Amphetamine
Topic: Chemistry
 From HandWiki - Reading time: 113 min
From HandWiki - Reading time: 113 min
Amphetamine[note 1] is a central nervous system (CNS) stimulant that is used in the treatment of attention deficit hyperactivity disorder (ADHD) and narcolepsy; it is also used to treat binge eating disorder in the form of lisdexamfetamine. Historically, it has been used to treat nasal congestion and depression. Amphetamine is also used as an athletic performance enhancer and cognitive enhancer, and recreationally as an aphrodisiac and euphoriant. It is a prescription drug in many countries, and unauthorized possession and distribution of amphetamine are often tightly controlled due to the significant health risks associated with recreational use.[sources 1]
Amphetamine was discovered as a chemical in 1887 by Lazăr Edeleanu, and then as a drug in the late 1920s. It exists as two enantiomers:[note 2] levoamphetamine and dextroamphetamine. The first amphetamine pharmaceutical was Benzedrine. Pharmaceutical amphetamine is prescribed as racemic amphetamine, Adderall,[note 3] dextroamphetamine, or lisdexamfetamine. Amphetamine increases monoamine and excitatory neurotransmission in the brain, with its most pronounced effects targeting the norepinephrine and dopamine neurotransmitter systems.[sources 2]
At therapeutic doses, amphetamine causes emotional and cognitive effects such as euphoria, change in desire for sex, increased wakefulness, and improved cognitive control. It induces physical effects such as improved reaction time, fatigue resistance, decreased appetite, elevated heart rate, and increased muscle strength. Larger doses of amphetamine may impair cognitive function and induce rapid muscle breakdown. Addiction is a serious risk with heavy recreational amphetamine use, but is unlikely to occur from long-term medical use at therapeutic doses. Very high doses can result in psychosis, which rarely occurs at therapeutic doses even during long-term use. Recreational doses are generally much larger than prescribed therapeutic doses and carry a far greater risk of serious side effects.[sources 3]
Amphetamine belongs to the phenethylamine class. It is also the parent compound of its own structural class, the substituted amphetamines,[note 4] which includes prominent substances such as bupropion, cathinone, MDMA, and methamphetamine. As a member of the phenethylamine class, amphetamine is also chemically related to the naturally occurring trace amine neuromodulators, specifically phenethylamine and N-methylphenethylamine, both of which are produced within the human body. Phenethylamine is the parent compound of amphetamine, while N-methylphenethylamine is a positional isomer of amphetamine that differs only in the placement of the methyl group.[sources 4]
Uses
Medical
Amphetamine is used to treat attention deficit hyperactivity disorder (ADHD), narcolepsy, and, in the form of lisdexamfetamine, binge eating disorder.[35][16][17] It is sometimes prescribed off-label for its past medical indications, particularly for depression and chronic pain.[35][36]
ADHD
Long-term amphetamine exposure at sufficiently high doses in some animal species is known to produce abnormal dopamine system development or nerve damage,[37][38] but, in humans with ADHD, long-term use of pharmaceutical amphetamines at therapeutic doses appears to improve brain development and nerve growth.[39][40][41] Reviews of magnetic resonance imaging (MRI) studies suggest that long-term treatment with amphetamine decreases abnormalities in brain structure and function found in subjects with ADHD, and improves function in several parts of the brain, such as the right caudate nucleus of the basal ganglia.[39][40][41]
Reviews of clinical stimulant research have established the safety and effectiveness of long-term continuous amphetamine use for the treatment of ADHD.[27][42][43] Randomized controlled trials of continuous stimulant therapy for the treatment of ADHD spanning 2 years have demonstrated treatment effectiveness and safety.[27][42] Two reviews have indicated that long-term continuous stimulant therapy for ADHD is effective for reducing the core symptoms of ADHD (i.e., hyperactivity, inattention, and impulsivity), enhancing quality of life and academic achievement, and producing improvements in a large number of functional outcomes[note 5] across 9 categories of outcomes related to academics, antisocial behavior, driving, non-medicinal drug use, obesity, occupation, self-esteem, service use (i.e., academic, occupational, health, financial, and legal services), and social function.[27][43] Additionally, a 2024 meta-analytic systematic review reported moderate improvements in quality of life when amphetamine treatment is used for ADHD.[45] One review highlighted a nine-month randomized controlled trial of amphetamine treatment for ADHD in children that found an average increase of 4.5 IQ points, continued increases in attention, and continued decreases in disruptive behaviors and hyperactivity.[42] Another review indicated that, based upon the longest follow-up studies conducted to date, lifetime stimulant therapy that begins during childhood is continuously effective for controlling ADHD symptoms and reduces the risk of developing a substance use disorder as an adult.[27]
Models of ADHD suggest that it is associated with functional impairments in some of the brain's neurotransmitter systems;[46] these functional impairments involve impaired dopamine neurotransmission in the mesocorticolimbic projection and norepinephrine neurotransmission in the noradrenergic projections from the locus coeruleus to the prefrontal cortex.[46] Stimulants like methylphenidate and amphetamine are effective in treating ADHD because they increase neurotransmitter activity in these systems.[7][46][47] Approximately 80% of those who use these stimulants see improvements in ADHD symptoms.[48] Children with ADHD who use stimulant medications generally have better relationships with peers and family members, perform better in school, are less distractible and impulsive, and have longer attention spans.[49][50] The Cochrane reviews[note 6] on the treatment of ADHD in children, adolescents, and adults with pharmaceutical amphetamines stated that short-term studies have demonstrated that these drugs decrease the severity of symptoms, but they have higher discontinuation rates than non-stimulant medications due to their adverse side effects.[52][53] However, a 2025 meta-analytic systematic review of 113 randomized controlled trials found that stimulant medications were the only intervention with robust short-term efficacy, and were associated with lower all-cause treatment discontinuation rates than non-stimulant medications (e.g., atomoxetine).[note 7][54] A Cochrane review on the treatment of ADHD in children with tic disorders such as Tourette syndrome indicated that stimulants in general do not make tics worse, but high doses of dextroamphetamine could exacerbate tics in some individuals.[55]
Binge eating disorder
Binge eating disorder (BED) is characterized by recurrent and persistent episodes of compulsive binge eating.[56] These episodes are often accompanied by marked distress and a feeling of loss of control over eating.[56] The pathophysiology of BED is not fully understood, but it is believed to involve dysfunctional dopaminergic reward circuitry along the cortico-striatal-thalamic-cortical loop.[57][58] As of July 2024, lisdexamfetamine is the only USFDA- and TGA-approved pharmacotherapy for BED.[17][59] Evidence suggests that lisdexamfetamine's treatment efficacy in BED is underpinned at least in part by a psychopathological overlap between BED and ADHD, with the latter conceptualized as a cognitive control disorder that also benefits from treatment with lisdexamfetamine.[57][58]
Lisdexamfetamine's therapeutic effects for BED primarily involve direct action in the central nervous system after conversion to its pharmacologically active metabolite, dextroamphetamine.[59] Dextroamphetamine increases dopaminergic and noradrenergic neurotransmission in prefrontal cortex, a region of the brain that regulates cognitive control of behavior.[57][59] Similar to its therapeutic effect in ADHD, dextroamphetamine enhances cognitive control and may reduce impulsivity in patients with BED by enhancing the cognitive processes responsible for overriding prepotent feeding responses that precede binge eating episodes.[57][60][61] Dextroamphetamine is also a full agonist of trace amine-associated receptor 1 (TAAR1), a G-protein coupled receptor that regulates monoaminergic systems in the brain;[62][63] activation of TAAR1 may restore impaired dopaminergic signaling in the prefrontal cortex and thereby correct deficits in inhibitory control associated with binge eating behaviors.[63] Alongside its effect on cognitive control, appetite suppression is a common side effect of lisdexamfetamine and is associated with reduced food intake.[61][64] Amphetamines are believed to dampen the sensation of hunger through actions in the lateral hypothalamus;[65] this is thought to involve increased catecholaminergic signaling and the induction of cocaine- and amphetamine-regulated transcript (CART), a hypothalamic neuropeptide involved the regulation of food intake.[65] While appetite suppression may influence the magnitude of reductions in food intake, three medical reviews indicate that lisdexamfetamine's enhancement of cognitive control is the primary therapeutic effect for reducing binge-eating symptoms and is likely required for addressing the disorder's underlying psychopathology.[57][64][61] This view is supported by the failure of anti-obesity medications and other appetite suppressants to significantly reduce BED symptom severity, despite their ability to produce weight loss.[64]
Medical reviews of randomized controlled trials have demonstrated that lisdexamfetamine, at doses between 50–70 mg, is safe and effective for the treatment of moderate-to-severe BED in adults.[sources 5] These reviews suggest that lisdexamfetamine is persistently effective at treating BED and is associated with significant reductions in the number of binge eating days and binge eating episodes per week.[sources 5] Furthermore, a meta-analytic systematic review highlighted an open-label, 12-month extension safety and tolerability study that reported lisdexamfetamine remained effective at reducing the number of binge eating days for the duration of the study.[61] In addition, both a review and a meta-analytic systematic review found lisdexamfetamine to be superior to placebo in several secondary outcome measures, including persistent binge eating cessation, reduction of obsessive-compulsive related binge eating symptoms, reduction of body-weight, and reduction of triglycerides.[58][61] Lisdexamfetamine, like all pharmaceutical amphetamines, has direct appetite suppressant effects that may be therapeutically useful in both BED and its comorbidities.[17][61] Based on reviews of neuroimaging studies involving BED-diagnosed participants, therapeutic neuroplasticity in dopaminergic and noradrenergic pathways from long-term use of lisdexamfetamine may be implicated in lasting improvements in the regulation of eating behaviors that are observed.[17][59][61]
Narcolepsy
Narcolepsy is a chronic sleep-wake disorder that is associated with excessive daytime sleepiness, cataplexy, and sleep paralysis.[67] Patients with narcolepsy are diagnosed as either type 1 or type 2, with only the former presenting cataplexy symptoms.[68] Type 1 narcolepsy results from the loss of approximately 70,000 orexin-releasing neurons in the lateral hypothalamus, leading to significantly reduced cerebrospinal orexin levels;[69][70] this reduction is a diagnostic biomarker for type 1 narcolepsy.[68] Lateral hypothalamic orexin neurons innervate every component of the ascending reticular activating system (ARAS), which includes noradrenergic, dopaminergic, histaminergic, and serotonergic nuclei that promote wakefulness.[70][71]
Amphetamine's therapeutic mode of action in narcolepsy primarily involves increasing monoamine neurotransmitter activity in the ARAS.[69][72][73] This includes noradrenergic neurons in the locus coeruleus, dopaminergic neurons in the ventral tegmental area, histaminergic neurons in the tuberomammillary nucleus, and serotonergic neurons in the dorsal raphe nucleus.[71][73] Dextroamphetamine, the more dopaminergic enantiomer of amphetamine, is particularly effective at promoting wakefulness because dopamine release has the greatest influence on cortical activation and cognitive arousal, relative to other monoamines.[69][74] In contrast, levoamphetamine may have a greater effect on cataplexy, a symptom more sensitive to the effects of norepinephrine and serotonin.[69] Noradrenergic and serotonergic nuclei in the ARAS are involved in the regulation of the REM sleep cycle and function as "REM-off" cells, with amphetamine's effect on norepinephrine and serotonin contributing to the suppression of REM sleep and a possible reduction of cataplexy at high doses.[69][68][71]
The American Academy of Sleep Medicine (AASM) 2021 clinical practice guideline conditionally recommends dextroamphetamine for the treatment of both type 1 and type 2 narcolepsy.[75] Treatment with pharmaceutical amphetamines is generally less preferred relative to other stimulants (e.g., modafinil) and is considered a third-line treatment option.[30][76][77] Medical reviews indicate that amphetamine is safe and effective for the treatment of narcolepsy.[69][30][75] Amphetamine appears to be most effective at improving symptoms associated with hypersomnolence, with three reviews finding clinically significant reductions in daytime sleepiness in patients with narcolepsy.[69][30][75] Additionally, these reviews suggest that amphetamine may dose-dependently improve cataplexy symptoms.[69][30][75] However, the quality of evidence for these findings is low and is consequently reflected in the AASM's conditional recommendation for dextroamphetamine as a treatment option for narcolepsy.[75]
Enhancing performance
Cognitive performance
In 2015, a systematic review and a meta-analysis of high quality clinical trials found that, when used at low (therapeutic) doses, amphetamine produces modest yet unambiguous improvements in cognition, including working memory, long-term episodic memory, inhibitory control, and some aspects of attention, in normal healthy adults;[78][79] these cognition-enhancing effects of amphetamine are known to be partially mediated through the indirect activation of both dopamine D1 receptor and α2-adrenergic receptor in the prefrontal cortex.[7][78] A systematic review from 2014 found that low doses of amphetamine also improve memory consolidation, in turn leading to improved recall of information.[80] Therapeutic doses of amphetamine also enhance cortical network efficiency, an effect which mediates improvements in working memory in all individuals.[7][81] Amphetamine and other ADHD stimulants also improve task saliency (motivation to perform a task) and increase arousal (wakefulness), in turn promoting goal-directed behavior.[7][82][83] Stimulants such as amphetamine can improve performance on difficult and boring tasks and are used by some students as a study and test-taking aid.[7][83][84] Based upon studies of self-reported illicit stimulant use, 5–35% of college students use diverted ADHD stimulants, which are primarily used for enhancement of academic performance rather than as recreational drugs.[85][86][87] However, high amphetamine doses that are above the therapeutic range can interfere with working memory and other aspects of cognitive control.[7][83]
Physical performance
Amphetamine is used by some athletes for its psychological and athletic performance-enhancing effects, such as increased endurance and alertness;[8][23] however, non-medical amphetamine use is prohibited at sporting events that are regulated by collegiate, national, and international anti-doping agencies.[88][89] In healthy people at oral therapeutic doses, amphetamine has been shown to increase muscle strength, acceleration, athletic performance in anaerobic conditions, and endurance (i.e., it delays the onset of fatigue), while improving reaction time.[8][90][91] Amphetamine improves endurance and reaction time primarily through reuptake inhibition and release of dopamine in the central nervous system.[90][91][92] Amphetamine and other dopaminergic drugs also increase power output at fixed levels of perceived exertion by overriding a "safety switch", allowing the core temperature limit to increase in order to access a reserve capacity that is normally off-limits.[91][93][94] At therapeutic doses, the adverse effects of amphetamine do not impede athletic performance;[8][90] however, at much higher doses, amphetamine can induce effects that severely impair performance, such as rapid muscle breakdown and elevated body temperature.[9][90]
Recreational
