracellular toxin levels to accumulate, since the production of saxitoxin is not slowed. This also implies that the maximum concentration of toxin is partially limited by the speed of cell division.
Economic Impact of Harmful Algal Blooms
Harmful algal blooms created by A. tamarense can have significant negative economic impacts on the shellfish industry[12][13][14]. In the Korean aquaculture industry there has been a loss of US $121 million during the last 3 decades due to harmful algal blooms[12]. In the United States harmful algal blooms have had significant economic impacts on public health, commercial fisheries, recreation, tourism, environmental monitoring and bloom management[13]. In the United States of America the preliminary and conservative estimate provided by the Woods Hole Oceanographic Institution is that the nationwide average annual costs of harmful algal blooms is approximately $50 million[13]. For the other industries impacted by harmful algal blooms the economic costs are the following: public health the estimate is $20 million spent annually, commercial fisheries is $18 million, $7 million for recreation and tourism impacts, and $2 million for monitoring and management[13]. The National Centers for Coastal Ocean Science states that costs from a single major harmful algal bloom can reach tens of millions of dollars in the United States[14].
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Deng, Huiyun; Shang, Xinrui; Zhu, Hu; Huang, Ning; Wang, Lianghua; Sun, Mingjuan (July 2, 2025). "Saxitoxin: A Comprehensive Review of Its History, Structure, Toxicology, Biosynthesis, Detection, and Preventive Implications". Mar. Drugs 23 (7): 277. doi:10.3390/md23070277. PMID 40710502.
- ↑ 2.0 2.1 2.2 Scholin, Christopher A.; Herzog, Michel; Sogin, Mitchell; Anderson, Donald M. (December 1994). "Identification of Group- and Strain-Specific Genetic Markers for Globally Distributed Alexandrium (Dinophyceae). II. Sequence Analysis of a Fragment of the LSU rRNA Gene 1" (in en). Journal of Phycology 30 (6): 1003–1005. doi:10.1111/j.0022-3646.1994.00999.x. ISSN 0022-3646. https://onlinelibrary.wiley.com/doi/10.1111/j.0022-3646.1994.00999.x.
- ↑ 3.0 3.1 3.2 3.3 3.4 Miranda, L. N.; Zhuang, Y.; Zhang, H.; Lin, S. (2012). "Phylogenetic analysis guided by intragenomic SSU rDNA polymorphism refines classification of "Alexandrium tamarense" species complex". Harmful Algae 16: 35–41–42. doi:10.1016/j.hal.2012.01.002. Bibcode: 2012HAlga..16...35M.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 Fauchot, Juliette; Saucier, François J.; Levasseur, Maurice; Roy, Suzanne; Zakardjian, Bruno (2008-02-01). "Wind-driven river plume dynamics and toxic Alexandrium tamarense blooms in the St. Lawrence estuary (Canada): A modeling study". Harmful Algae 7 (2): 214–227. doi:10.1016/j.hal.2007.08.002. ISSN 1568-9883. Bibcode: 2008HAlga...7..214F. https://www.sciencedirect.com/science/article/pii/S1568988307001242.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 Genovesi, Benjamin; Laabir, Mohamed; Masseret, Estelle; Collos, Yves; Vaquer, André; Grzebyk, Daniel (October 2009). "Dormancy and germination features in resting cysts of Alexandrium tamarense species complex (Dinophyceae) can facilitate bloom formation in a shallow lagoon (Thau, southern France)". Journal of Plankton Research 31 (10): 1209–1224. doi:10.1093/plankt/fbp066.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 6.14 6.15 6.16 6.17 6.18 6.19 6.20 6.21 6.22 Weise, Andréa; Levasseur, Maurice; Saucier, François; Senneville, Simon; Bonneau, Esther; Roy, Suzanne; Sauvé, Gilbert; Michaud, Sonia et al. (2002). "The link between precipitation, river runoff, and blooms of the toxic dinoflagellate Alexandrium tamarense in the St. Lawrence". Canadian Journal of Fisheries and Aquatic Sciences 59 (3). https://doi.org/10.1139/f02-024.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 Fauchot, Juliette; Levasseur, Maurice; Roy, Suzanne; Gagnon, Réal; Weise, Andréa M. (2005). "ENVIRONMENTAL FACTORS CONTROLLING ALEXANDRIUM TAMARENSE (DINOPHYCEAE) GROWTH RATE DURING A RED TIDE EVENT IN THE ST. LAWRENCE ESTUARY (CANADA) 1" (in en). Journal of Phycology 41 (2): 263–272. doi:10.1111/j.1529-8817.2005.03092.x. ISSN 0022-3646. https://onlinelibrary.wiley.com/doi/10.1111/j.1529-8817.2005.03092.x.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 MacIntyre, J.; Cullen, John; Cembella, Allan (1997). "Vertical migration, nutrition and toxicity in the dinoflagellate Alexandrium tamarense". Marine Ecology Progress Series 148: 201-216. https://www.int-res.com/articles/meps/148/m148p201.pdf.
