1,N6-Ethanoadenine
Topic: Chemistry
 From HandWiki - Reading time: 2 min
From HandWiki - Reading time: 2 min
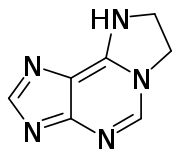
| |
| Names | |
|---|---|
| IUPAC name
8,9-dihydro-7H-imidazo[2,1-f]purine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| MeSH | 1,N(6)-ethanoadenine |
PubChem CID
|
|
| |
| |
| Properties | |
| C7H7N5 | |
| Molar mass | 161.168 g·mol−1 |
| Related compounds | |
Related compounds
|
3,N4-Ethenocytosine; 1,N6-Ethenoadenine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
1,N6-Ethanoadenine or epsilonA is a tricyclic derivative of adenine, where an ethylene bridge connect the amine group to the adjacent carbon on the six member ring, to add another five membered ring. This kind of modification can take place as a mutation of DNA. As a DNA modification it is called an etheno (ε) DNA adduct.[1] Chloroacetaldehyde[1] and 1,3-bis(2-chloroethyl)-1-nitrosourea (BCNU) can react with adenine to form 1,N6-ethanoadenine.[2]
Escherichia coli can fix these modifications using AlkB protein.[1]
Another way to produce it is via cyclisation of N6-hydroxyethyladenine with DIAD and triphenylphosphine in the Mitsunobu reaction in a THF/MeCN solvent or by reacting with SOCl2.[3]
References
- ↑ 1.0 1.1 1.2 MacIejewska, A. M.; Sokolowska, B.; Nowicki, A.; Kusmierek, J. T. (2011). "The role of AlkB protein in repair of 1,N6-ethenoadenine in Escherichia coli cells". Mutagenesis 26 (3): 401–406. doi:10.1093/mutage/geq107. PMID 21193516.
- ↑ Hang, B (29 October 2003). "Miscoding properties of 1,N6-ethanoadenine, a DNA adduct derived from reaction with the antitumor agent 1,3-bis(2-chloroethyl)-1-nitrosourea". Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 531 (1–2): 191–203. doi:10.1016/j.mrfmmm.2003.07.006. PMID 14637255. https://digital.library.unt.edu/ark:/67531/metadc785755/.
- ↑ Šimůnková, Naděžda; Tobrman, Tomáš; Eigner, Václav; Dvořák, Dalimil (November 2017). "A Study on the Intramolecular Mitsunobu Reaction of N6-(ω-Hydroxyalkyl)adenines". Journal of Heterocyclic Chemistry 54 (6): 3565–3573. doi:10.1002/jhet.2982.
 |
Licensed under CC BY-SA 3.0 | Source: https://handwiki.org/wiki/Chemistry:1,N6-Ethanoadenine1 views | ↧ Download this article as ZWI file
 KSF
KSF