Amphetamine, specifically the more dopaminergic dextrorotatory enantiomer (dextroamphetamine), is also used recreationally as a euphoriant and aphrodisiac, and like other amphetamines; is used as a club drug for its energetic and euphoric high. Dextroamphetamine (d-amphetamine) is considered to have a high potential for misuse in a recreational manner since individuals typically report feeling euphoric, more alert, and more energetic after taking the drug.[95][96][97] A notable part of the 1960s mod subculture in the UK was recreational amphetamine use, which was used to fuel all-night dances at clubs like Manchester's Twisted Wheel. Newspaper reports described dancers emerging from clubs at 5 a.m. with dilated pupils.[98] Mods used the drug for stimulation and alertness, which they viewed as different from the intoxication caused by alcohol and other drugs.[98] Dr. Andrew Wilson argues that for a significant minority, "amphetamines symbolised the smart, on-the-ball, cool image" and that they sought "stimulation not intoxication [...] greater awareness, not escape" and "confidence and articulacy" rather than the "drunken rowdiness of previous generations."[98] Dextroamphetamine's dopaminergic (rewarding) properties affect the mesocorticolimbic circuit; a group of neural structures responsible for incentive salience (i.e., "wanting"; desire or craving for a reward and motivation), positive reinforcement and positively-valenced emotions, particularly ones involving pleasure.[99] Large recreational doses of dextroamphetamine may produce symptoms of dextroamphetamine overdose.[97] Recreational users sometimes open dexedrine capsules and crush the contents in order to insufflate (snort) it or subsequently dissolve it in water and inject it.[97] Immediate-release formulations have higher potential for abuse via insufflation (snorting) or intravenous injection due to a more favorable pharmacokinetic profile and easy crushability (especially tablets).[100][101] Injection into the bloodstream can be dangerous because insoluble fillers within the tablets can block small blood vessels.[97] Chronic overuse of dextroamphetamine can lead to severe drug dependence, resulting in withdrawal symptoms when drug use stops.[97]
Contraindications
According to the International Programme on Chemical Safety (IPCS) and the U.S. Food and Drug Administration (FDA),[note 8] amphetamine is contraindicated in people with a history of drug abuse,[note 9] cardiovascular disease, severe agitation, or severe anxiety.[16][9][103] It is also contraindicated in individuals with advanced arteriosclerosis (hardening of the arteries), glaucoma (increased eye pressure), hyperthyroidism (excessive production of thyroid hormone), or moderate to severe hypertension.[16][9][103] These agencies indicate that people who have experienced allergic reactions to other stimulants or who are taking monoamine oxidase inhibitors (MAOIs) should not take amphetamine,[16][9][103] although safe concurrent use of amphetamine and monoamine oxidase inhibitors has been documented.[104][105] These agencies also state that anyone with anorexia nervosa, bipolar disorder, depression, hypertension, liver or kidney problems, mania, psychosis, Raynaud's phenomenon, seizures, thyroid problems, tics, or Tourette syndrome should monitor their symptoms while taking amphetamine.[9][103] Evidence from human studies indicates that therapeutic amphetamine use does not cause developmental abnormalities in the fetus or newborns (i.e., it is not a human teratogen), but amphetamine abuse does pose risks to the fetus.[103] Amphetamine has also been shown to pass into breast milk, so the IPCS and the FDA advise mothers to avoid breastfeeding when using it.[9][103] Due to the potential for reversible growth impairments,[note 10] the FDA advises monitoring the height and weight of children and adolescents prescribed an amphetamine pharmaceutical.[9]
Adverse effects
The adverse side effects of amphetamine are many and varied, and the amount of amphetamine used is the primary factor in determining the likelihood and severity of adverse effects.[9][23] Amphetamine products such as Adderall, Vyvanse, and their generic equivalents are currently approved by the U.S. FDA for long-term therapeutic use.[19][9] Recreational use of amphetamine generally involves much larger doses, which have a greater risk of serious adverse drug effects than dosages used for therapeutic purposes.[23]
Physical
Cardiovascular side effects can include hypertension or hypotension from a vasovagal response, Raynaud's phenomenon (reduced blood flow to the hands and feet), and tachycardia (increased heart rate).[107][23][106] Sexual side effects in males may include erectile dysfunction, frequent erections, or prolonged erections.[9] Gastrointestinal side effects may include abdominal pain, constipation, diarrhea, and nausea.[35][9][108] Other potential physical side effects include appetite loss, blurred vision, dry mouth, excessive grinding of the teeth, nosebleed, profuse sweating, rhinitis medicamentosa (drug-induced nasal congestion), reduced seizure threshold, tics (a type of movement disorder), and weight loss.[sources 6] Dangerous physical side effects are rare at typical pharmaceutical doses.[23]
Amphetamine stimulates the medullary respiratory centers, producing faster and deeper breaths.[23] In a normal person at therapeutic doses, this effect is usually not noticeable, but when respiration is already compromised, it may be evident.[23] Amphetamine also induces contraction in the urinary bladder sphincter, the muscle which controls urination, which can result in difficulty urinating.[23] This effect can be useful in treating bed wetting and loss of bladder control.[23] The effects of amphetamine on the gastrointestinal tract are unpredictable.[23] If intestinal activity is high, amphetamine may reduce gastrointestinal motility (the rate at which content moves through the digestive system);[23] however, amphetamine may increase motility when the smooth muscle of the tract is relaxed.[23] Amphetamine also has a slight analgesic effect and can enhance the pain relieving effects of opioids.[110]
FDA-commissioned studies from 2011 indicate that in children, young adults, and adults there is no association between serious adverse cardiovascular events (sudden death, heart attack, and stroke) and the medical use of amphetamine or other ADHD stimulants.[sources 7] These findings were subsequently corroborated by a 2022 meta-analysis that sampled nearly four million participants, which found no association between therapeutic use of amphetamine and the development of cardiovascular disease in any age group.[115] However, amphetamine pharmaceuticals are contraindicated in individuals with preexisting cardiovascular disease.[sources 8]
Psychological
At normal therapeutic doses, the most common psychological side effects of amphetamine include increased alertness, apprehension, concentration, initiative, self-confidence and sociability, mood swings (elated mood followed by mildly depressed mood), insomnia or wakefulness, and decreased sense of fatigue.[9][23] Less common side effects include anxiety, change in libido, grandiosity, irritability, repetitive or obsessive behaviors, and restlessness;[sources 9] these effects depend on the user's personality and current mental state.[23] Amphetamine psychosis (e.g., delusions and paranoia) can occur in heavy users.[9][24][25] Although very rare, this psychosis can also occur at therapeutic doses during long-term therapy.[9][25][26] According to the FDA, "there is no systematic evidence" that stimulants produce aggressive behavior or hostility.[9]
Amphetamine has also been shown to produce a conditioned place preference in humans taking therapeutic doses,[52][117] meaning that individuals acquire a preference for spending time in places where they have previously used amphetamine.[117][118]
Reinforcement disorders
Addiction
| Addiction and dependence glossary[118][119][120][121] | |
|---|---|
| |
Template:Transcription factor glossary
Signaling cascade in the nucleus accumbens that results in amphetamine addiction
|
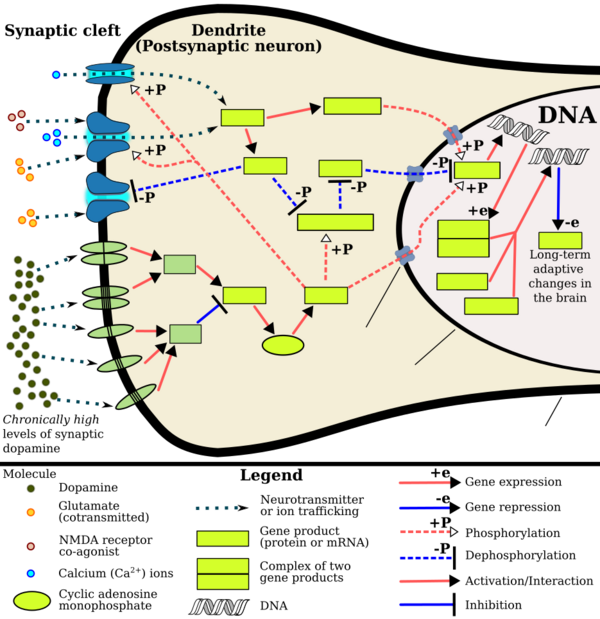
Addiction is a serious risk with heavy recreational amphetamine use, but is unlikely to occur from long-term medical use at therapeutic doses;[28][29][30] in fact, lifetime stimulant therapy for ADHD that begins during childhood reduces the risk of developing substance use disorders as an adult.[27] Pathological overactivation of the mesolimbic pathway, a dopamine pathway that connects the ventral tegmental area to the nucleus accumbens, plays a central role in amphetamine addiction.[128][129] Individuals who frequently self-administer high doses of amphetamine have a high risk of developing an amphetamine addiction, since chronic use at high doses gradually increases the level of accumbal ΔFosB, a "molecular switch" and "master control protein" for addiction.[119][130][131] Once nucleus accumbens ΔFosB is sufficiently overexpressed, it begins to increase the severity of addictive behavior (i.e., compulsive drug-seeking) with further increases in its expression.[130][132] While there are currently no effective drugs for treating amphetamine addiction, regularly engaging in sustained aerobic exercise appears to reduce the risk of developing such an addiction.[133][134] Exercise therapy improves clinical treatment outcomes and may be used as an adjunct therapy with behavioral therapies for addiction.[133][135][sources 10]
Biomolecular mechanisms
Chronic use of amphetamine at excessive doses causes alterations in gene expression in the mesocorticolimbic projection, which arise through transcriptional and epigenetic mechanisms.[131][136][137] The most important transcription factors[note 11] that produce these alterations are Delta FBJ murine osteosarcoma viral oncogene homolog B (ΔFosB), cAMP response element binding protein (CREB), and nuclear factor-kappa B (NF-κB).[131] ΔFosB is the most significant biomolecular mechanism in addiction because ΔFosB overexpression (i.e., an abnormally high level of gene expression which produces a pronounced gene-related phenotype) in the D1-type medium spiny neurons in the nucleus accumbens is necessary and sufficient[note 12] for many of the neural adaptations and regulates multiple behavioral effects (e.g., reward sensitization and escalating drug self-administration) involved in addiction.[119][130][131] Once ΔFosB is sufficiently overexpressed, it induces an addictive state that becomes increasingly more severe with further increases in ΔFosB expression.[119][130] It has been implicated in addictions to alcohol, cannabinoids, cocaine, methylphenidate, nicotine, opioids, phencyclidine, propofol, and substituted amphetamines, among others.[sources 11]
ΔJunD, a transcription factor, and G9a, a histone methyltransferase enzyme, both oppose the function of ΔFosB and inhibit increases in its expression.[119][131][141] Sufficiently overexpressing ΔJunD in the nucleus accumbens with viral vectors can completely block many of the neural and behavioral alterations seen in chronic drug abuse (i.e., the alterations mediated by ΔFosB).[131] Similarly, accumbal G9a hyperexpression results in markedly increased histone 3 lysine residue 9 dimethylation (H3K9me2) and blocks the induction of ΔFosB-mediated neural and behavioral plasticity by chronic drug use,[sources 12] which occurs via H3K9me2-mediated repression of transcription factors for ΔFosB and H3K9me2-mediated repression of various ΔFosB transcriptional targets (e.g., CDK5).[131][141][142] ΔFosB also plays an important role in regulating behavioral responses to natural rewards, such as palatable food, sex, and exercise.[132][131][145] Since both natural rewards and addictive drugs induce the expression of ΔFosB (i.e., they cause the brain to produce more of it), chronic acquisition of these rewards can result in a similar pathological state of addiction.[132][131] Consequently, ΔFosB is the most significant factor involved in both amphetamine addiction and amphetamine-induced sexual addictions, which are compulsive sexual behaviors that result from excessive sexual activity and amphetamine use.[132][146][147] These sexual addictions are associated with a dopamine dysregulation syndrome which occurs in some patients taking dopaminergic drugs.[132][145]
The effects of amphetamine on gene regulation are both dose- and route-dependent.[137] Most of the research on gene regulation and addiction is based upon animal studies with intravenous amphetamine administration at very high doses.[137] The few studies that have used equivalent (weight-adjusted) human therapeutic doses and oral administration show that these changes, if they occur, are relatively minor.[137] This suggests that medical use of amphetamine does not significantly affect gene regulation.[137]
Pharmacological treatments
As of December 2019,[update] there is no effective pharmacotherapy for amphetamine addiction.[148][149][150] Reviews from 2015 and 2016 indicated that TAAR1-selective agonists have significant therapeutic potential as a treatment for psychostimulant addictions;[22][151] however, as of February 2016,[update] the only compounds which are known to function as TAAR1-selective agonists are experimental drugs.[22][151] Amphetamine addiction is largely mediated through increased activation of dopamine receptors and co-localized NMDA receptors[note 13] in the nucleus accumbens;[129] magnesium ions inhibit NMDA receptors by blocking the receptor calcium channel.[129][152] One review suggested that, based upon animal testing, pathological (addiction-inducing) psychostimulant use significantly reduces the level of intracellular magnesium throughout the brain.[129] Supplemental magnesium[note 14] treatment has been shown to reduce amphetamine self-administration (i.e., doses given to oneself) in humans, but it is not an effective monotherapy for amphetamine addiction.[129]
A systematic review and meta-analysis from 2019 assessed the efficacy of 17 different pharmacotherapies used in randomized controlled trials (RCTs) for amphetamine and methamphetamine addiction;[149] it found only low-strength evidence that methylphenidate might reduce amphetamine or methamphetamine self-administration.[149] There was low- to moderate-strength evidence of no benefit for most of the other medications used in RCTs, which included antidepressants (bupropion, mirtazapine, sertraline), antipsychotics (aripiprazole), anticonvulsants (topiramate, baclofen, gabapentin), naltrexone, varenicline, citicoline, ondansetron, prometa, riluzole, atomoxetine, dextroamphetamine, and modafinil.[149]
Behavioral treatments
A 2018 systematic review and network meta-analysis of 50 trials involving 12 different psychosocial interventions for amphetamine, methamphetamine, or cocaine addiction found that combination therapy with both contingency management and community reinforcement approach had the highest efficacy (i.e., abstinence rate) and acceptability (i.e., lowest dropout rate).[153] Other treatment modalities examined in the analysis included monotherapy with contingency management or community reinforcement approach, cognitive behavioral therapy, 12-step programs, non-contingent reward-based therapies, psychodynamic therapy, and other combination therapies involving these.[153]
Additionally, research on the neurobiological effects of physical exercise suggests that daily aerobic exercise, especially endurance exercise (e.g., marathon running), prevents the development of drug addiction and is an effective adjunct therapy (i.e., a supplemental treatment) for amphetamine addiction.[sources 10] Exercise leads to better treatment outcomes when used as an adjunct treatment, particularly for psychostimulant addictions.[133][135][154] In particular, aerobic exercise decreases psychostimulant self-administration, reduces the reinstatement (i.e., relapse) of drug-seeking, and induces increased dopamine receptor D2 (DRD2) density in the striatum.[132][154] This is the opposite of pathological stimulant use, which induces decreased striatal DRD2 density.[132] One review noted that exercise may also prevent the development of a drug addiction by altering ΔFosB or c-Fos immunoreactivity in the striatum or other parts of the reward system.[134]
| Form of neuroplasticity or behavioral plasticity |
Type of reinforcer | Sources | |||||
|---|---|---|---|---|---|---|---|
| Opiates | Psychostimulants | High fat or sugar food | Sexual intercourse | Physical exercise (aerobic) |
Environmental enrichment | ||
| ΔFosB expression in nucleus accumbens D1-type MSNs |
↑ | ↑ | ↑ | ↑ | ↑ | ↑ | [132] |
| Behavioral plasticity | |||||||
| Escalation of intake | Yes | Yes | Yes | [132] | |||
| Psychostimulant cross-sensitization |
Yes | Not applicable | Yes | Yes | Attenuated | Attenuated | [132] |
| Psychostimulant self-administration |
↑ | ↑ | ↓ | ↓ | ↓ | [132] | |
| Psychostimulant conditioned place preference |
↑ | ↑ | ↓ | ↑ | ↓ | ↑ | [132] |
| Reinstatement of drug-seeking behavior | ↑ | ↑ | ↓ | ↓ | [132] | ||
| Neurochemical plasticity | |||||||
| CREB phosphorylation in the nucleus accumbens |
↓ | ↓ | ↓ | ↓ | ↓ | [132] | |
| Sensitized dopamine response in the nucleus accumbens |
No | Yes | No | Yes | [132] | ||
| Altered striatal dopamine signaling | ↓DRD2, ↑DRD3 | ↑DRD1, ↓DRD2, ↑DRD3 | ↑DRD1, ↓DRD2, ↑DRD3 | ↑DRD2 | ↑DRD2 | [132] | |
| Altered striatal opioid signaling | No change or ↑μ-opioid receptors |
↑μ-opioid receptors ↑κ-opioid receptors |
↑μ-opioid receptors | ↑μ-opioid receptors | No change | No change | [132] |
| Changes in striatal opioid peptides | ↑dynorphin No change: enkephalin |
↑dynorphin | ↓enkephalin | ↑dynorphin | ↑dynorphin | [132] | |
| Mesocorticolimbic synaptic plasticity | |||||||
| Number of dendrites in the nucleus accumbens | ↓ | ↑ | ↑ | [132] | |||
| Dendritic spine density in the nucleus accumbens |
↓ | ↑ | ↑ | [132] | |||
Dependence and withdrawal