- ↑ 9.0 9.1 9.2 Bricelj, V. Monica; Shumway, Sandra E. (December 1998). "Paralytic Shellfish Toxins in Bivalve Molluscs: Occurrence, Transfer Kinetics, and Biotransformation" (in en). Reviews in Fisheries Science 6 (4): 315–383. doi:10.1080/10641269891314294. ISSN 1064-1262. Bibcode: 1998RvFS....6..315B. https://www.tandfonline.com/doi/full/10.1080/10641269891314294.
- ↑ 10.0 10.1 10.2 Turner, Jefferson T.; Tester, Patricia A. (July 1997). "Toxic marine phytoplankton, zooplankton grazers, and pelagic food webs" (in en). Limnology and Oceanography 42 (5part2): 1203–1213. doi:10.4319/lo.1997.42.5_part_2.1203. ISSN 0024-3590. Bibcode: 1997LimOc..42.1203T.
- ↑ 11.0 11.1 11.2 Starr, Michel; Lair, Stéphane; Michaud, Sonia; Scarratt, Michael; Quilliam, Michael; Lefaivre, Denis; Robert, Michel; Wotherspoon, Andrew et al. (2017). "Multispecies mass mortality of marine fauna linked to a toxic dinoflagellate bloom". PLOS ONE 12 (5). doi:10.1371/journal.pone.0176299. ISSN 1932-6203. PMID 28472048. Bibcode: 2017PLoSO..1276299S.
- ↑ 12.0 12.1 12.2 12.3 Park, Tae Gyu; Lim, Weol Ae; Park, Young Tae; Lee, Chang Kyu; Jeong, Hae Jin (2013-12-01). "Economic impact, management and mitigation of red tides in Korea". Harmful Algae 30: S131–S143. doi:10.1016/j.hal.2013.10.012. ISSN 1568-9883. Bibcode: 2013HAlga..30S.131P. https://www.sciencedirect.com/science/article/pii/S1568988313001534.
- ↑ 13.0 13.1 13.2 13.3 13.4 "Socioeconomic – Harmful Algal Blooms" (in en-US). https://hab.whoi.edu/impacts/impacts-socioeconomic/.
- ↑ 14.0 14.1 14.2 "Assessing Environmental and Economic Impacts" (in en-US). https://coastalscience.noaa.gov/science-areas/habs/assessing-environmental-and-economic-impacts/.
- ↑ 15.00 15.01 15.02 15.03 15.04 15.05 15.06 15.07 15.08 15.09 15.10 15.11 15.12 Uwe, John; Litaker, R. Wayne; Montresor, Marina; Murray, Shauna; Brosnahan, Michael L.; Anderson, Donald M. (2014-12-01). "Formal Revision of the Alexandrium tamarense Species Complex (Dinophyceae) Taxonomy: The Introduction of Five Species with Emphasis on Molecular-based (rDNA) Classification". Protist 165 (6): 789–793. doi:10.1016/j.protis.2014.10.001. ISSN 1434-4610. PMID 25460230.
- ↑ "Alexandrium tamarense (Lebour) Balech :: AlgaeBase" (in en). https://www.algaebase.org/search/species/detail/?species_id=40299.
- ↑ Lebour, M.V (1925). The Dinoflagellates of the Northern Seas. Plymouth Marine Biol. pp. 1–250.
- ↑ Balech, Enrique (1995). The Genus Alexandrium Halim (Dinoflagellata). Sherkin Island Marine Station. pp. 24–43. ISBN 1-870492-61-7.
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 19.7 Anderson, Donald M.; Alpermann, Tilman J.; Cembella, Allan D.; Collos, Yves; Masseret, Estelle; Montresor, Marina (February 2012). "The globally distributed genus Alexandrium: multifaceted roles in marine ecosystems and impacts on human health". Harmful Algae 14: 13. doi:10.1016/j.hal.2011.10.012. ISSN 1568-9883. PMID 22308102. Bibcode: 2012HAlga..14...10A.
