Benzoxazinone biosynthesis
Topic: Biology
 From HandWiki - Reading time: 2 min
From HandWiki - Reading time: 2 min
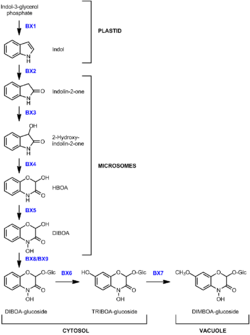
The biosynthesis of benzoxazinone, a cyclic hydroxamate and a natural insecticide, has been well-characterized in maize and related grass species.[1] In maize, genes in the pathway are named using the symbol bx. Maize Bx-genes are tightly linked, a feature that has been considered uncommon for plant genes of a biosynthetic pathways. Especially notable are genes encoding the different enzymatic functions BX1, BX2 and BX8 and which are found within about 50 kilobases.[1][2] Results from wheat and rye indicate that the cluster is an ancient feature.[3] In wheat the cluster is split into two parts. The wheat genes Bx1 and Bx2 are located in close proximity on chromosome 4 and wheat Bx3, Bx4 and Bx5 map to the short arm of chromosome 5; an additional Bx3 copy was detected on the long arm of chromosome 5B.[3] Recently, additional biosynthetic clusters have been detected in other plants for other biosynthetic pathways and this organization might be common in plants.[4]
Maize genes
The bx1 gene encodes a protein, BX1, that forms indol from indol-3-glycerol phosphate in the plastid. It is the first step in the pathway and determines much of the natural variation in levels of DIMBOA in maize.[5] The next steps in the pathway occur in the endoplasmic reticulum, also referred to as the microsomes in cell fractionation experiments, and are carried by proteins encoded by genes bx2, bx3, bx4, and bx5.
References
- ↑ 1.0 1.1 Frey, M; Chomet, P; Glawischnig, E; Stettner, C; Grün, S; Winklmair, A; Eisenreich, W; Bacher, A et al. (Aug 1, 1997). "Analysis of a chemical plant defense mechanism in grasses.". Science 277 (5326): 696–9. doi:10.1126/science.277.5326.696. PMID 9235894.
- ↑ Frey, M; Kliem, R; Saedler, H; Gierl, A (Jan 6, 1995). "Expression of a cytochrome P450 gene family in maize.". Molecular & General Genetics 246 (1): 100–9. doi:10.1007/bf00290138. PMID 7823905.
- ↑ 3.0 3.1 Nomura, T; Ishihara, A; Imaishi, H; Ohkawa, H; Endo, TR; Iwamura, H (Sep 2003). "Rearrangement of the genes for the biosynthesis of benzoxazinones in the evolution of Triticeae species.". Planta 217 (5): 776–82. doi:10.1007/s00425-003-1040-5. PMID 12734755.
- ↑ Osbourn, A (Oct 2010). "Gene clusters for secondary metabolic pathways: an emerging theme in plant biology.". Plant Physiology 154 (2): 531–5. doi:10.1104/pp.110.161315. PMID 20921179.
- ↑ Butrón, A; Chen, YC; Rottinghaus, GE; McMullen, MD (Feb 2010). "Genetic variation at bx1 controls DIMBOA content in maize.". Theoretical and Applied Genetics 120 (4): 721–34. doi:10.1007/s00122-009-1192-1. PMID 19911162. https://digital.csic.es/bitstream/10261/24875/1/Genetic%20Variation%20at%20bx1%20Controls%20DIMBOA%20Content%20in%20Maize.pdf.
 |
 KSF
KSF