Drug tolerance develops rapidly in amphetamine abuse (i.e., recreational amphetamine use), so periods of extended abuse require increasingly larger doses of the drug in order to achieve the same effect.[155][156] According to a Cochrane review on withdrawal in individuals who compulsively use amphetamine and methamphetamine, "when chronic heavy users abruptly discontinue amphetamine use, many report a time-limited withdrawal syndrome that occurs within 24 hours of their last dose."[157] This review noted that withdrawal symptoms in chronic, high-dose users are frequent, occurring in roughly 88% of cases, and persist for 3–4 weeks with a marked "crash" phase occurring during the first week.[157] Amphetamine withdrawal symptoms can include anxiety, drug craving, depressed mood, fatigue, increased appetite, increased movement or decreased movement, lack of motivation, sleeplessness or sleepiness, and lucid dreams.[157] The review indicated that the severity of withdrawal symptoms is positively correlated with the age of the individual and the extent of their dependence.[157]
According to a 2025 review, the discontinuation of amphetamine at therapeutic doses does not typically result in withdrawal symptoms.[158] Discontinuation of amphetamine may cause a recurrence of ADHD symptoms due to the cessation of treatment-related drug effects.[158] In cases where mild withdrawal symptoms do occur, they can be avoided by tapering the dose.[35] Unlike amphetamine abuse, where drug tolerance necessitates escalating doses to achieve the same effect, tolerance to clinically relevant doses of amphetamine plateaus after the initial titration period, and "drug holidays" (i.e., temporary treatment discontinuation) are not required to prevent the development of tolerance.[158][159] A 2026 systematic review on ADHD medication tolerance found evidence of tolerance developing to some affective state effects of amphetamine (e.g., positive mood and drug liking) within two weeks of daily use, but little to no evidence of long-term tolerance to its therapeutic or cardiovascular effects.[159]
Overdose
An amphetamine overdose can lead to many different symptoms, but is rarely fatal with appropriate care.[35][103][160] The severity of overdose symptoms increases with dosage and decreases with drug tolerance to amphetamine.[23][103] Tolerant individuals have been known to take as much as 5 grams of amphetamine in a day, which is roughly 100 times the maximum daily therapeutic dose.[103] Symptoms of a moderate and extremely large overdose are listed below; fatal amphetamine poisoning usually also involves convulsions and coma.[9][23] In 2013, overdose on amphetamine, methamphetamine, and other compounds implicated in an "amphetamine use disorder" resulted in an estimated 3,788 deaths worldwide (3,425–4,145 deaths, 95% confidence).[note 15][161]
| System | Minor or moderate overdose[9][23][103] | Severe overdose[sources 13] |
|---|---|---|
| Cardiovascular |
|
|
| Central nervous system |
|
|
| Musculoskeletal |
| |
| Respiratory |
|
|
| Urinary |
|
|
| Other |
|
|
Toxicity
In rodents and primates, sufficiently high doses of amphetamine cause dopaminergic neurotoxicity, or damage to dopamine neurons, which is characterized by dopamine terminal degeneration and reduced transporter and receptor function.[163][164] There is no evidence that amphetamine is directly neurotoxic in humans.[165][166] However, large doses of amphetamine may indirectly cause dopaminergic neurotoxicity as a result of hyperpyrexia, the excessive formation of reactive oxygen species, and increased autoxidation of dopamine.[sources 14] Animal models of neurotoxicity from high-dose amphetamine exposure indicate that the occurrence of hyperpyrexia (i.e., core body temperature ≥ 40 °C) is necessary for the development of amphetamine-induced neurotoxicity.[164] Prolonged elevations of brain temperature above 40 °C likely promote the development of amphetamine-induced neurotoxicity in laboratory animals by facilitating the production of reactive oxygen species, disrupting cellular protein function, and transiently increasing blood–brain barrier permeability.[164]
Psychosis
An amphetamine overdose can result in a stimulant psychosis that may involve a variety of symptoms, such as delusions and paranoia.[24][25] A Cochrane review on treatment for amphetamine, dextroamphetamine, and methamphetamine psychosis states that about 5–15% of users fail to recover completely.[24][169] According to the same review, there is at least one trial that shows antipsychotic medications effectively resolve the symptoms of acute amphetamine psychosis.[24] Psychosis rarely arises from therapeutic use.[9][25][26]
Drug interactions
Many types of substances are known to interact with amphetamine, resulting in altered drug action or metabolism of amphetamine, the interacting substance, or both.[9] Inhibitors of enzymes that metabolize amphetamine (e.g., CYP2D6 and FMO3) will prolong its elimination half-life, meaning that its effects will last longer.[170][9] Amphetamine also interacts with MAOIs, particularly monoamine oxidase A inhibitors, since both MAOIs and amphetamine increase plasma catecholamines (i.e., norepinephrine and dopamine);[9] therefore, concurrent use of both is dangerous.[9] Amphetamine modulates the activity of most psychoactive drugs. In particular, amphetamine may decrease the effects of sedatives and depressants and increase the effects of stimulants and antidepressants.[9][110] Amphetamine may also decrease the effects of antihypertensives and antipsychotics due to its effects on blood pressure and dopamine respectively.[9][110] Zinc supplementation may reduce the minimum effective dose of amphetamine when it is used for the treatment of ADHD.[note 16][175] Norepinephrine reuptake inhibitors (NRIs) like atomoxetine prevent norepinephrine release induced by amphetamines and have been found to reduce the stimulant, euphoriant, and sympathomimetic effects of dextroamphetamine in humans.[176][177][178]
In general, there is no significant interaction when consuming amphetamine with food, but the pH of gastrointestinal content and urine affects the absorption and excretion of amphetamine, respectively.[9] Acidic substances reduce the absorption of amphetamine and increase urinary excretion, and alkaline substances do the opposite.[9] Due to the effect pH has on absorption, amphetamine also interacts with gastric acid reducers such as proton pump inhibitors and H2 antihistamines, which increase gastrointestinal pH (i.e., make it less acidic).[9]
Pharmacology
Pharmacodynamics
Pharmacodynamics of amphetamine in a dopamine neuron
|
Amphetamine exerts its behavioral effects by altering the use of monoamines as neuronal signals in the brain, primarily in catecholamine neurons in the reward and executive function pathways of the brain.[21][47] The concentrations of the main neurotransmitters involved in reward circuitry and executive functioning, dopamine and norepinephrine, increase dramatically in a dose-dependent manner by amphetamine because of its effects on monoamine transporters.[21][47][179] The reinforcing and motivational salience-promoting effects of amphetamine are due mostly to enhanced dopaminergic activity in the mesolimbic pathway.[7] The euphoric and locomotor-stimulating effects of amphetamine are dependent upon the magnitude and speed by which it increases synaptic dopamine and norepinephrine concentrations in the striatum.[4]
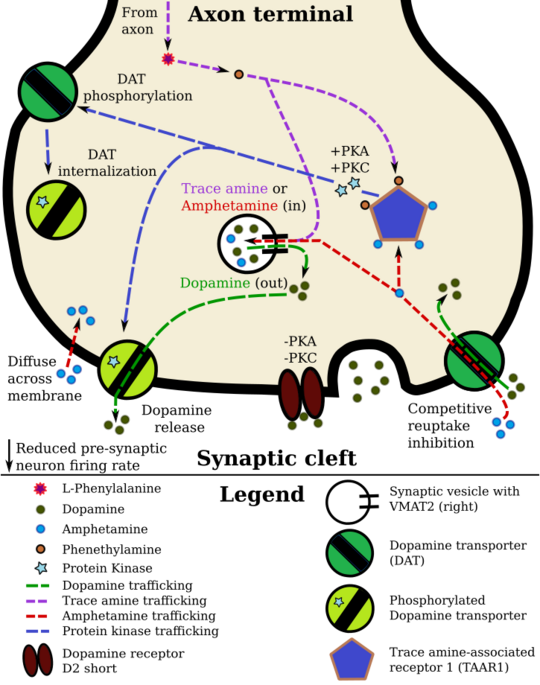
Amphetamine potentiates monoaminergic neurotransmission primarily by entering axon terminals either through active transport by monoamine transporters (DAT, NET, and SERT) or by passive diffusion across neuronal membranes.[185][186] The uptake of amphetamine through these transporters produces competitive reuptake inhibition, since amphetamine competes with endogenous monoamines for transporter-mediated clearance from the synaptic cleft.[185][187] Once inside the neuronal cytosol, amphetamine can interact with its receptor protein targets to initiate signaling cascades that activate intracellular effectors which regulate monoamine transporter function and surface expression at the plasma membrane.[107][186][187] The effect of amphetamine on monoamine transporters appears to involve phosphorylation, in which activated protein kinases attach a phosphate group to a specific amino acid residue on the transporter protein.[188][187] Depending on the protein kinase involved and the residue(s) phosphorylated, phosphorylation can shift transporter function into an efflux-permissive state that causes the reverse transport of cytosolic monoamines into the synaptic cleft, or it can promote transporter internalization, whereby phosphorylated transporters are withdrawn from the plasma membrane and lower total reuptake capacity (i.e., non-competitive reuptake inhibition).[186][188] As of January 2026, protein kinase A (PKA), protein kinase C (PKC), Ca2+/calmodulin-dependent protein kinase II (CaMKII), and Ras homolog family member A (RhoA) have all been demonstrated experimentally to regulate monoamine transporter function activity following amphetamine exposure.[186][189]
Amphetamine has been identified as a full agonist of trace amine-associated receptor 1 (TAAR1), a Gs-coupled and G13-coupled G protein-coupled receptor (GPCR) discovered in 2001, which is important for regulation of brain monoamines.[21][190][189] Several reviews have linked amphetamine's agonism at TAAR1 to modulation of monoamine transporter function and subsequent neurotransmitter efflux and reuptake inhibition at monoaminergic synapses.[sources 15] Upon activation by amphetamine, TAAR1 can couple to the Gs alpha subunit and increase intracellular cAMP production via adenylyl cyclase activation, which triggers PKA- and PKC-mediated transporter phosphorylation.[21][62][193] When TAAR1 couples to the G13 alpha subunit, RhoA activity increases near the endoplasmic reticulum and leads to the downstream internalization of monoamine transporters;[186][189] TAAR1-dependent RhoA signaling has also been shown to internalize EAAT3, a neuronal glutamate transporter expressed in some monoaminergic neurons.[186][189] Monoamine autoreceptors (e.g., D2 short, presynaptic α2, and presynaptic 5-HT1A) have the opposite effect of TAAR1, and together these receptors provide a regulatory system for monoamines.[21][22][190] Notably, amphetamine and trace amines possess high binding affinities for TAAR1, but not for monoamine autoreceptors.[21][22] Although TAAR1 is implicated in amphetamine-induced transporter phosphorylation, the magnitude of TAAR1-mediated monoamine release in humans remains unclear.[sources 15][190] Beyond its Gs- and G13-coupled receptor-mediated effects on monoamine transporter function, TAAR1 also opens G protein-coupled inwardly rectifying potassium channels through a separate pathway, an action that reduces neuronal firing.[194][195][190]
Amphetamine is also a substrate for the vesicular monoamine transporters VMAT1 and VMAT2.[180][196] Under normal conditions, VMAT2 transports cytosolic monoamines into synaptic vesicles for storage and later exocytotic release. When amphetamine accumulates in the presynaptic terminal, it collapses the vesicular pH gradient and releases vesicular monoamines into the neuronal cytosol.[180][196] These displaced monoamines expand the cytosolic pool available for reverse transport, thereby increasing the capacity for monoamine efflux beyond that achieved by amphetamine-mediated transporter phosphorylation alone.[187][197][196] Although VMAT2 is recognized as a major target in amphetamine-induced monoamine release at higher doses, some reviews have challenged its relevance at therapeutic doses.[185][196][198]
In addition to membrane and vesicular monoamine transporters, amphetamine also inhibits SLC1A1, SLC22A3, and SLC22A5.[sources 16] SLC1A1 is excitatory amino acid transporter 3 (EAAT3), a glutamate transporter located in neurons, SLC22A3 is an extraneuronal monoamine transporter that is present in astrocytes, and SLC22A5 is a high-affinity carnitine transporter.[sources 16] Amphetamine is known to strongly induce cocaine- and amphetamine-regulated transcript (CART) gene expression,[2][204] a neuropeptide involved in feeding behavior, stress, and reward, which induces observable increases in neuronal development and survival in vitro.[2][205][206] The CART receptor has yet to be identified, but there is significant evidence that CART binds to a unique Gi/Go-coupled GPCR.[206][207] Amphetamine also inhibits monoamine oxidases at very high doses, resulting in less monoamine and trace amine metabolism and consequently higher concentrations of synaptic monoamines.[198][1][208] In humans, the only post-synaptic receptor at which amphetamine is known to bind is the 5-HT1A receptor, where it acts as an agonist with low micromolar affinity.[209][210]
The full profile of amphetamine's short-term drug effects in humans is mostly derived through increased cellular communication or neurotransmission of dopamine,[21] serotonin,[21] norepinephrine,[21] epinephrine,[179] histamine,[179] CART peptides,[2][204] endogenous opioids,[211][212][213] adrenocorticotropic hormone,[214][215] corticosteroids,[214][215] and glutamate,[183][200] which it affects through interactions with CART, 5-HT1A, EAAT3, TAAR1, VMAT1, VMAT2, and possibly other biological targets.[sources 17] Amphetamine also activates seven human carbonic anhydrase enzymes, several of which are expressed in the human brain.[216]
Dextroamphetamine displays higher binding affinity for DAT than levoamphetamine, whereas both enantiomers share comparable affinity at NET;[185] Consequently, dextroamphetamine produces greater CNS stimulation than levoamphetamine, roughly three to four times more, but levoamphetamine has slightly stronger cardiovascular and peripheral effects.[185][23] Dextroamphetamine is also a more potent agonist of TAAR1 than levoamphetamine.[217][218]
Dopamine
In certain brain regions, amphetamine increases the concentration of dopamine in the synaptic cleft by modulating DAT through several overlapping processes.[197][186][62] Amphetamine can enter the presynaptic neuron either through DAT or, to a lesser extent, by diffusing across the neuronal membrane directly.[21][186] As a consequence of DAT uptake, amphetamine produces competitive reuptake inhibition at the transporter.[185][187] Upon entering the presynaptic neuron, amphetamine provokes the release of Ca2+ from endoplasmic reticulum stores, an effect that raises intracellular calcium to levels sufficient for downstream kinase-dependent signalling.[188][189][197] In parallel, amphetamine also increases intracellular cAMP, which activates protein kinase A (PKA) and protein kinase C (PKC), whilst elevated intracellular Ca2+ activates PKC alone.[193][186][62] Phosphorylation of DAT by either kinase induces transporter internalization (non-competitive reuptake inhibition), but PKC-mediated phosphorylation alone induces the reversal of dopamine transport through DAT (i.e., dopamine efflux).[186][188]
TAAR1 has been identified as a biomolecular target of amphetamine that initiates some of amphetamine's kinase-dependent signaling cascades.[193][186][62] When TAAR1 signals via Gs-coupled receptors, intracellular cAMP increases through adenylyl cyclase activation and activates PKA and PKC, in turn phosphorylating DAT.[62][193] TAAR1 also couples G-protein alpha subunit G13;[219] when triggered by amphetamine, this pathway activates Ras homolog A (RhoA) and its downstream protein kinase, Rho-associated coiled-coil kinase (ROCK), an effect that internalizes both DAT and the neuronal glutamate transporter EAAT3.[note 17][220][186] Transporter internalization via TAAR1's G13-coupled pathway is transient because Gs-cAMP-PKA signaling functionally inhibits RhoA's downstream activity;[219][221] once intracellular cAMP sufficiently accumulates, PKA is activated and phosphorylates RhoA, thereby terminating ROCK-mediated transporter internalization.[220][186] In addition to presynaptic actions that regulate DAT, TAAR1 activation exerts a somatodendritic inhibitory influence on midbrain dopamine neurons by reducing their firing rate via G protein-coupled inwardly-rectifying potassium channels, an effect that is expected to reduce action potential-dependent (vesicular) dopamine release into the synaptic cleft.[195][190][181]
Amphetamine's effect on intracellular calcium is associated with DAT phosphorylation through Ca2+/calmodulin-dependent protein kinase II alpha (CAMKIIα), in turn producing dopamine efflux.[186][188][184] Because conventional PKC isoforms can be activated by Ca2+ and diacylglycerol, elevated intracellular calcium can promote PKC-dependent DAT phosphorylation independent of TAAR1.[197]
| Biological target of amphetamine | Initial effector / G-protein | Second messenger(s) | Secondary effector protein kinase |
Phosphorylated transporter | Effect on transporter function | Effect on neurotransmission | Sources |
|---|---|---|---|---|---|---|---|
| Unidentified | Unidentified intracellular effector | IP3-mediated intracellular Ca2+ release | CAMKIIα | DAT | Reverse transport of dopamine | Dopamine efflux into synaptic cleft | [197][188] |
| TAAR1 | G13 | RhoA–GTP | ROCK† | DAT | Transporter internalization | Dopamine reuptake inhibition | [186][221][220] |
| TAAR1 | G13 | RhoA–GTP | ROCK† | EAAT3 | Transporter internalization | Glutamate reuptake inhibition | [186][190][220] |
| TAAR1 | Gs | ↑ cAMP | PKA | DAT | Transporter internalization | Dopamine reuptake inhibition | [21][186][62] |
| TAAR1 | Gs | ↑ cAMP | PKC | DAT | Reverse transport of dopamine Transporter internalization |
Dopamine efflux into synaptic cleft Dopamine reuptake inhibition |
[21][62][193] |
| Unidentified | Unidentified intracellular effector | IP3/DAG pathway‡ | PKC | DAT | Reverse transport of dopamine Transporter internalization |
Dopamine efflux into synaptic cleft Dopamine reuptake inhibition |
[197][188] |
| †ROCK-mediated transporter internalization is transient due to the inactivation of RhoA (which activates ROCK) by PKA.