- ↑ 20.0 20.1 20.2 20.3 20.4 20.5 20.6 Lilly, Emily L.; Halanych, Kenneth M.; Anderson, Donald M. (December 2007). "Species boundaries and global biogeography of the Alexandrium tamarense complex (Dinophyceae)1" (in en). Journal of Phycology 43 (6): 1334–1335. doi:10.1111/j.1529-8817.2007.00420.x. ISSN 0022-3646. https://onlinelibrary.wiley.com/doi/10.1111/j.1529-8817.2007.00420.x.
- ↑ 21.0 21.1 21.2 Fritz, Lawrence; Triemer, Richard E. (December 1985). "A Rapid Simple Technique Utilizing Calcofluor White M2R for the Visualization of Dinoflagellate Thecal Plates 1" (in en). Journal of Phycology 21 (4): 662–664. doi:10.1111/j.0022-3646.1985.00662.x. ISSN 0022-3646. https://onlinelibrary.wiley.com/doi/10.1111/j.0022-3646.1985.00662.x.
- ↑ 22.0 22.1 22.2 22.3 Brown, Lyndsay; Bresnan, Eileen; Graham, Jennifer; Lacaze, Jean-Pierre; Turrell, Elizabeth; Collins, Catherine (13 December 2010). "Distribution, diversity and toxin composition of the genus Alexandrium (Dinophyceae) in Scottish waters". European Journal of Phycology 45 (4): 375–393. doi:10.1080/09670262.2010.495164. Bibcode: 2010EJPhy..45..375B.
- ↑ 23.0 23.1 23.2 Klemm, Kerstin; Cembella, Allan; Clarke, Dave; Cusack, Caroline; Arneborg, Lars; Karlson, Bengt; Liu, Ye; Naustvoll, Lars et al. (2022-11-01). "Apparent biogeographical trends in Alexandrium blooms for northern Europe: identifying links to climate change and effective adaptive actions". Harmful Algae 119. doi:10.1016/j.hal.2022.102335. ISSN 1568-9883. PMID 36344194. Bibcode: 2022HAlga.11902335K. https://www.sciencedirect.com/science/article/pii/S1568988322001639.
- ↑ 24.0 24.1 Lilly, Emily L.; Halanych, Kenneth M.; Anderson, Donald M. (December 2007). "Species boundaries and global biogeography of the Alexandrium tamarense complex (Dinophyceae)1" (in en). Journal of Phycology 43 (6): 1334–1335. doi:10.1111/j.1529-8817.2007.00420.x. ISSN 0022-3646. https://onlinelibrary.wiley.com/doi/10.1111/j.1529-8817.2007.00420.x.
- ↑ 25.0 25.1 25.2 Stüken, Anke; Orr, Russell J. S.; Kellmann, Ralf; Murray, Shauna A.; Neilan, Brett A.; Jakobsen, Kjetill S. (2011-05-18). "Discovery of Nuclear-Encoded Genes for the Neurotoxin Saxitoxin in Dinoflagellates" (in en). PLOS ONE 6 (5). doi:10.1371/journal.pone.0020096. ISSN 1932-6203. PMID 21625593. Bibcode: 2011PLoSO...620096S.
- ↑ 26.00 26.01 26.02 26.03 26.04 26.05 26.06 26.07 26.08 26.09 26.10 26.11 26.12 Anderson, Donald (January 1998). "Physiology and bloom dynamics of toxic Alexandrium species, with emphasis on life cycle transitions". Woods Hole Oceanographic Institution: 29–48. https://www.researchgate.net/publication/301838910.
- ↑ Kamiyama, T; Suzuki, T; Okumura, Y (2006-09-01). "Feeding of the tintinnid ciliate Favella taraikaensis on the toxic dinoflagellate Alexandrium tamarense and the fate of prey toxins". African Journal of Marine Science 28 (2): 343–346. doi:10.2989/18142320609504174. ISSN 1814-232X. Bibcode: 2006AfJMS..28..343K.
- ↑ Kang, Hee Chang; Jin Jeong, Hae; So Jin, Kim; You, Ji Hyun; Hee Ok, Jin (2018-09-01). "Differential feeding by common heterotrophic protists on 12 different Alexandrium species". Harmful Algae 78: 106–117. doi:10.1016/j.hal.2018.08.005. ISSN 1568-9883. PMID 30196918. Bibcode: 2018HAlga..78..106K. https://www.sciencedirect.com/science/article/pii/S1568988318301239.
