18-Hydroxy-11-deoxycorticosterone
Topic: Chemistry
 From HandWiki - Reading time: 5 min
From HandWiki - Reading time: 5 min
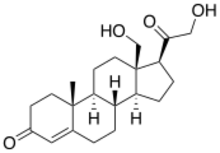
| |
| Names | |
|---|---|
| IUPAC name
18,21-Dihydroxypregn-4-ene-3,20-dione
| |
| Systematic IUPAC name
(1S,3aS,3bR,9aR,9bS,11aR)-1-(Hydroxyacetyl)-11a-(hydroxymethyl)-9a-methyl-1,2,3,3a,3b,4,5,8,9,9a,9b,10,11,11a-tetradecahydro-7H-cyclopenta[a]phenanthren-7-one | |
| Other names
18,21-dihydroxy-4-pregnene-3,20-dione
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
| MeSH | 18-hydroxydeoxycorticosterone |
PubChem CID
|
|
| |
| |
| Properties | |
| C21H30O4 | |
| Molar mass | 346.5 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
18-Hydroxy-11-deoxycorticosterone (also known as 18-OH-DOC, 18,21-dihydroxyprogesterone, and 18,21-dihydroxypregn-4-ene-3,20-dione) is an endogenous steroid and a mineralocorticoid. It is a hydroxylated metabolite of 11-deoxycorticosterone.[1]
In rats, conversion of 11-deoxycorticosterone into 18-OH-DOC is catalyzed by the CYP11B3 enzyme.[2]
In humans, 18-OH-DOC is a weak mineralocorticoid.[3] It may be increased in 17α-hydroxylase (CYP17A1) deficiency,[4] in aldosterone synthase (CYP11B2) deficiency,[5] in primary aldosteronism, and may also indicate a histologic variant of the aldosteronoma.[4] Excessive secretion of 18-OH-DOC can cause mineralocorticoid excess syndrome, although these cases are very rare.[6]
References
- ↑ "18,19-Dihydroxydeoxycorticosterone, a new metabolite produced from 18-hydroxydeoxycorticosterone by cytochrome P-450(11) beta. Chemical synthesis and structural analysis by 1H NMR". Biochemistry 23 (12): 2558–64. June 1984. doi:10.1021/bi00307a004. PMID 6466598.
- ↑ "Cloning and expression of the rat adrenal cytochrome P-450 11B3 (CYP11B3) enzyme cDNA: preferential 18-hydroxylation over 11 beta-hydroxylation of DOC". Molecular and Cellular Endocrinology 114 (1–2): 137–45. October 1995. doi:10.1016/0303-7207(95)03653-o. PMID 8674838.
- ↑ "The regulation of plasma 18-hydroxy 11-deoxycorticosterone in man". The Journal of Clinical Investigation 58 (1): 221–9. July 1976. doi:10.1172/JCI108453. PMID 180059.
- ↑ 4.0 4.1 "Adrenocortical factors in hypertension. I. Significance of 18-hydroxy-11-deoxycorticosterone". The American Journal of Cardiology 38 (6): 814–24. November 1976. doi:10.1016/0002-9149(76)90360-x. PMID 187051.
- ↑ "Chromatographic system for the simultaneous measurement of plasma 18-hydroxy-11-deoxycorticosterone and 18-hydroxycorticosterone by radioimmunoassay: reference data for neonates and infants and its application in aldosterone-synthase deficiency". Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences 785 (2): 293–301. March 2003. doi:10.1016/s1570-0232(02)00921-2. PMID 12554142.
- ↑ "Effect of uni-adrenalectomy on blood pressure in a patient with excessive adrenal 18-hydroxy-11-deoxycorticosterone production bilaterally". Internal Medicine (Tokyo, Japan) 42 (6): 507–12. June 2003. doi:10.2169/internalmedicine.42.507. PMID 12857050.
External links
- 18-hydroxydeoxycorticosterone at the US National Library of Medicine Medical Subject Headings (MeSH)
 |
Licensed under CC BY-SA 3.0 | Source: https://handwiki.org/wiki/Chemistry:18-Hydroxy-11-deoxycorticosterone1 | Status: cached on February 10 2026 11:38:48↧ Download this article as ZWI file
 KSF
KSF