‡IP3 binds to its receptors on the endoplasmic reticulum to release intracellular Ca2+ stores, and together with diacylglycerol activates conventional PKC isoforms. |
[186][188][220] | ||||||
Amphetamine is also a substrate for the presynaptic vesicular monoamine transporter, VMAT2.[180] Following amphetamine uptake at VMAT2, amphetamine induces the collapse of the vesicular pH gradient, which results in a dose-dependent release of dopamine molecules from synaptic vesicles into the cytosol via dopamine efflux through VMAT2.[180][196] Subsequently, the cytosolic dopamine molecules are released from the presynaptic neuron into the synaptic cleft via reverse transport at DAT.[187][180][196]
Norepinephrine
Similar to dopamine, amphetamine dose-dependently increases the level of synaptic norepinephrine, the direct precursor of epinephrine.[185] Amphetamine is believed to affect norepinephrine analogously to dopamine.[62] In other words, amphetamine induces competitive NET reuptake inhibition, TAAR1-mediated non-competitive reuptake inhibition and reverse transport at phosphorylated NET, CAMKIIα-mediated NET efflux independent of TAAR1, and norepinephrine release from VMAT2.[186][62][187] In locus coeruleus norepinephrine neurons, TAAR1-dependent RhoA signaling promotes EAAT3 internalization and subsequent glutamate reuptake inhibition.[189]
Serotonin
Amphetamine exerts analogous, yet less pronounced, effects on serotonin as on dopamine and norepinephrine.[21] Amphetamine affects serotonin via VMAT2 inhibition and SERT phosphorylation.[62] Like dopamine, amphetamine has low, micromolar affinity at the human 5-HT1A receptor.[209][210]
Other neurotransmitters, peptides, hormones, and enzymes
| Enzyme | KA (nM) | Sources |
|---|---|---|
| hCA4 | 94 | [216] |
| hCA5A | 810 | [216][222] |
| hCA5B | 2560 | [216] |
| hCA7 | 910 | [216][222] |
| hCA12 | 640 | [216] |
| hCA13 | 24100 | [216] |
| hCA14 | 9150 | [216] |
Acute amphetamine administration in humans increases endogenous opioid release in several brain structures in the reward system.[211][212][213] Extracellular levels of glutamate, the primary excitatory neurotransmitter in the brain, have been shown to increase in the striatum following exposure to amphetamine.[183] This increase in extracellular glutamate presumably occurs via the amphetamine-induced internalization of EAAT3, a glutamate reuptake transporter in some dopamine and norepinephrine neurons.[183][200] This internalization is mediated by RhoA activation and its downstream effector ROCK, a process that is TAAR1-dependent.[186][189] Amphetamine also induces the selective release of histamine from mast cells and efflux from histaminergic neurons through VMAT2.[179] Acute amphetamine administration can also increase adrenocorticotropic hormone and corticosteroid levels in blood plasma by stimulating the hypothalamic–pituitary–adrenal axis.[16][214][215]
In December 2017, the first study assessing the interaction between amphetamine and human carbonic anhydrase enzymes was published;[216] of the eleven carbonic anhydrase enzymes it examined, it found that amphetamine potently activates seven, four of which are highly expressed in the human brain, with low nanomolar through low micromolar activating effects.[216] Based upon preclinical research, cerebral carbonic anhydrase activation has cognition-enhancing effects;[223] but, based upon the clinical use of carbonic anhydrase inhibitors, carbonic anhydrase activation in other tissues may be associated with adverse effects, such as ocular activation exacerbating glaucoma.[223]
Sex-dependent differences
Clinical research indicates that the pharmacological effects of amphetamine may vary depending on sex and menstrual cycle phase, possibly due to fluctuations in estrogen and progesterone.[sources 18] In menstruating individuals, subjective and behavioral responses to amphetamine are heightened during the follicular phase (i.e., when estrogen levels are higher), and reduced during the luteal phase (i.e., when progesterone is elevated).[224][225][227] Reviews of human studies have also noted that men typically report stronger positive subjective responses to amphetamine compared to women tested during the luteal phase, whereas these sex differences are absent when women are tested during the follicular phase;[sources 18] subjective responses to amphetamine appear to correlate positively with plasma or salivary estrogen concentrations.[224][227] Moreover, neuroimaging studies have reported significant sex differences in the neural response to amphetamine in humans, including differences in dopamine release within the striatum and other brain regions.[228][229]
Preclinical studies have also produced findings of sex-dependent differences in drug response to amphetamine.[229][230] In contrast to human studies, adult female rats exhibit markedly greater dopamine release in the nucleus accumbens and more pronounced behavioral effects from amphetamine administration relative to males, effects that may be modulated by fluctuating estradiol levels across the estrous cycle or more broadly by adult gonadal hormones.[228][229][230]
Some evidence suggests that amphetamine may be more strongly modulated by ovarian sex hormones than other psychostimulants such as methylphenidate, which may result in relatively greater variability in drug response across the menstrual cycle.[224][226] Although preliminary observational evidence suggests potential benefit from adjusting amphetamine doses according to menstrual cycle phases, randomized controlled trials have not evaluated this practice.[224][225][158]
Pharmacokinetics
The oral bioavailability of amphetamine varies with gastrointestinal pH;[9] it is well absorbed from the gut, and bioavailability is typically 90%.[231] Amphetamine is a weak base with a pKa of 9.9;[232] consequently, when the pH is basic, more of the drug is in its lipid soluble free base form, and more is absorbed through the lipid-rich cell membranes of the gut epithelium.[232][9] Conversely, an acidic pH means the drug is predominantly in a water-soluble cationic (salt) form, and less is absorbed.[232] Approximately 20% of amphetamine circulating in the bloodstream is bound to plasma proteins.[2] Following absorption, amphetamine readily distributes into most tissues in the body, with high concentrations occurring in cerebrospinal fluid and brain tissue.[233]
The half-lives of amphetamine enantiomers differ and vary with urine pH.[232] At normal urine pH, the half-lives of dextroamphetamine and levoamphetamine are 9–11 hours and 11–14 hours, respectively.[232] Highly acidic urine will reduce the enantiomer half-lives to 7 hours;[233] highly alkaline urine will increase the half-lives up to 34 hours.[233] The immediate-release and extended release variants of salts of both isomers reach peak plasma concentrations at 3 hours and 7 hours post-dose respectively.[232] Amphetamine is eliminated via the kidneys, with 30–40% of the drug being excreted unchanged at normal urinary pH.[232] When the urinary pH is basic, amphetamine is in its free base form, so less is excreted.[232] When urine pH is abnormal, the urinary recovery of amphetamine may range from a low of 1% to a high of 75%, depending mostly upon whether urine is too basic or acidic, respectively.[232] Following oral administration, amphetamine appears in urine within 3 hours.[233] Roughly 90% of ingested amphetamine is eliminated 3 days after the last oral dose.[233]
Lisdexamfetamine is a prodrug of dextroamphetamine.[234][235] It is not as sensitive to pH as amphetamine when being absorbed in the gastrointestinal tract.[235] Following absorption into the blood stream, lisdexamfetamine is completely converted by red blood cells to dextroamphetamine and the amino acid L-lysine by hydrolysis via undetermined aminopeptidase enzymes.[235][234][236] This is the rate-limiting step in the bioactivation of lisdexamfetamine.[234] The elimination half-life of lisdexamfetamine is generally less than 1 hour.[235][234] Due to the necessary conversion of lisdexamfetamine into dextroamphetamine, levels of dextroamphetamine with lisdexamfetamine peak about one hour later than with an equivalent dose of immediate-release dextroamphetamine.[234][236] Presumably due to its rate-limited activation by red blood cells, intravenous administration of lisdexamfetamine shows greatly delayed time to peak and reduced peak levels compared to intravenous administration of an equivalent dose of dextroamphetamine.[234] The pharmacokinetics of lisdexamfetamine are similar regardless of whether it is administered orally, intranasally, or intravenously.[234][236] Hence, in contrast to dextroamphetamine, parenteral use does not enhance the subjective effects of lisdexamfetamine.[234][236] Because of its behavior as a prodrug and its pharmacokinetic differences, lisdexamfetamine has a longer duration of therapeutic effect than immediate-release dextroamphetamine and shows reduced misuse potential.[234][236]
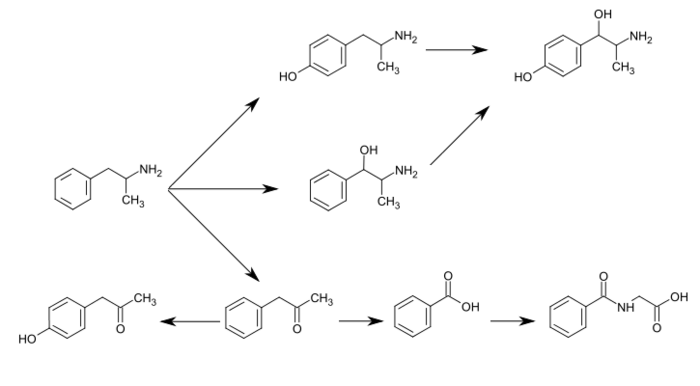
CYP2D6, dopamine β-hydroxylase (DBH), flavin-containing monooxygenase 3 (FMO3), butyrate-CoA ligase (XM-ligase), and glycine N-acyltransferase (GLYAT) are the enzymes known to metabolize amphetamine or its metabolites in humans.[sources 19] Amphetamine has a variety of excreted metabolic products, including 4-hydroxyamphetamine, 4-hydroxynorephedrine, 4-hydroxyphenylacetone, N-hydroxyamphetamine, benzoic acid, hippuric acid, norephedrine, and phenylacetone.[232][237][238] Among these metabolites, the active sympathomimetics are 4-hydroxyamphetamine,[239] 4-hydroxynorephedrine,[240] norephedrine,[241] and N-hydroxyamphetamine.[242][243] The main metabolic pathways involve aromatic para-hydroxylation, aliphatic alpha- and beta-hydroxylation, N-oxidation, N-dealkylation, and deamination.[232][244] The known metabolic pathways, detectable metabolites, and metabolizing enzymes in humans include the following:
Metabolic pathways of amphetamine in humans[sources 19]
|
Pharmacomicrobiomics
The human metagenome (i.e., the genetic composition of an individual and all microorganisms that reside on or within the individual's body) varies considerably between individuals.[252][253] Since the total number of microbial and viral cells in the human body (over 100 trillion) greatly outnumbers human cells (tens of trillions),[note 20][252][254] there is considerable potential for interactions between drugs and an individual's microbiome, including: drugs altering the composition of the human microbiome, drug metabolism by microbial enzymes modifying the drug's pharmacokinetic profile, and microbial drug metabolism affecting a drug's clinical efficacy and toxicity profile.[252][253][255] The field that studies these interactions is known as pharmacomicrobiomics.[252]
Similar to most biomolecules and other orally administered xenobiotics (i.e., drugs), amphetamine is predicted to undergo promiscuous metabolism by human gastrointestinal microbiota (primarily bacteria) prior to absorption into the blood stream.[255] The first amphetamine-metabolizing microbial enzyme, tyramine oxidase from a strain of E. coli commonly found in the human gut, was identified in 2019.[255] This enzyme was found to metabolize amphetamine, tyramine, and phenethylamine with roughly the same binding affinity for all three compounds.[255]
Related endogenous compounds
Amphetamine has a very similar structure and function to the endogenous trace amines, which are naturally occurring neuromodulator molecules produced in the human body and brain.[21][32][256] Among this group, the most closely related compounds are phenethylamine, the parent compound of amphetamine, and N-methylphenethylamine, a structural isomer of amphetamine (i.e., it has an identical molecular formula).[21][32][257] In humans, phenethylamine is produced directly from L-phenylalanine by the aromatic amino acid decarboxylase (AADC) enzyme, which converts L-DOPA into dopamine as well.[32][257] In turn, N-methylphenethylamine is metabolized from phenethylamine by phenylethanolamine N-methyltransferase, the same enzyme that metabolizes norepinephrine into epinephrine.[32][257] Like amphetamine, both phenethylamine and N-methylphenethylamine regulate monoamine neurotransmission via TAAR1;[21][256][257] unlike amphetamine, both of these substances are broken down by monoamine oxidase B, and therefore have a shorter half-life than amphetamine.[32][257]
Chemistry
Racemic amphetamine
|
Phenyl-2-nitropropene (right cups)
Amphetamine is a methyl homolog of the mammalian neurotransmitter phenethylamine with the chemical formula C
9H
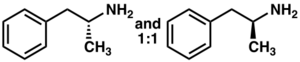
13N. The carbon atom adjacent to the primary amine is a stereogenic center, and amphetamine is composed of a racemic 1:1 mixture of two enantiomers.[2] This racemic mixture can be separated into its optical isomers:[note 21] levoamphetamine and dextroamphetamine.[2] At room temperature, the pure free base of amphetamine is a mobile, colorless, and volatile liquid with a characteristically strong amine odor, and acrid, burning taste.[258] Frequently prepared solid salts of amphetamine include amphetamine adipate,[259] aspartate,[9] hydrochloride,[260] phosphate,[261] saccharate,[9] sulfate,[9] and tannate.[262] Dextroamphetamine sulfate is the most common enantiopure salt.[33] Amphetamine is also the parent compound of its own structural class, which includes a number of psychoactive derivatives.[31][2] In organic chemistry, amphetamine is an excellent chiral ligand for the stereoselective synthesis of 1,1'-bi-2-naphthol.[263]
Substituted derivatives
The substituted derivatives of amphetamine, or "substituted amphetamines", are a broad range of chemicals that contain amphetamine as a "backbone";[31][34][264] specifically, this chemical class includes derivative compounds that are formed by replacing one or more hydrogen atoms in the amphetamine core structure with substituents.[31][34][265] The class includes amphetamine itself, stimulants like methamphetamine, serotonergic empathogens like MDMA, and decongestants like ephedrine, among other subgroups.[31][34][264]
Synthesis
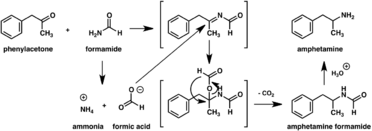
Since the first preparation was reported in 1887,[266] numerous synthetic routes to amphetamine have been developed.[267][268] The most common route of both legal and illicit amphetamine synthesis employs a non-metal reduction known as the Leuckart reaction (method 1).[33][269] In the first step, a reaction between phenylacetone and formamide, either using additional formic acid or formamide itself as a reducing agent, yields N-formylamphetamine. This intermediate is then hydrolyzed using hydrochloric acid, and subsequently basified, extracted with organic solvent, concentrated, and distilled to yield the free base. The free base is then dissolved in an organic solvent, sulfuric acid added, and amphetamine precipitates out as the sulfate salt.[269][270]
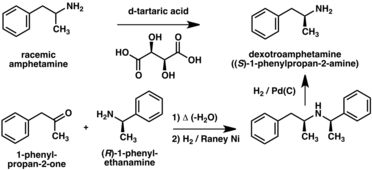
A number of chiral resolutions have been developed to separate the two enantiomers of amphetamine.[267] For example, racemic amphetamine can be treated with d-tartaric acid to form a diastereoisomeric salt which is fractionally crystallized to yield dextroamphetamine.[271] Chiral resolution remains the most economical method for obtaining optically pure amphetamine on a large scale.[272] In addition, several enantioselective syntheses of amphetamine have been developed. In one example, optically pure (R)-1-phenyl-ethanamine is condensed with phenylacetone to yield a chiral Schiff base. In the key step, this intermediate is reduced by catalytic hydrogenation with a transfer of chirality to the carbon atom alpha to the amino group. Cleavage of the benzylic amine bond by hydrogenation yields optically pure dextroamphetamine.[272]
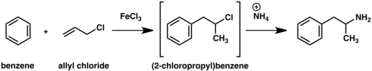
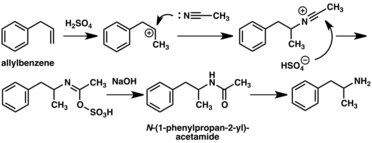
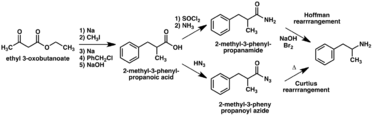
A large number of alternative synthetic routes to amphetamine have been developed based on classic organic reactions.[267][268] One example is the Friedel–Crafts alkylation of benzene by allyl chloride to yield beta chloropropylbenzene which is then reacted with ammonia to produce racemic amphetamine (method 2).[273] Another example employs the Ritter reaction (method 3). In this route, allylbenzene is reacted acetonitrile in sulfuric acid to yield an organosulfate which in turn is treated with sodium hydroxide to give amphetamine via an acetamide intermediate.[274][275] A third route starts with ethyl 3-oxobutanoate which through a double alkylation with methyl iodide followed by benzyl chloride can be converted into 2-methyl-3-phenyl-propanoic acid. This synthetic intermediate can be transformed into amphetamine using either a Hofmann or Curtius rearrangement (method 4).[276]
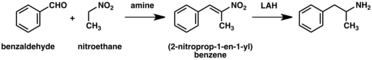
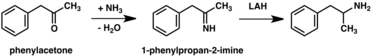