- ↑ 29.0 29.1 Wohlrab, Sylke; Iversen, Morten H.; John, Uwe (2010-11-29). "A Molecular and Co-Evolutionary Context for Grazer Induced Toxin Production in Alexandrium tamarense" (in en). PLOS ONE 5 (11). doi:10.1371/journal.pone.0015039. ISSN 1932-6203. PMID 21124775. Bibcode: 2010PLoSO...515039W.
- ↑ Bergkvist, Johanna; Selander, Erik; Pavia, Henrik (2008-05-01). "Induction of toxin production in dinoflagellates: the grazer makes a difference" (in en). Oecologia 156 (1): 147–154. doi:10.1007/s00442-008-0981-6. ISSN 1432-1939. PMID 18283499. Bibcode: 2008Oecol.156..147B.
- ↑ "Paralytic Shellfish Poisoning" (in en-CA). https://www.bccdc.ca/health-info/diseases-conditions/paralytic-shellfish-poisoning.
- ↑ Franks, P. J. S.; Anderson, D. M. (1992-01-01). "Alongshore transport of a toxic phytoplankton bloom in a buoyancy current: Alexandrium tamarense in the Gulf of Maine" (in en). Marine Biology 112 (1): 153–164. doi:10.1007/BF00349739. ISSN 1432-1793.
- ↑ 33.0 33.1 Nagai, Satoshi; Matsuyama, Yukihiko; Oh, Seok-Jin; Itakura, Shigeru (February 24, 2004). "Effect of nutrients and temperature on encystment of the toxic dinoflagellate Alexandrium tamarense (Dinophyceae) isolated from Hiroshima Bay, Japan". Pankton Biology & Ecology 51 (2): 103-109. https://plankton.jp/PBE/issue/vol51_2/vol51_2_103.pdf.
- ↑ Lilly, Emily L.; Halanych, Kenneth M.; Anderson, Donald M. (2007). "Species boundaries and global biogeography of the Alexandrium tamarense complex (Dinophyceae) 1" (in en). Journal of Phycology 43 (6): 1329–1338. doi:10.1111/j.1529-8817.2007.00420.x. ISSN 0022-3646. https://onlinelibrary.wiley.com/doi/10.1111/j.1529-8817.2007.00420.x.
- ↑ Hadjadji, Imene; Frehi, Hocine; Ayada, Lembarek; Abadie, Eric; Collos, Yves (2014-02-01). "A comparative analysis of Alexandrium catenella/tamarense blooms in Annaba Bay (Algeria) and Thau lagoon (France); phosphorus limitation as a trigger". Comptes Rendus Biologies 337 (2): 117–122. doi:10.1016/j.crvi.2013.11.006. ISSN 1631-0691. https://www.sciencedirect.com/science/article/pii/S1631069113002746.
- ↑ 36.0 36.1 Natsuike, Masafumi; Matsuno, Kohei; Hirawake, Toru; Yamaguchi, Atsushi; Nishino, Shigeto; Imai, Ichiro (2017-01-01). "Possible spreading of toxic Alexandrium tamarense blooms on the Chukchi Sea shelf with the inflow of Pacific summer water due to climatic warming". Harmful Algae 61: 80–86. doi:10.1016/j.hal.2016.11.019. ISSN 1568-9883. Bibcode: 2017HAlga..61...80N. https://www.sciencedirect.com/science/article/pii/S1568988316301020.
- ↑ 37.0 37.1 37.2 37.3 "Paralytic Shellfish Poisoning" (in en-CA). https://www.bccdc.ca/health-info/diseases-conditions/paralytic-shellfish-poisoning.
- ↑ Akbar, Muhamad Afiq; Mohd Yusof, Nurul Yuziana; Tahir, Noor Idayu; Ahmad, Asmat; Usup, Gires; Sahrani, Fathul Karim; Bunawan, Hamidun (2020-02-05). "Biosynthesis of Saxitoxin in Marine Dinoflagellates: An Omics Perspective". Marine Drugs 18 (2): 103. doi:10.3390/md18020103. ISSN 1660-3397. PMID 32033403. Bibcode: 2020MarDr..18..103A.