A significant number of amphetamine syntheses feature a reduction of a nitro, imine, oxime, or other nitrogen-containing functional groups.[268] In one such example, a Knoevenagel condensation of benzaldehyde with nitroethane yields phenyl-2-nitropropene. The double bond and nitro group of this intermediate is reduced using either catalytic hydrogenation or by treatment with lithium aluminium hydride (method 5).[269][277] Another method is the reaction of phenylacetone with ammonia, producing an imine intermediate that is reduced to the primary amine using hydrogen over a palladium catalyst or lithium aluminum hydride (method 6).[269]
|
|
|
Detection in body fluids
Amphetamine is frequently measured in urine or blood as part of a drug test for sports, employment, poisoning diagnostics, and forensics.[sources 20] Techniques such as immunoassay, which is the most common form of amphetamine test, may cross-react with a number of sympathomimetic drugs.[281] Chromatographic methods specific for amphetamine are employed to prevent false positive results.[282] Chiral separation techniques may be employed to help distinguish the source of the drug, whether prescription amphetamine, prescription amphetamine prodrugs, (e.g., selegiline), over-the-counter drug products that contain levomethamphetamine,[note 22] or illicitly obtained substituted amphetamines.[282][285][286] Several prescription drugs produce amphetamine as a metabolite, including benzphetamine, clobenzorex, famprofazone, fenproporex, lisdexamfetamine, mesocarb, methamphetamine, prenylamine, and selegiline, among others.[4][287][288] These compounds may produce positive results for amphetamine on drug tests.[287][288] Amphetamine is generally only detectable by a standard drug test for approximately 24 hours, although a high dose may be detectable for 2–4 days.[281]
For the assays, a study noted that an enzyme multiplied immunoassay technique (EMIT) assay for amphetamine and methamphetamine may produce more false positives than liquid chromatography–tandem mass spectrometry.[285] Gas chromatography–mass spectrometry (GC–MS) of amphetamine and methamphetamine with the derivatizing agent (S)-(−)-trifluoroacetylprolyl chloride allows for the detection of methamphetamine in urine.[282] GC–MS of amphetamine and methamphetamine with the chiral derivatizing agent Mosher's acid chloride allows for the detection of both dextroamphetamine and dextromethamphetamine in urine.[282] Hence, the latter method may be used on samples that test positive using other methods to help distinguish between the various sources of the drug.[282]
History, society, and culture
| Substance | Best estimate |
Low estimate |
High estimate |
|---|---|---|---|
| Amphetamine- type stimulants |
34.16 | 13.42 | 55.24 |
| Cannabis | 192.15 | 165.76 | 234.06 |
| Cocaine | 18.20 | 13.87 | 22.85 |
| Ecstasy | 20.57 | 8.99 | 32.34 |
| Opiates | 19.38 | 13.80 | 26.15 |
| Opioids | 34.26 | 27.01 | 44.54 |
Amphetamine was first synthesized in 1887 in Germany by Romanian chemist Lazăr Edeleanu who named it phenylisopropylamine;[266][290][291] its stimulant effects remained unknown until 1927, when it was independently resynthesized by Gordon Alles and reported to have sympathomimetic properties.[291] Amphetamine had no medical use until late 1933, when Smith, Kline and French began selling it as an inhaler under the brand name Benzedrine as a decongestant.[10] Benzedrine sulfate was introduced 3 years later and was used to treat a wide variety of medical conditions, including narcolepsy, obesity, low blood pressure, low libido, and chronic pain, among others.[36][10] During World War II, amphetamine and methamphetamine were used extensively by both the Allied and Axis forces for their stimulant and performance-enhancing effects.[266][292][293] As the addictive properties of the drug became known, governments began to place strict controls on the sale of amphetamine.[266] For example, during the early 1970s in the United States, amphetamine became a schedule II controlled substance under the Controlled Substances Act.[294] In spite of strict government controls, amphetamine has been used legally or illicitly by people from a variety of backgrounds, including authors,[295] musicians,[296] mathematicians,[297] and athletes.[8]
Amphetamine is illegally synthesized in clandestine labs and sold on the black market, primarily in European countries.[298] Among European Union (EU) member states in 2018,[update] 11.9 million adults of ages 15–64 have used amphetamine or methamphetamine at least once in their lives and 1.7 million have used either in the last year.[299] During 2012, approximately 5.9 metric tons of illicit amphetamine were seized within EU member states;[300] the "street price" of illicit amphetamine within the EU ranged from €6–38 per gram during the same period.[300] Outside Europe, the illicit market for amphetamine is much smaller than the market for methamphetamine and MDMA.[298]
Legal status
As a result of the United Nations 1971 Convention on Psychotropic Substances, amphetamine became a schedule II controlled substance, as defined in the treaty, in all 183 state parties.[11] Consequently, it is heavily regulated in most countries.[301][302] Some countries, such as South Korea and Japan, have banned substituted amphetamines even for medical use.[303][304] In other nations, such as Brazil (class A3),[305] Canada (schedule I drug),[306] the Netherlands (List I drug),[307] the United States (schedule II drug),[294] Australia (schedule 8),[308] Thailand (category 1 narcotic),[309] and United Kingdom (class B drug),[310] amphetamine is in a restrictive national drug schedule that allows for its use as a medical treatment.[298][12]
Pharmaceutical products
Several currently marketed amphetamine formulations contain both enantiomers, including those marketed under the brand names Adderall, Adderall XR, Mydayis,[note 23] Adzenys ER, Adzenys XR-ODT, Dyanavel XR, Evekeo, and Evekeo ODT. Of those, Evekeo (including Evekeo ODT) is the only product containing only racemic amphetamine (as amphetamine sulfate), and is therefore the only one whose active moiety can be accurately referred to simply as "amphetamine".[35][16][108] Dextroamphetamine, marketed under the brand names Dexedrine and Zenzedi, is the only enantiopure amphetamine product currently available. A prodrug form of dextroamphetamine, lisdexamfetamine, is also available and is marketed under the brand name Vyvanse. As it is a prodrug, lisdexamfetamine is structurally different from dextroamphetamine, and is inactive until it metabolizes into dextroamphetamine.[19][235] The free base of racemic amphetamine was previously available as Benzedrine, Psychedrine, and Sympatedrine.[4] Levoamphetamine was previously available as Cydril.[4] Many current amphetamine pharmaceuticals are salts due to the comparatively high volatility of the free base.[4][19][33] However, oral suspension and orally disintegrating tablet (ODT) dosage forms composed of the free base were introduced in 2015 and 2016, respectively.[108][311][312] Some of the current brands and their generic equivalents are listed below.
| Brand name |
United States Adopted Name |
(D:L) ratio |
Dosage form |
Marketing start date |
Sources |
|---|---|---|---|---|---|
| Adderall | – | 3:1 (salts) | tablet | 1996 | [4][19] |
| Adderall XR | – | 3:1 (salts) | capsule | 2001 | [4][19] |
| Mydayis | – | 3:1 (salts) | capsule | 2017 | [313][314] |
| Adzenys ER | amphetamine | 3:1 (base) | suspension | 2017 | [315] |
| Adzenys XR-ODT | amphetamine | 3:1 (base) | ODT | 2016 | [312][316] |
| Dyanavel XR | amphetamine | 3.2:1 (base) | suspension | 2015 | [108][311] |
| Evekeo | amphetamine sulfate | 1:1 (salts) | tablet | 2012 | [16][317] |
| Evekeo ODT | amphetamine sulfate | 1:1 (salts) | ODT | 2019 | [318] |
| Dexedrine | dextroamphetamine sulfate | 1:0 (salts) | capsule | 1976 | [4][19] |
| Zenzedi | dextroamphetamine sulfate | 1:0 (salts) | tablet | 2013 | [19][319] |
| Vyvanse | lisdexamfetamine dimesylate | 1:0 (prodrug) | capsule | 2007 | [4][235][320] |
| tablet | |||||
| Xelstrym | dextroamphetamine | 1:0 (base) | patch | 2022 | [321] |
| drug | formula | molecular mass [note 24] |
amphetamine base [note 25] |
amphetamine base in equal doses |
doses with equal base content [note 26] | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| (g/mol) | (percent) | (30 mg dose) | ||||||||
| total | base | total | dextro- | levo- | dextro- | levo- | ||||
| dextroamphetamine sulfate[323][324] | (C9H13N)2•H2SO4 | |||||||||
| amphetamine sulfate[325] | (C9H13N)2•H2SO4 | |||||||||
| Adderall | ||||||||||
| 25% | dextroamphetamine sulfate[323][324] | (C9H13N)2•H2SO4 | ||||||||
| 25% | amphetamine sulfate[325] | (C9H13N)2•H2SO4 | ||||||||
| 25% | dextroamphetamine saccharate[326] | (C9H13N)2•C6H10O8 | ||||||||
| 25% | amphetamine aspartate monohydrate[327] | (C9H13N)•C4H7NO4•H2O | ||||||||
| lisdexamfetamine dimesylate[235] | C15H25N3O•(CH4O3S)2 | |||||||||
| amphetamine base suspension[note 27][108] | C9H13N | |||||||||
Notes
- ↑ Amphetamine is contracted from alpha-methylphenethylamine. Synonyms and alternate spellings include: 1-phenylpropan-2-amine (IUPAC name), α-methylphenethylamine, amfetamine (International Nonproprietary Name [INN]), β-phenylisopropylamine, thyramine, and speed.[1][2][3]
- ↑ Enantiomers are molecules that are mirror images of one another; they are structurally identical, but of the opposite orientation.[18]
Levoamphetamine and dextroamphetamine are also known as L-amph or levamfetamine (INN) and D-amph or dexamfetamine (INN) respectively.[1] - ↑ The brand name Adderall is used throughout this article to refer to the amphetamine four-salt mixture it contains (dextroamphetamine sulfate 25%, dextroamphetamine saccharate 25%, amphetamine sulfate 25%, and amphetamine aspartate 25%). The nonproprietary name, which lists all four active constituent chemicals, is excessively lengthy.[19]
- ↑ The term "amphetamines" also refers to a chemical class, but, unlike the class of substituted amphetamines,[31] the "amphetamines" class does not have a standardized definition in academic literature.[6] One of the more restrictive definitions of this class includes only the racemate and enantiomers of amphetamine and methamphetamine.[6] The most general definition of the class encompasses a broad range of pharmacologically and structurally related compounds.[6]
Due to confusion that may arise from use of the plural form, this article will only use the terms "amphetamine" and "amphetamines" to refer to racemic amphetamine, levoamphetamine, and dextroamphetamine and reserve the term "substituted amphetamines" for its structural class. - ↑ The ADHD-related outcome domains with the greatest proportion of significantly improved outcomes from long-term continuous stimulant therapy include academics (≈55% of academic outcomes improved), driving (100% of driving outcomes improved), non-medical drug use (47% of addiction-related outcomes improved), obesity (≈65% of obesity-related outcomes improved), self-esteem (50% of self-esteem outcomes improved), and social function (67% of social function outcomes improved).[43]
The largest effect sizes for outcome improvements from long-term stimulant therapy occur in the domains involving academics (e.g., grade point average, achievement test scores, length of education, and education level), self-esteem (e.g., self-esteem questionnaire assessments, number of suicide attempts, and suicide rates), and social function (e.g., peer nomination scores, social skills, and quality of peer, family, and romantic relationships).[43]
Long-term combination therapy for ADHD (i.e., treatment with both a stimulant and behavioral therapy) produces even larger effect sizes for outcome improvements and improves a larger proportion of outcomes across each domain compared to long-term stimulant therapy alone.[43] These findings were further supported by a 2025 review of interventions for adolescents, which concluded that medications and cognitive-behavioral treatments (CBT) provide complementary benefits. Medications demonstrated strong short-term efficacy on core symptoms, while CBT contributed modest to strong, and sometimes long-lasting, improvements in functional impairments and executive skills when used as part of combination therapy.[44] - ↑ Cochrane reviews are high quality meta-analytic systematic reviews of randomized controlled trials.[51]
- ↑ In contrast to the Cochrane reviews that observed higher treatment discontinuation from adverse effects alone, this figure represents any cause of discontinuation (e.g., insufficient perceived treatment benefit).[54]
- ↑ The statements supported by the USFDA come from prescribing information, which is the copyrighted intellectual property of the manufacturer and approved by the USFDA. USFDA contraindications are not necessarily intended to limit medical practice but limit claims by pharmaceutical companies.[102]
- ↑ According to one review, amphetamine can be prescribed to individuals with a history of abuse provided that appropriate medication controls are employed, such as requiring daily pick-ups of the medication from the prescribing physician.[4]
- ↑ In individuals who experience sub-normal height and weight gains, a rebound to normal levels is expected to occur if stimulant therapy is briefly interrupted.[27][42][106] The average reduction in final adult height from 3 years of continuous stimulant therapy is 2 cm.[106]
- ↑ Transcription factors are proteins that increase or decrease the expression of specific genes.[138]
- ↑ In simpler terms, this necessary and sufficient relationship means that ΔFosB overexpression in the nucleus accumbens and addiction-related behavioral and neural adaptations always occur together and never occur alone.
- ↑ NMDA receptors are voltage-dependent ligand-gated ion channels that requires simultaneous binding of glutamate and a co-agonist (D-serine or glycine) to open the ion channel.[152]
- ↑ The review indicated that magnesium L-aspartate and magnesium chloride produce significant changes in addictive behavior;[129] other forms of magnesium were not mentioned.
- ↑ The 95% confidence interval indicates that there is a 95% probability that the true number of deaths lies between 3,425 and 4,145.
- ↑ The human dopamine transporter (hDAT) contains a high-affinity, extracellular, and allosteric Zn2+ (zinc ion) binding site which, upon zinc binding, inhibits dopamine reuptake, inhibits amphetamine-induced hDAT internalization, and amplifies amphetamine-induced dopamine efflux.[171][172][173][174] The human serotonin transporter and norepinephrine transporter do not contain zinc binding sites.[173]
- ↑ Mesolimbic dopamine neurons co-express the glutamate transporter EAAT3 alongside DAT, permitting amphetamine-induced EAAT3 internalization to influence glutamatergic signaling in the mesolimbic pathway.[183][186]
- ↑ Amphetamine interacts with its receptor protein target(s), TAAR1 and an as-yet-unidentified biomolecular target, which initiates signaling cascades that generate second messengers and activate protein kinases.[187] The activated kinases then phosphorylate their respective transporter(s), which in turn causes a conformational change in transporter protein, thereby altering its function and affecting dopaminergic/glutamatergic neurotransmission at dopaminergic synapses.[186][188]
- ↑ 4-Hydroxyamphetamine has been shown to be metabolized into 4-hydroxynorephedrine by dopamine beta-hydroxylase (DBH) in vitro and it is presumed to be metabolized similarly in vivo.[31][247] Evidence from studies that measured the effect of serum DBH concentrations on 4-hydroxyamphetamine metabolism in humans suggests that a different enzyme may mediate the conversion of 4-hydroxyamphetamine to 4-hydroxynorephedrine;[247][249] however, other evidence from animal studies suggests that this reaction is catalyzed by DBH in synaptic vesicles within noradrenergic neurons in the brain.[250][251]
- ↑ There is substantial variation in microbiome composition and microbial concentrations by anatomical site.[252][253] Fluid from the human colon – which contains the highest concentration of microbes of any anatomical site – contains approximately one trillion (10^12) bacterial cells/ml.[252]
- ↑ Enantiomers are molecules that are mirror images of one another; they are structurally identical, but of the opposite orientation.[18]
- ↑ The active ingredient in some OTC inhalers in the United States is listed as levmetamfetamine, the INN and USAN of levomethamphetamine.[283][284]
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedAdderallDiff - ↑ For uniformity, molecular masses were calculated using the Lenntech Molecular Weight Calculator[322] and were within 0.01g/mol of published pharmaceutical values.
- ↑ Amphetamine base percentage = molecular massbase / molecular masstotal. Amphetamine base percentage for Adderall = sum of component percentages / 4.
- ↑ dose = (1 / amphetamine base percentage) × scaling factor = (molecular masstotal / molecular massbase) × scaling factor. The values in this column were scaled to a 30 mg dose of dextroamphetamine sulfate. Due to pharmacological differences between these medications (e.g., differences in the release, absorption, conversion, concentration, differing effects of enantiomers, half-life, etc.), the listed values should not be considered equipotent doses.
- ↑ This product (Dyanavel XR) is an oral suspension (i.e., a drug that is suspended in a liquid and taken by mouth) that contains 2.5 mg/mL of amphetamine base.[108] The product uses an ion exchange resin to achieve extended release of the amphetamine base.[108]
- Image legend
- ↑ (Text color) Transcription factors
Reference notes
- ↑ [4][5][6][7][8][9][10][11][12][13][14][15][16][17]
- ↑ [4][20][7][10][16][21][22]
- ↑ [20][7][8][9][13][23][24][25][26][27][28][29][30]
- ↑ [32][33][34]
- ↑ 5.0 5.1 [58][17][61][59][66]
- ↑ [35][9][23][106][108][109]
- ↑ [111][112][113][114]
- ↑ [9][103][111][113]
- ↑ [13][9][23][116]
- ↑ 10.0 10.1 [132][133][134][135][154]
- ↑ [130][132][131][139][140]
- ↑ [131][142][143][144]
- ↑ [3][9][23][160][162]
- ↑ [37][164][167][168]
- ↑ 15.0 15.1 [107][187][191][21][186][62][192][193]
- ↑ 16.0 16.1 [179][183][199][200][201][202][203]
- ↑ [21][179][199][200][204][209]
- ↑ 18.0 18.1 [224][225][226][227][228]
- ↑ 19.0 19.1 [232][31][245][170][246][237][247][248]
- ↑ [8][278][279][280]
References
- ↑ 1.0 1.1 1.2 "Amphetamine". Amphetamine. United States National Library of Medicine – National Center for Biotechnology Information. PubChem Compound Database. 11 April 2015.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Template:Cite DrugBank
- ↑ 3.0 3.1 "Review article: amphetamines and related drugs of abuse". Emergency Medicine Australasia 20 (5): 391–402. October 2008. doi:10.1111/j.1742-6723.2008.01114.x. PMID 18973636.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 "Amphetamine, past and present – a pharmacological and clinical perspective". Journal of Psychopharmacology 27 (6): 479–496. June 2013. doi:10.1177/0269881113482532. PMID 23539642. "The intravenous use of d-amphetamine and other stimulants still pose major safety risks to the individuals indulging in this practice. Some of this intravenous abuse is derived from the diversion of ampoules of d-amphetamine, which are still occasionally prescribed in the UK for the control of severe narcolepsy and other disorders of excessive sedation. ... For these reasons, observations of dependence and abuse of prescription d-amphetamine are rare in clinical practice, and this stimulant can even be prescribed to people with a history of drug abuse provided certain controls, such as daily pick-ups of prescriptions, are put in place (Jasinski and Krishnan, 2009b).".
- ↑ "Amphetamine-Type Stimulants". The Neuropsychiatric Complications of Stimulant Abuse. International Review of Neurobiology. 120. Academic Press. January 2015. pp. 9–25. doi:10.1016/bs.irn.2015.02.001. ISBN 978-0-12-802978-7.
- ↑ 6.0 6.1 6.2 6.3 "Chapter 1: Use and Misuse of Amphetamines: An International Overview". Amphetamine Misuse: International Perspectives on Current Trends. Amsterdam, Netherlands: Harwood Academic Publishers. 1997. p. 2. ISBN 978-90-5702-081-0. https://books.google.com/books?id=gVw_wzZU4x8C&pg=PA2. "Amphetamine, in the singular form, properly applies to the racemate of 2-amino-1-phenylpropane. ... In its broadest context, however, the term [amphetamines] can even embrace a large number of structurally and pharmacologically related substances."
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 "Chapter 13: Higher Cognitive Function and Behavioral Control". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York, US: McGraw-Hill Medical. 2009. pp. 318, 321. ISBN 978-0-07-148127-4. "Therapeutic (relatively low) doses of psychostimulants, such as methylphenidate and amphetamine, improve performance on working memory tasks both in normal subjects and those with ADHD. ... stimulants act not only on working memory function, but also on general levels of arousal and, within the nucleus accumbens, improve the saliency of tasks. Thus, stimulants improve performance on effortful but tedious tasks ... through indirect stimulation of dopamine and norepinephrine receptors. ...
Beyond these general permissive effects, dopamine (acting via D1 receptors) and norepinephrine (acting at several receptors) can, at optimal levels, enhance working memory and aspects of attention." - ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 "Nutritional supplements and ergogenic AIDS". Primary Care: Clinics in Office Practice 40 (2): 487–505. June 2013. doi:10.1016/j.pop.2013.02.009. PMID 23668655. "Amphetamines and caffeine are stimulants that increase alertness, improve focus, decrease reaction time, and delay fatigue, allowing for an increased intensity and duration of training ...
Physiologic and performance effects
• Amphetamines increase dopamine/norepinephrine release and inhibit their reuptake, leading to central nervous system (CNS) stimulation
• Amphetamines seem to enhance athletic performance in anaerobic conditions 39 40
• Improved reaction time
• Increased muscle strength and delayed muscle fatigue
• Increased acceleration
• Increased alertness and attention to task". - ↑ 9.00 9.01 9.02 9.03 9.04 9.05 9.06 9.07 9.08 9.09 9.10 9.11 9.12 9.13 9.14 9.15 9.16 9.17 9.18 9.19 9.20 9.21 9.22 9.23 9.24 9.25 9.26 9.27 9.28 9.29 9.30 9.31 9.32 9.33 9.34 9.35 9.36 9.37 "Adderall XR- dextroamphetamine sulfate, dextroamphetamine saccharate, amphetamine sulfate and amphetamine aspartate capsule, extended release". Shire US Inc.. 17 July 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=aff45863-ffe1-4d4f-8acf-c7081512a6c0.
- ↑ 10.0 10.1 10.2 10.3 "Making the first anti-depressant: amphetamine in American medicine, 1929–1950". Journal of the History of Medicine and Allied Sciences 61 (3): 288–323. July 2006. doi:10.1093/jhmas/jrj039. PMID 16492800. "However the firm happened to discover the drug, SKF first packaged it as an inhaler so as to exploit the base's volatility and, after sponsoring some trials by East Coast otolaryngological specialists, began to advertise the Benzedrine Inhaler as a decongestant in late 1933.".
- ↑ 11.0 11.1 "Convention on psychotropic substances". United Nations. http://treaties.un.org/Pages/ViewDetails.aspx?src=TREATY&mtdsg_no=VI-16&chapter=6&lang=en.
- ↑ 12.0 12.1 "Misuse and diversion of stimulants prescribed for ADHD: a systematic review of the literature". Journal of the American Academy of Child & Adolescent Psychiatry 47 (1): 21–31. January 2008. doi:10.1097/chi.0b013e31815a56f1. PMID 18174822. "Stimulant misuse appears to occur both for performance enhancement and their euphorogenic effects, the latter being related to the intrinsic properties of the stimulants (e.g., IR versus ER profile) ...
Although useful in the treatment of ADHD, stimulants are controlled II substances with a history of preclinical and human studies showing potential abuse liability.". - ↑ 13.0 13.1 13.2 Montgomery KA (June 2008). "Sexual desire disorders". Psychiatry 5 (6): 50–55. PMID 19727285.
- ↑ "Amphetamine". United States National Library of Medicine. https://meshb.nlm.nih.gov/#/record/ui?name=Amphetamine.
- ↑ "Guidelines on the Use of International Nonproprietary Names (INNS) for Pharmaceutical Substances". World Health Organization. 1997. http://apps.who.int/medicinedocs/en/d/Jh1806e/2.4.html. "In principle, INNs are selected only for the active part of the molecule which is usually the base, acid or alcohol. In some cases, however, the active molecules need to be expanded for various reasons, such as formulation purposes, bioavailability or absorption rate. In 1975 the experts designated for the selection of INN decided to adopt a new policy for naming such molecules. In future, names for different salts or esters of the same active substance should differ only with regard to the inactive moiety of the molecule. ... The latter are called modified INNs (INNMs)."
- ↑ 16.0 16.1 16.2 16.3 16.4 16.5 16.6 16.7 16.8 "Evekeo- amphetamine sulfate tablet". Arbor Pharmaceuticals, LLC. 14 August 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=f469fb38-0380-4621-9db3-a4f429126156.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 "Pharmacotherapy, alternative and adjunctive therapies for eating disorders: findings from a rapid review". Journal of Eating Disorders 11 (1). July 2023. doi:10.1186/s40337-023-00833-9. PMID 37415200. "LDX is commonly used in the treatment of ADHD, and is the only treatment for BED that is approved by the Food and Drug Administration (FDA) and the Therapeutic Goods Administration (TGA). LDX, like all amphetamine stimulants, has direct appetite suppressant effects that may be therapeutically useful in BED, although long-term neuroadaptations in dopaminergic and noradrenergic systems caused by LDX may also be relevant, leading to improved regulation of eating behaviours, attentional processes and goal-directed behaviours. ...
Evidently, there is a substantial volume of trials with high-quality evidence supporting the efficacy of LDX in reducing binge eating frequency in treatment of adults with moderate to severe BED at 50–70 mg/day.". - ↑ 18.0 18.1 "Enantiomer". IUPAC Compendium of Chemical Terminology. International Union of Pure and Applied Chemistry. IUPAC Goldbook. 2009. doi:10.1351/goldbook.E02069. ISBN 978-0-9678550-9-7. http://goldbook.iupac.org/E02069.html. Retrieved 14 March 2014. "One of a pair of molecular entities which are mirror images of each other and non-superposable."
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 19.7 "National Drug Code Amphetamine Search Results". United States Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/ndc/results.cfm?beginrow=1&numberperpage=160&searchfield=amphetamine&searchtype=ActiveIngredient&OrderBy=ProprietaryName.
- ↑ 20.0 20.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedAdderall IR - ↑ 21.00 21.01 21.02 21.03 21.04 21.05 21.06 21.07 21.08 21.09 21.10 21.11 21.12 21.13 21.14 21.15 21.16 21.17 21.18 21.19 21.20 21.21 21.22 21.23 "The emerging role of trace amine-associated receptor 1 in the functional regulation of monoamine transporters and dopaminergic activity". Journal of Neurochemistry 116 (2): 164–176. January 2011. doi:10.1111/j.1471-4159.2010.07109.x. PMID 21073468.
- ↑ 22.0 22.1 22.2 22.3 22.4 ""TAARgeting Addiction"-The Alamo Bears Witness to Another Revolution: An Overview of the Plenary Symposium of the 2015 Behavior, Biology and Chemistry Conference". Drug and Alcohol Dependence 159: 9–16. February 2016. doi:10.1016/j.drugalcdep.2015.11.014. PMID 26644139. "When considered together with the rapidly growing literature in the field a compelling case emerges in support of developing TAAR1-selective agonists as medications for preventing relapse to psychostimulant abuse.".
- ↑ 23.00 23.01 23.02 23.03 23.04 23.05 23.06 23.07 23.08 23.09 23.10 23.11 23.12 23.13 23.14 23.15 23.16 23.17 23.18 23.19 23.20 23.21 Goodman & Gilman's Pharmacological Basis of Therapeutics (12th ed.). New York, US: McGraw-Hill. 2010. ISBN 978-0-07-162442-8. "Dextrorotatory substitution on the α-carbon generally results in a more potent compound. d-Amphetamine is more potent than l-amphetamine in central but not peripheral activity. ... In eliciting CNS excitatory effects, the d-isomer (dextroamphetamine) is three to four times more potent than the l-isomer."
- ↑ 24.0 24.1 24.2 24.3 24.4 "Treatment for amphetamine psychosis". Cochrane Database of Systematic Reviews 2009 (1). January 2009. doi:10.1002/14651858.CD003026.pub3. PMID 19160215. "A minority of individuals who use amphetamines develop full-blown psychosis requiring care at emergency departments or psychiatric hospitals. In such cases, symptoms of amphetamine psychosis commonly include paranoid and persecutory delusions as well as auditory and visual hallucinations in the presence of extreme agitation. More common (about 18%) is for frequent amphetamine users to report psychotic symptoms that are sub-clinical and that do not require high-intensity intervention ...
About 5–15% of the users who develop an amphetamine psychosis fail to recover completely (Hofmann 1983) ...
Findings from one trial indicate use of antipsychotic medications effectively resolves symptoms of acute amphetamine psychosis.
psychotic symptoms of individuals with amphetamine psychosis may be due exclusively to heavy use of the drug or heavy use of the drug may exacerbate an underlying vulnerability to schizophrenia.". - ↑ 25.0 25.1 25.2 25.3 25.4 "Amphetamine-induced psychosis—a separate diagnostic entity or primary psychosis triggered in the vulnerable?". BMC Psychiatry 12. December 2012. doi:10.1186/1471-244X-12-221. PMID 23216941. "In these studies, amphetamine was given in consecutively higher doses until psychosis was precipitated, often after 100–300 mg of amphetamine ... Secondly, psychosis has been viewed as an adverse event, although rare, in children with ADHD who have been treated with amphetamine".
- ↑ 26.0 26.1 26.2 Greydanus D. "Stimulant Misuse: Strategies to Manage a Growing Problem". ACHA Professional Development Program. p. 20. http://www.acha.org/prof_dev/ADHD_docs/ADHD_PDprogram_Article2.pdf.
- ↑ 27.0 27.1 27.2 27.3 27.4 27.5 27.6 "Long-term outcomes with medications for attention-deficit hyperactivity disorder: current status of knowledge". CNS Drugs 25 (7): 539–554. July 2011. doi:10.2165/11589380-000000000-00000. PMID 21699268. "Several other studies,[97-101] including a meta-analytic review[98] and a retrospective study,[97] suggested that stimulant therapy in childhood is associated with a reduced risk of subsequent substance use, cigarette smoking and alcohol use disorders. ... Recent studies have demonstrated that stimulants, along with the non-stimulants atomoxetine and extended-release guanfacine, are continuously effective for more than 2-year treatment periods with few and tolerable adverse effects. The effectiveness of long-term therapy includes not only the core symptoms of ADHD, but also improved quality of life and academic achievements. The most concerning short-term adverse effects of stimulants, such as elevated blood pressure and heart rate, waned in long-term follow-up studies. ... The current data do not support the potential impact of stimulants on the worsening or development of tics or substance abuse into adulthood. In the longest follow-up study (of more than 10 years), lifetime stimulant treatment for ADHD was effective and protective against the development of adverse psychiatric disorders.".
- ↑ 28.0 28.1 "Chapter 16: Reinforcement and Addictive Disorders". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (3rd ed.). New York: McGraw-Hill Medical. 2015. ISBN 978-0-07-182770-6. "Such agents also have important therapeutic uses; cocaine, for example, is used as a local anesthetic (Chapter 2), and amphetamines and methylphenidate are used in low doses to treat attention deficit hyperactivity disorder and in higher doses to treat narcolepsy (Chapter 12). Despite their clinical uses, these drugs are strongly reinforcing, and their long-term use at high doses is linked with potential addiction, especially when they are rapidly administered or when high-potency forms are given."
- ↑ 29.0 29.1 "A qualitative review of issues arising in the use of psycho-stimulant medications in patients with ADHD and co-morbid substance use disorders". Current Medical Research and Opinion 24 (5): 1345–1357. May 2008. doi:10.1185/030079908X280707. PMID 18384709. "When oral formulations of psychostimulants are used at recommended doses and frequencies, they are unlikely to yield effects consistent with abuse potential in patients with ADHD.".
- ↑ 30.0 30.1 30.2 30.3 30.4 30.5 "Management of Narcolepsy". Current Treatment Options in Neurology 18 (10). October 2016. doi:10.1007/s11940-016-0429-y. PMID 27549768. "The usefulness of amphetamines is limited by a potential risk of abuse, and their cardiovascular adverse effects (Table 1). That is why, even though they are cheaper than other drugs, and efficient, they remain third-line therapy in narcolepsy. Three class II studies showed an improvement of EDS in that disease. ...
Despite the potential for drug abuse or tolerance using stimulants, patients with narcolepsy rarely exhibit addiction to their medication. ...
Some stimulants, such as mazindol, amphetamines, and pitolisant, may also have some anticataplectic effects.". - ↑ 31.0 31.1 31.2 31.3 31.4 31.5 31.6 Glennon RA (2013). Foye's principles of medicinal chemistry (7th ed.). Philadelphia, USA: Wolters Kluwer Health/Lippincott Williams & Wilkins. pp. 646–648. ISBN 9781609133450. "The simplest unsubstituted phenylisopropylamine, 1-phenyl-2-aminopropane, or amphetamine, serves as a common structural template for hallucinogens and psychostimulants. Amphetamine produces central stimulant, anorectic, and sympathomimetic actions, and it is the prototype member of this class (39). ... The phase 1 metabolism of amphetamine analogs is catalyzed by two systems: cytochrome P450 and flavin monooxygenase. ... Amphetamine can also undergo aromatic hydroxylation to p-hydroxyamphetamine. ... Subsequent oxidation at the benzylic position by DA β-hydroxylase affords p-hydroxynorephedrine. Alternatively, direct oxidation of amphetamine by DA β-hydroxylase can afford norephedrine."
- ↑ 32.0 32.1 32.2 32.3 32.4 32.5 Broadley KJ (March 2010). "The vascular effects of trace amines and amphetamines". Pharmacology & Therapeutics 125 (3): 363–375. doi:10.1016/j.pharmthera.2009.11.005. PMID 19948186.