- ↑ 39.0 39.1 Wiese, Maria; D'Agostino, Paul M.; Mihali, Troco K.; Moffitt, Michelle C.; Neilan, Brett A. (2010-07-20). "Neurotoxic alkaloids: saxitoxin and its analogs". Marine Drugs 8 (7): 2185–2211. doi:10.3390/md8072185. ISSN 1660-3397. PMID 20714432.
- ↑ 40.0 40.1 Ichimi, Kazuhiko; Suzuki, Toshiyuki; Ito, Akira (2002-07-03). "Variety of PSP toxin profiles in various culture strains of Alexandrium tamarense and change of toxin profile in natural A. tamarense population". Journal of Experimental Marine Biology and Ecology 273 (1): 51–60. doi:10.1016/S0022-0981(02)00137-5. ISSN 0022-0981. Bibcode: 2002JEMBE.273...51I. https://www.sciencedirect.com/science/article/pii/S0022098102001375.
- ↑ 41.0 41.1 41.2 41.3 41.4 Akbar, Muhamad Afiq; Mohd Yusof, Nurul Yuziana; Tahir, Noor Idayu; Ahmad, Asmat; Usup, Gires; Sahrani, Fathul Karim; Bunawan, Hamidun (2020-02-05). "Biosynthesis of Saxitoxin in Marine Dinoflagellates: An Omics Perspective". Marine Drugs 18 (2): 103. doi:10.3390/md18020103. ISSN 1660-3397. PMID 32033403. Bibcode: 2020MarDr..18..103A.
- ↑ 42.0 42.1 42.2 42.3 42.4 42.5 Jiang, Xi-Wen; Wang, Jing; Gao, Yue; Chan, Leo Lai; Lam, Paul Kwan Sing; Gu, Ji-Dong (2015-10-01). "Relationship of proteomic variation and toxin synthesis in the dinoflagellate Alexandrium tamarense CI01 under phosphorus and inorganic nitrogen limitation" (in en). Ecotoxicology 24 (7): 1744–1753. doi:10.1007/s10646-015-1513-x. ISSN 1573-3017. PMID 26239440. Bibcode: 2015Ecotx..24.1744J.
- ↑ Haines, Ricci J.; Pendleton, Laura C.; Eichler, Duane C. (2011). "Argininosuccinate synthase: at the center of arginine metabolism". International Journal of Biochemistry and Molecular Biology 2 (1): 8–23. ISSN 2152-4114. PMID 21494411.
- ↑ Dagenais-Bellefeuille, Steve; Morse, David (2013-12-04). "Putting the N in dinoflagellates". Frontiers in Microbiology 4: 369. doi:10.3389/fmicb.2013.00369. ISSN 1664-302X. PMID 24363653. Bibcode: 2013FrMic...400369D.
- ↑ Deng, Huiyun; Shang, Xinrui; Zhu, Hu; Huang, Ning; Wang, Lianghua; Sun, Mingjuan (2025-07-02). "Saxitoxin: A Comprehensive Review of Its History, Structure, Toxicology, Biosynthesis, Detection, and Preventive Implications". Marine Drugs 23 (7): 277. doi:10.3390/md23070277. ISSN 1660-3397. PMID 40710502.
- ↑ Ohtsu, Iwao; Kawano, Yusuke; Suzuki, Marina; Morigasaki, Susumu; Saiki, Kyohei; Yamazaki, Shunsuke; Nonaka, Gen; Takagi, Hiroshi (2015). "Uptake of L-cystine via an ABC transporter contributes defense of oxidative stress in the L-cystine export-dependent manner in Escherichia coli". PLOS ONE 10 (3). doi:10.1371/journal.pone.0120619. ISSN 1932-6203. PMID 25837721. Bibcode: 2015PLoSO..1020619O.
- ↑ Saldivia, Pablo; Hernández, Mauricio; Isla, Adolfo; Fritz, Rocío; Varela, Daniel; González-Jartín, Jesús M.; Figueroa, Jaime; Botana, Luis M. et al. (2023-06-01). "Proteomic and toxicological analysis of the response of dinoflagellate Alexandrium catenella to changes in NaNO3 concentration". Harmful Algae 125. doi:10.1016/j.hal.2023.102428. ISSN 1568-9883. PMID 37220981. https://www.sciencedirect.com/science/article/pii/S1568988323000549.