- ↑ 33.0 33.1 33.2 33.3 "Amphetamine". http://www.emcdda.europa.eu/publications/drug-profiles/amphetamine.
- ↑ 34.0 34.1 34.2 34.3 "Biosynthesis of amphetamine analogs in plants". Trends in Plant Science 17 (7): 404–412. 2012. doi:10.1016/j.tplants.2012.03.004. PMID 22502775. Bibcode: 2012TPS....17..404H. "Substituted amphetamines, which are also called phenylpropylamino alkaloids, are a diverse group of nitrogen-containing compounds that feature a phenethylamine backbone with a methyl group at the α-position relative to the nitrogen (Figure 1). ... Beyond (1R,2S)-ephedrine and (1S,2S)-pseudoephedrine, myriad other substituted amphetamines have important pharmaceutical applications. ... For example, (S)-amphetamine (Figure 4b), a key ingredient in Adderall and Dexedrine, is used to treat attention deficit hyperactivity disorder (ADHD) [79]. ...
[Figure 4](b) Examples of synthetic, pharmaceutically important substituted amphetamines.". - ↑ 35.0 35.1 35.2 35.3 35.4 35.5 35.6 "Amphetamine (D,L)". Prescriber's Guide: Stahl's Essential Psychopharmacology (6th ed.). Cambridge, United Kingdom: Cambridge University Press. March 2017. pp. 45–51. ISBN 978-1-108-22874-9. https://books.google.com/books?id=9hssDwAAQBAJ&pg=PA45. Retrieved 5 August 2017.
- ↑ 36.0 36.1 "Benzedrine sulphate in clinical medicine; a survey of the literature". Postgraduate Medical Journal 22 (250): 205–218. August 1946. doi:10.1136/pgmj.22.250.205. PMID 20997404.
- ↑ 37.0 37.1 "Toxicity of amphetamines: an update". Archives of Toxicology 86 (8): 1167–1231. August 2012. doi:10.1007/s00204-012-0815-5. PMID 22392347. Bibcode: 2012ArTox..86.1167C.
- ↑ "Abuse of amphetamines and structural abnormalities in the brain". Annals of the New York Academy of Sciences 1141 (1): 195–220. October 2008. doi:10.1196/annals.1441.031. PMID 18991959.
- ↑ 39.0 39.1 "Meta-analysis of functional magnetic resonance imaging studies of inhibition and attention in attention-deficit/hyperactivity disorder: exploring task-specific, stimulant medication, and age effects". JAMA Psychiatry 70 (2): 185–198. February 2013. doi:10.1001/jamapsychiatry.2013.277. PMID 23247506.
- ↑ 40.0 40.1 "Effect of psychostimulants on brain structure and function in ADHD: a qualitative literature review of magnetic resonance imaging-based neuroimaging studies". The Journal of Clinical Psychiatry 74 (9): 902–917. September 2013. doi:10.4088/JCP.12r08287. PMID 24107764.
- ↑ 41.0 41.1 "Meta-analysis of structural MRI studies in children and adults with attention deficit hyperactivity disorder indicates treatment effects". Acta Psychiatrica Scandinavica 125 (2): 114–126. February 2012. doi:10.1111/j.1600-0447.2011.01786.x. PMID 22118249. "Basal ganglia regions like the right globus pallidus, the right putamen, and the nucleus caudatus are structurally affected in children with ADHD. These changes and alterations in limbic regions like ACC and amygdala are more pronounced in non-treated populations and seem to diminish over time from child to adulthood. Treatment seems to have positive effects on brain structure.".
- ↑ 42.0 42.1 42.2 42.3 "Chapter 9: Medications for ADHD". Attention Deficit Hyperactivity Disorder Handbook: A Physician's Guide to ADHD (2nd ed.). New York, US: Springer. 2010. pp. 121–123, 125–127. ISBN 978-1-4419-1396-8. "Ongoing research has provided answers to many of the parents' concerns, and has confirmed the effectiveness and safety of the long-term use of medication."
- ↑ 43.0 43.1 43.2 43.3 43.4 "Effect of treatment modality on long-term outcomes in attention-deficit/hyperactivity disorder: a systematic review". PLOS ONE 10 (2). February 2015. doi:10.1371/journal.pone.0116407. PMID 25714373. "The highest proportion of improved outcomes was reported with combination treatment (83% of outcomes). Among significantly improved outcomes, the largest effect sizes were found for combination treatment. The greatest improvements were associated with academic, self-esteem, or social function outcomes.".
Figure 3: Treatment benefit by treatment type and outcome group - ↑ "Research Review: Pharmacological and non-pharmacological treatments for adolescents with attention deficit/hyperactivity disorder - a systematic review of the literature". Journal of Child Psychology and Psychiatry, and Allied Disciplines 66 (1): 132–149. January 2025. doi:10.1111/jcpp.14056. PMID 39370392. "The main efficacy-related conclusions of this review are: (a) medications demonstrated the strongest and most consistent effects on core ADHD symptoms (especially inattention), (b) heterogeneous C/BTs demonstrated inconsistent effects on ADHD symptoms, strong consistent effects on impairment and executive function skills, and modest consistent effects on internalizing symptoms and analogue note-taking performance, (c) C/BTs demonstrated consistent maintenance effects for executive function skills and impairment up to 6 months and possibly 3 years post-treatment, (d) though comparing the efficacy of two C/BTs rarely led to significant differences, which C/BT worked best for whom could be reliably predicted from patient- and provider-level moderators ...
Thus, maximal therapeutic benefit (in terms of breadth of response and maintenance of effects) might be achieved by combining medication and C/BTs, a recommendation generally reflected in current practice parameters (AACAP, 2007; AADPA, 2022; NICE, 2018; Wolraich et al., 2019).". - ↑ "Systematic Review and Meta-Analysis: Effects of Pharmacological Treatment for Attention-Deficit/Hyperactivity Disorder on Quality of Life". Journal of the American Academy of Child and Adolescent Psychiatry 64 (3): S0890–8567(24)00304–6. 30 May 2024. doi:10.1016/j.jaac.2024.05.023. PMID 38823477. "We conducted the first systematic review and meta-analysis investigating the effects of medication for ADHD on quality of life (QoL) in parallel or crossover RCTs. Overall, we found that methylphenidate, amphetamines, and atomoxetine were significantly more efficacious than placebo in improving QoL in people with ADHD. ...
Four studies on amphetamines (950 participants with ADHD in total; 45% adults) reported relevant data for effect sizes to be computed. The meta-analysis on 14 effect sizes showed that amphetamines led to better QoL than placebo in individuals with ADHD.". - ↑ 46.0 46.1 46.2 "Chapter 6: Widely Projecting Systems: Monoamines, Acetylcholine, and Orexin". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York, US: McGraw-Hill Medical. 2009. pp. 154–157. ISBN 978-0-07-148127-4.
- ↑ 47.0 47.1 47.2 "Cognitive enhancers for the treatment of ADHD". Pharmacology Biochemistry and Behavior 99 (2): 262–274. August 2011. doi:10.1016/j.pbb.2011.05.002. PMID 21596055.
- ↑ "The long-term outcomes of interventions for the management of attention-deficit hyperactivity disorder in children and adolescents: a systematic review of randomized controlled trials". Psychology Research and Behavior Management 6: 87–99. September 2013. doi:10.2147/PRBM.S49114. PMID 24082796. "Only one paper53 examining outcomes beyond 36 months met the review criteria. ... There is high level evidence suggesting that pharmacological treatment can have a major beneficial effect on the core symptoms of ADHD (hyperactivity, inattention, and impulsivity) in approximately 80% of cases compared with placebo controls, in the short term.".
- ↑ "Chapter 9: Medications for ADHD". Attention Deficit Hyperactivity Disorder Handbook: A Physician's Guide to ADHD (2nd ed.). New York, US: Springer. 2010. pp. 111–113. ISBN 978-1-4419-1396-8.
- ↑ "Stimulants for Attention Deficit Hyperactivity Disorder". Healthwise. 12 April 2010. http://www.webmd.com/add-adhd/childhood-adhd/stimulants-for-attention-deficit-hyperactivity-disorder.
- ↑ "The Cochrane Collaboration". European Journal of Clinical Nutrition 59 (Suppl 1): S147–S149; discussion S195–S196. August 2005. doi:10.1038/sj.ejcn.1602188. PMID 16052183.
- ↑ 52.0 52.1 "Amphetamines for attention deficit hyperactivity disorder (ADHD) in adults". Cochrane Database of Systematic Reviews 2018 (8). August 2018. doi:10.1002/14651858.CD007813.pub3. PMID 30091808.
- ↑ "Amphetamines for attention deficit hyperactivity disorder (ADHD) in children and adolescents". Cochrane Database of Systematic Reviews 2016 (2). February 2016. doi:10.1002/14651858.CD009996.pub2. PMID 26844979.
- ↑ 54.0 54.1 "Comparative efficacy and acceptability of pharmacological, psychological, and neurostimulatory interventions for ADHD in adults: a systematic review and component network meta-analysis". The Lancet. Psychiatry 12 (1): 32–43. 2025. doi:10.1016/S2215-0366(24)00360-2. PMID 39701638. "Our findings were based on 113 RCTs, including 14 887 participants, and indicated that stimulants were the only intervention that was supported by evidence of efficacy in the short term (ie, at timepoints closest to 12 weeks) for core symptoms of ADHD in adults (both self-reported and clinician-reported) and was associated with good acceptability (all-cause discontinuation).".
- ↑ "Pharmacological treatment for attention deficit hyperactivity disorder (ADHD) in children with comorbid tic disorders". Cochrane Database of Systematic Reviews 2018 (6). June 2018. doi:10.1002/14651858.CD007990.pub3. PMID 29944175.
- ↑ 56.0 56.1 "Binge eating disorder". Nature Reviews. Disease Primers 8 (1). March 2022. doi:10.1038/s41572-022-00344-y. PMID 35301358.
- ↑ 57.0 57.1 57.2 57.3 57.4 "Prospects for new drugs to treat binge-eating disorder: Insights from psychopathology and neuropharmacology". Journal of Psychopharmacology 36 (6): 680–703. June 2022. doi:10.1177/02698811211032475. PMID 34318734. "BED subjects have substantial decrements in their ventral striatal reward pathways and diminished ability to recruit fronto-cortical impulse-control circuits to implement dietary restraint. ...
There is not only substantial overlap between the psychopathology of BED and ADHD but also a clear association between these two disorders. Lisdexamfetamine's ability to reduce impulsivity and increase cognitive control in ADHD supports the hypothesis that efficacy in BED is dependent on treating its core obsessive, compulsive and impulsive behaviours.". - ↑ 58.0 58.1 58.2 58.3 "Pharmacologic Treatments for Binge-Eating Disorder". The Journal of Clinical Psychiatry 78 (Suppl 1): 14–19. 2017. doi:10.4088/JCP.sh16003su1c.03. PMID 28125174. "Genetic polymorphisms associated with abnormal dopaminergic signaling have been found in individuals who exhibit binge-eating behavior, and the binge-eating episodes, which often involve the consumption of highly palatable food, further stimulate the dopaminergic system. This ongoing stimulation may contribute to progressive impairments in dopamine signaling. Lisdexamfetamine is hypothesized to reduce binge-eating behavior by normalizing dopaminergic activity. ...
After 12 weeks, both studies found significant reductions in the number of binge-eating days per week in the active treatment group compared with placebo (P < .001 for both studies; Figure 1). Lisdexamfetamine was also found to be superior to placebo on a number of secondary outcome measures including global improvement, binge-eating cessation for 4 weeks, and reduction of obsessive-compulsive binge-eating symptoms, body weight, and triglycerides.". - ↑ 59.0 59.1 59.2 59.3 59.4 "The Neurobiology of Binge-eating Disorder Compared with Obesity: Implications for Differential Therapeutics". Clinical Therapeutics 43 (1): 50–69. January 2021. doi:10.1016/j.clinthera.2020.10.014. PMID 33257092. "Stimulant medications may be especially effective for individuals with BED because of dual effects on reward and executive function systems. Indeed, the only FDA-approved pharmacotherapy for BED is LDX, a d-amphetamine prodrug. ...
In humans, RCTs found that LDX reduced binge eating and impulsivity/compulsivity symptoms. Notably, there is a strong correlation between compulsivity symptoms and severity/frequency of binge eating episodes observed in LDX trials. Further, in individuals with BED, changes in prefrontal brain systems associated with LDX treatment were related to treatment outcome.". - ↑ "Chapter 14: Higher Cognitive Function and Behavioral Control". Molecular neuropharmacology: a foundation for clinical neuroscience (3rd ed.). New York: McGraw-Hill Medical. 2015. ISBN 978-0-07-182770-6. "Because behavioral responses in humans are not rigidly dictated by sensory inputs and drives, behavioral responses can instead be guided in accordance with short- or long-term goals, prior experience, and the environmental context. The response to a delicious-looking dessert is different depending on whether a person is alone staring into his or her refrigerator, is at a formal dinner party attended by his or her punctilious boss, or has just formulated the goal of losing 10 lb. ...
Adaptive responses depend on the ability to inhibit automatic or prepotent responses (eg, to ravenously eat the dessert or run from the snake) given certain social or environmental contexts or chosen goals and, in those circumstances, to select more appropriate responses. In conditions in which prepotent responses tend to dominate behavior, such as in drug addiction, where drug cues can elicit drug seeking (Chapter 16), or inattention deficit hyperactivity disorder (ADHD; described below), significant negative consequences can result." - ↑ 61.0 61.1 61.2 61.3 61.4 61.5 61.6 61.7 "Lisdexamfetamine and binge-eating disorder: A systematic review and meta-analysis of the preclinical and clinical data with a focus on mechanism of drug action in treating the disorder". European Neuropsychopharmacology 53: 49–78. December 2021. doi:10.1016/j.euroneuro.2021.08.001. PMID 34461386. http://pure-oai.bham.ac.uk/ws/files/147133958/LDX_final_pure.pdf. "Our meta-analysis of the four RCT data sets (Guerdjikova et al., 2016; McElroy et al., 2015b; McElroy et al., 2016a) showed an overall significant effect of LDX on binge-eating symptom change. ...
BED has been described as an impulse control disorder since one of the key symptoms of the disorder is a lack of control over eating (American Psychiatric Association, 2013) and it is possible that LDX may be effective in treating BED at least in part by reducing impulsivity, compulsivity, and the repetitive nature of binge eating. There is extensive evidence that loss of impulse control in BED is a causal factor in provoking bingeing symptoms (Colles et al., 2008; Galanti et al., 2007; Giel et al., 2017; McElroy et al., 2016a; Nasser et al., 2004; Schag et al., 2013). More specifically, BED is associated with motor impulsivity and non-planning impulsivity which could initiate and maintain binge eating (Nasser et al., 2004). Neuroimaging studies using the Stroop task to measure impulse control have shown that BED patients have decreased BOLD fMRI activity in brain areas involved in self-regulation and impulse control including VMPFC, inferior frontal gyrus (IFG), and insula during performance of the task compared to lean and obese controls (Balodis et al., 2013b). ...
It is conceivable that in BED patients a low 30 mg dose of LDX could reduce food intake by suppressing appetite or enhancing satiety and higher (50 and 70 mg) doses of the drug may have a dual suppressant effect on food intake and binge-eating frequency.". - ↑ 62.00 62.01 62.02 62.03 62.04 62.05 62.06 62.07 62.08 62.09 62.10 62.11 "Molecular Characterisation of the Mechanism of Action of Stimulant Drugs Lisdexamfetamine and Methylphenidate on ADHD Neurobiology: A Review". Neurology and Therapy 11 (4): 1489–1517. 11 August 2022. doi:10.1007/s40120-022-00392-2. PMID 35951288. "The active form of the drug has a central nervous system stimulating activity by the primary inhibition of DAT, NET, trace amine-associated receptor 1 (TAAR1) and vesicular monoamine transporter 2 (SLC18A2), among other targets, therefore regulating the reuptake and release of catecholamines (primarily NE and DA) on the synaptic cleft. ...
LDX can also promote the increase of DA in the synaptic cleft by activating protein TAAR1, which produces the efflux of monoamine NTs, mainly DA, from storage sites on presynaptic neurons. TAAR1 activation leads to intracellular cAMP signalling that results in PKA and PKC phosphorylation and activation. This PKC activation decreases DAT1, NET1 and SERT cell surface expression, intensifying the direct blockage of monoamine transporters by LDX and improving the neurotransmission imbalance in ADHD.". - ↑ 63.0 63.1 "Trace Amine Associated Receptor 1 (TAAR1) Modulation of Food Reward". Frontiers in Pharmacology 9. 2018. doi:10.3389/fphar.2018.00129. PMID 29535626. "As TAAR1 agonism has been shown to reduce various maladaptive eating behaviors, it can indeed be regarded as a very promising therapeutic target for compulsive eating behavior, even though much more research is needed at this point. Recently, Lisdexamfetamine (LDX) was approved for the treatment of BED, and has been shown to reduce binge eating behaviors as well as compulsive eating behaviors (assessed with the Yale-Brown Obsessive Compulsive Scale modified for Binge Eating; Y-BOCS-BE) (Hudson et al., 2017). Interestingly, TAAR1 is also activated by amphetamine, the active metabolite in LDX. LDX and TAAR1 agonism may, therefore, work through similar mechanisms to restore dysfunctional signaling in prefrontal areas, alleviating impaired inhibitory behaviors.".