- ↑ Girault, Mathias; Siano, Raffaele; Labry, Claire; Latimier, Marie; Jauzein, Cécile; Beneyton, Thomas; Buisson, Lionel; Del Amo, Yolanda et al. (July 2021). "Variable inter and intraspecies alkaline phosphatase activity within single cells of revived dinoflagellates". The ISME Journal 15 (7): 2057–2069. doi:10.1038/s41396-021-00904-2. ISSN 1751-7370. PMID 33568788. Bibcode: 2021ISMEJ..15.2057G.
- ↑ Zhang, Hao; Zhou, Youping; Liu, Tian-Qi; Yin, Xi-Jie; Lin, Lin; Lin, Qiang; Wang, Da-Zhi (June 2021). "Initiation of efficient C4 pathway in response to low ambient CO2 during the bloom period of a marine dinoflagellate". Environmental Microbiology 23 (6): 3196–3211. doi:10.1111/1462-2920.15545. ISSN 1462-2920. PMID 33938118. Bibcode: 2021EnvMi..23.3196Z.
- ↑ Suzuki, Yuji; Konno, Yume; Takegahara-Tamakawa, Yuki; Miyake, Chikahiro; Makino, Amane (2022-08-01). "Effects of suppression of chloroplast phosphoglycerate kinase on photosynthesis in rice" (in en). Photosynthesis Research 153 (1): 83–91. doi:10.1007/s11120-022-00923-w. ISSN 1573-5079. PMID 35635654. Bibcode: 2022PhoRe.153...83S.
- ↑ Jiang, Jing; Zhang, Hao; Kang, Yisheng; Bina, David; Lo, Cynthia S.; Blankenship, Robert E. (2012-07-01). "Characterization of the peridinin–chlorophyll a-protein complex in the dinoflagellate Symbiodinium". Biochimica et Biophysica Acta (BBA) - Bioenergetics 1817 (7): 983–989. doi:10.1016/j.bbabio.2012.03.027. ISSN 0005-2728. https://www.sciencedirect.com/science/article/pii/S0005272812001028.
- ↑ Dukic, Emilija; van Maldegem, Kim A.; Shaikh, Kashif Mohd; Fukuda, Kento; Töpel, Mats; Solymosi, Katalin; Hellsten, Jonna; Hansen, Thomas Hesselhøj et al. (2023). "Chloroplast magnesium transporters play essential but differential roles in maintaining magnesium homeostasis". Frontiers in Plant Science 14. doi:10.3389/fpls.2023.1221436. ISSN 1664-462X. PMID 37692441. Bibcode: 2023FrPS...1421436D.
- ↑ Li, Meizhen; Shi, Xinguo; Guo, Chentao; Lin, Senjie (2016-06-01). "Phosphorus Deficiency Inhibits Cell Division But Not Growth in the Dinoflagellate Amphidinium carterae" (in English). Frontiers in Microbiology 7: 826. doi:10.3389/fmicb.2016.00826. ISSN 1664-302X. PMID 27313570. Bibcode: 2016FrMic...700826L.
Further reading
- Liang, Zhongxiu; Li, Jian; Li, Jitao; Tan, Zhijun; Ren, Hai; Zhao, Fazhen (July 8, 2014). "Toxic Dinoflagellate Alexandrium tamarense Induces Oxidative Stress and Apoptosis in Hepatopancreas of Shrimp (Fenneropenaeus chinensis)". Ocean University of China 13 (6): 1005–1011. doi:10.1007/s11802-014-2397-8. Bibcode: 2014JOUC...13.1005L.
- Lu, Yameng; Wohlrab, Sylke; Gloeckner, Gernot; Guillou, Laure; Uwe, John (November 2014). "Genomic Insights into Processes Driving the Infection of Alexandrium tamarense by the Parasitoid Amoebophrya sp.". Eukaryotic Cell 13 (11): 1439–1449. doi:10.1128/EC.00139-14. PMID 25239978.
- Zou, Cheng; Ye, Rui-Min; Zheng, Jian-Wei; Luo, Zhao-He; Gu, Hai-Feng; Yang, Wei-Dong; Li, Hong-Ye; Liu, Jie-Sheng (15 December 2014). "Molecular phylogeny and PSP toxin profile of the Alexandrium tamarense species complex along the coast of China". Marine Pollution Bulletin 89 (1–2): 209–219. doi:10.1016/j.marpolbul.2014.09.056. PMID 25444620. Bibcode: 2014MarPB..89..209Z.
Wikidata ☰ Q2834271 entry
 |
 KSF
KSF