- ↑ 64.0 64.1 64.2 "Stimulant prodrugs: A pharmacological and clinical assessment of their role in treating ADHD and binge-eating disorder". Pharmacological Advances in Central Nervous System Stimulants. Advances in Pharmacology (San Diego, Calif.). 99. 2024. pp. 251–286. doi:10.1016/bs.apha.2023.10.002. ISBN 978-0-443-21933-7. https://pubmed.ncbi.nlm.nih.gov/38467483/. "Together, the findings indicate that LDX has independent actions to tackle the underlying psychopathology of BED to inhibit binge-eating and produce weight-loss by reducing food intake through appetite suppression or enhanced satiety. ... Although BED is a predisposing factor for the development of obesity, it is unresponsive to appetite suppressants or anti-obesity drugs, emphasizing their different pathophysiological causes."
- ↑ 65.0 65.1 "Chapter 6: Psychostimulants". Drugs and the neuroscience of behavior: an introduction to psychopharmacology (2nd ed.). Los Angeles: SAGE. 2018. pp. 315–20. ISBN 978-1-5063-3894-1. "The cocaine- and amphetamine-regulated transcript (CART) is a peptide neurotransmitter that is produced after psychostimulant administration. Acute administration of a psychostimulant drug causes the activation of the gene for CART, leading to the synthesis of the CART peptide, which is then stored in vesicles within axon terminals. ... Psychostimulants reduce hunger through actions in the hypothalamus, a key structure for the regulation of appetite. Within this structure, enhanced dopamine release contributes to appetite suppression. CART also plays a role in hunger. In the hypothalamus, CART is activated by the appetite-suppressing hormone leptin."
- ↑ "Psychopharmacologic Management of Eating Disorders". Current Psychiatry Reports 24 (7): 345–351. July 2022. doi:10.1007/s11920-022-01340-5. PMID 35576089. "An 11-week, double-blind RCT examined the effects of three doses of lisdexamfetamine (30 mg/day, 50 mg/day, 70 mg/day) and placebo on binge eating frequency. Results indicated that 50 mg and 70 mg doses were superior to placebo in reducing binge eating. Two follow-up 12-week RCTs confirmed the superiority of 50 and 70 mg doses to placebo in improving binge eating and secondary outcome measures, including obsessive–compulsive symptoms, body weight, and global improvement. ... Subsequent studies of lisdexamfetamine provided further support for the medication's safety and efficacy and provided additional evidence that continued use may be better than placebo in preventing relapse. While it is considered safe and effective, lisdexamfetamine's side effect profile and risk for misuse may make it inappropriate for certain patients.".
- ↑ "The autoimmune basis of narcolepsy". Current Opinion in Neurobiology 23 (5): 767–773. October 2013. doi:10.1016/j.conb.2013.04.013. PMID 23725858.
- ↑ 68.0 68.1 68.2 "Narcolepsy". Journal of Sleep Research 31 (4). August 2022. doi:10.1111/jsr.13631. PMID 35624073. "Narcolepsy type 1 was called "narcolepsy with cataplexy" before 2014 (AASM, 2005), but was renamed NT1 in the third and last international classification of sleep disorders (AASM, 2014). ... A low level of Hcrt-1 in the CSF is very sensitive and specific for the diagnosis of NT1. ...
All patients with low CSF Hcrt-1 levels are considered as NT1 patients, even if they report no cataplexy (in about 10–20% of cases), and all patients with normal CSF Hcrt-1 levels (or without cataplexy when the lumbar puncture is not performed) as NT2 patients (Baumann et al., 2014). ...
In patients with NT1, the absence of Hcrt leads to the inhibition of regions that suppress REM sleep, thus allowing the activation of descending pathways inhibiting motoneurons, leading to cataplexy.". - ↑ 69.0 69.1 69.2 69.3 69.4 69.5 69.6 69.7 "A practical guide to the therapy of narcolepsy and hypersomnia syndromes". Neurotherapeutics 9 (4): 739–752. October 2012. doi:10.1007/s13311-012-0150-9. PMID 23065655. "At the pathophysiological level, it is now clear that most narcolepsy cases with cataplexy, and a minority of cases (5–30 %) without cataplexy or with atypical cataplexy-like symptoms, are caused by a lack of hypocretin (orexin) of likely an autoimmune origin. In these cases, once the disease is established, the majority of the 70,000 hypocretin-producing cells have been destroyed, and the disorder is irreversible. ...
Amphetamines are exceptionally wake-promoting, and at high doses also reduce cataplexy in narcoleptic patients, an effect best explained by its action on adrenergic and serotoninergic synapses. ...
The D-isomer is more specific for DA transmission and is a better stimulant compound. Some effects on cataplexy (especially for the L-isomer), secondary to adrenergic effects, occur at higher doses. ...
Numerous studies have shown that increased dopamine release is the main property explaining wake-promotion, although norepinephrine effects also contribute.". - ↑ 70.0 70.1 "Chapter 10: Neural and Neuroendocrine Control of the Internal Milieu". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (3rd ed.). New York: McGraw-Hill Medical. 2015. pp. 456–457. ISBN 978-0-07-182770-6. "More recently, the lateral hypothalamus was also found to play a central role in arousal. Neurons in this region contain cell bodies that produce the orexin (also called hypocretin) peptides (Chapter 6). These neurons project widely throughout the brain and are involved in sleep, arousal, feeding, reward, aspects of emotion, and learning. In fact, orexin is thought to promote feeding primarily by promoting arousal. Mutations in orexin receptors are responsible for narcolepsy in a canine model, knockout of the orexin gene produces narcolepsy in mice, and humans with narcolepsy have low or absent levels of orexin peptides in cerebrospinal fluid (Chapter 13). Lateral hypothalamus neurons have reciprocal connections with neurons that produce monoamine neurotransmitters (Chapter 6)."
- ↑ 71.0 71.1 71.2 "Chapter 13: Sleep and Arousal". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (3rd ed.). McGraw-Hill Medical. 2015. p. 521. ISBN 978-0-07-182770-6. "The ARAS consists of several different circuits including the four main monoaminergic pathways discussed in Chapter 6. The norepinephrine pathway originates from the LC and related brainstem nuclei; the serotonergic neurons originate from the RN within the brainstem as well; the dopaminergic neurons originate in the ventral tegmental area (VTA); and the histaminergic pathway originates from neurons in the tuberomammillary nucleus (TMN) of the posterior hypothalamus. As discussed in Chapter 6, these neurons project widely throughout the brain from restricted collections of cell bodies. Norepinephrine, serotonin, dopamine, and histamine have complex modulatory functions and, in general, promote wakefulness. The PT in the brainstem is also an important component of the ARAS. Activity of PT cholinergic neurons (REM-on cells) promotes REM sleep, as noted earlier. During waking, REM-on cells are inhibited by a subset of ARAS norepinephrine and serotonin neurons called REM-off cells."
- ↑ Sleep medicine a guide to sleep and its disorders (2nd ed.). John Wiley & Sons. 2009. p. 81. ISBN 978-1-4051-7851-8. https://books.google.com/books?id=kWxWEdqvue4C&pg=PA81. "All the amphetamines enhance activity at dopamine, noradrenaline and 5HT synapses. They cause presynaptic release of preformed transmitters, and also inhibit the re-uptake of dopamine and noradrenaline. These actions are most prominent in the brainstem ascending reticular activating system and the cerebral cortex."
- ↑ 73.0 73.1 "Neurophysiology of sleep and wakefulness: basic science and clinical implications". Current Neuropharmacology 6 (4): 367–378. 2008. doi:10.2174/157015908787386050. PMID 19587857. "Alertness and associated forebrain and cortical arousal are mediated by several ascending pathways with distinct neuronal components that project from the upper brain stem near the junction of the pons and the midbrain. ...
Key cell populations of the ascending arousal pathway include cholinergic, noradrenergic, serotoninergic, dopaminergic, and histaminergic neurons located in the pedunculopontine and laterodorsal tegmental nucleus (PPT/LDT), locus coeruleus, dorsal and median raphe nucleus, and tuberomammillary nucleus (TMN), respectively. ...
The mechanism of action of sympathomimetic alerting drugs (eg, dextro- and methamphetamine, methylphenidate) is direct or indirect stimulation of dopaminergic and noradrenergic nuclei, which in turn heightens the efficacy of the ventral periaqueductal grey area and locus coeruleus, both components of the secondary branch of the ascending arousal system. ...
Sympathomimetic drugs have long been used to treat narcolepsy". - ↑ "Medications for daytime sleepiness in individuals with idiopathic hypersomnia". The Cochrane Database of Systematic Reviews 2021 (5). 2021-05-25. doi:10.1002/14651858.CD012714.pub2. PMID 34031871. "Amphetamines both increase the release of and block the reuptake of monoaminergic neurotransmitters (i.e. dopamine, norepinephrine, and serotonin). Their effects on dopaminergic neurotransmission are particularly important for promoting wakefulness (Banerjee 2004; Gowda 2014).".
- ↑ 75.0 75.1 75.2 75.3 75.4 "Treatment of central disorders of hypersomnolence: an American Academy of Sleep Medicine clinical practice guideline". Journal of Clinical Sleep Medicine 17 (9): 1881–1893. September 2021. doi:10.5664/jcsm.9328. PMID 34743789. "The TF identified 1 double-blind RCT, 1 single-blind RCT, and 1 retrospective observational long-term self-reported case series assessing the efficacy of dextroamphetamine in patients with narcolepsy type 1 and narcolepsy type 2. These studies demonstrated clinically significant improvements in excessive daytime sleepiness and cataplexy.".
- ↑ "Narcolepsy and Other Central Hypersomnias". Continuum 23 (4, Sleep Neurology): 989–1004. August 2017. doi:10.1212/CON.0000000000000492. PMID 28777172. "Recent clinical trials and practice guidelines have confirmed that stimulants such as modafinil, armodafinil, or sodium oxybate (as first line); methylphenidate and pitolisant (as second line [pitolisant is currently only available in Europe]); and amphetamines (as third line) are appropriate medications for excessive daytime sleepiness.".
- ↑ "Update on the pharmacologic management of narcolepsy: mechanisms of action and clinical implications". Sleep Medicine 68: 97–109. April 2020. doi:10.1016/j.sleep.2019.09.001. PMID 32032921. "The first agents used to treat EDS (ie, amphetamines, methylphenidate) are now considered second- or third-line options because newer medications have been developed with improved tolerability and lower abuse potential (eg, modafinil/armodafinil, solriamfetol, pitolisant)".
- ↑ 78.0 78.1 "The Cognition-Enhancing Effects of Psychostimulants Involve Direct Action in the Prefrontal Cortex". Biological Psychiatry 77 (11): 940–950. June 2015. doi:10.1016/j.biopsych.2014.09.013. PMID 25499957. PMC 4377121. https://rdw.rowan.edu/cgi/viewcontent.cgi?article=1056&context=som_facpub. "The procognitive actions of psychostimulants are only associated with low doses. Surprisingly, despite nearly 80 years of clinical use, the neurobiology of the procognitive actions of psychostimulants has only recently been systematically investigated. Findings from this research unambiguously demonstrate that the cognition-enhancing effects of psychostimulants involve the preferential elevation of catecholamines in the PFC and the subsequent activation of norepinephrine α2 and dopamine D1 receptors. ... This differential modulation of PFC-dependent processes across dose appears to be associated with the differential involvement of noradrenergic α2 versus α1 receptors. Collectively, this evidence indicates that at low, clinically relevant doses, psychostimulants are devoid of the behavioral and neurochemical actions that define this class of drugs and instead act largely as cognitive enhancers (improving PFC-dependent function). ... In particular, in both animals and humans, lower doses maximally improve performance in tests of working memory and response inhibition, whereas maximal suppression of overt behavior and facilitation of attentional processes occurs at higher doses.".
- ↑ "Prescription Stimulants' Effects on Healthy Inhibitory Control, Working Memory, and Episodic Memory: A Meta-analysis". Journal of Cognitive Neuroscience 27 (6): 1069–1089. June 2015. doi:10.1162/jocn_a_00776. PMID 25591060. https://repository.upenn.edu/neuroethics_pubs/130. "Specifically, in a set of experiments limited to high-quality designs, we found significant enhancement of several cognitive abilities. ... The results of this meta-analysis ... do confirm the reality of cognitive enhancing effects for normal healthy adults in general, while also indicating that these effects are modest in size.".
- ↑ "Efficacy of stimulants for cognitive enhancement in non-attention deficit hyperactivity disorder youth: a systematic review". Addiction 109 (4): 547–557. April 2014. doi:10.1111/add.12460. PMID 24749160. "Amphetamine has been shown to improve consolidation of information (0.02 ≥ P ≤ 0.05), leading to improved recall.".
- ↑ "Regional cerebral blood flow response to oral amphetamine challenge in healthy volunteers". Journal of Nuclear Medicine 42 (4): 535–542. April 2001. PMID 11337538.
- ↑ "Chapter 10: Neural and Neuroendocrine Control of the Internal Milieu". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York, US: McGraw-Hill Medical. 2009. p. 266. ISBN 978-0-07-148127-4. "Dopamine acts in the nucleus accumbens to attach motivational significance to stimuli associated with reward."
- ↑ 83.0 83.1 83.2 "Psychostimulants and cognition: a continuum of behavioral and cognitive activation". Pharmacological Reviews 66 (1): 193–221. January 2014. doi:10.1124/pr.112.007054. PMID 24344115.
- ↑ Twohey M (26 March 2006). "Pills become an addictive study aid". http://www.jsonline.com/story/index.aspx?id=410902.
- ↑ "Illicit use of specific prescription stimulants among college students: prevalence, motives, and routes of administration". Pharmacotherapy 26 (10): 1501–1510. October 2006. doi:10.1592/phco.26.10.1501. PMID 16999660.
- ↑ "Pharmacological interventions for adolescents and adults with ADHD: stimulant and nonstimulant medications and misuse of prescription stimulants". Psychology Research and Behavior Management 7: 223–249. September 2014. doi:10.2147/PRBM.S47013. PMID 25228824. "misuse of prescription stimulants has become a serious problem on college campuses across the US and has been recently documented in other countries as well. ... Indeed, large numbers of students claim to have engaged in the nonmedical use of prescription stimulants, which is reflected in lifetime prevalence rates of prescription stimulant misuse ranging from 5% to nearly 34% of students.".
- ↑ "The potential for misuse and abuse of medications in ADHD: a review". Postgraduate Medicine 126 (5): 64–81. September 2014. doi:10.3810/pgm.2014.09.2801. PMID 25295651. "Overall, the data suggest that ADHD medication misuse and diversion are common health care problems for stimulant medications, with the prevalence believed to be approximately 5% to 10% of high school students and 5% to 35% of college students, depending on the study.".
- ↑ "National Study of Substance Use Trends Among NCAA College Student-Athletes". National Collegiate Athletic Association. January 2012. http://www.ncaapublications.com/productdownloads/SAHS09.pdf.
- ↑ Docherty JR (June 2008). "Pharmacology of stimulants prohibited by the World Anti-Doping Agency (WADA)". British Journal of Pharmacology 154 (3): 606–622. doi:10.1038/bjp.2008.124. PMID 18500382.
- ↑ 90.0 90.1 90.2 90.3 Parr JW (July 2011). "Attention-deficit hyperactivity disorder and the athlete: new advances and understanding". Clinics in Sports Medicine 30 (3): 591–610. doi:10.1016/j.csm.2011.03.007. PMID 21658550. "In 1980, Chandler and Blair47 showed significant increases in knee extension strength, acceleration, anaerobic capacity, time to exhaustion during exercise, pre-exercise and maximum heart rates, and time to exhaustion during maximal oxygen consumption (VO2 max) testing after administration of 15 mg of dextroamphetamine versus placebo. Most of the information to answer this question has been obtained in the past decade through studies of fatigue rather than an attempt to systematically investigate the effect of ADHD drugs on exercise.".
- ↑ 91.0 91.1 91.2 "Neurophysiological determinants of theoretical concepts and mechanisms involved in pacing". Sports Medicine 43 (5): 301–311. May 2013. doi:10.1007/s40279-013-0030-4. PMID 23456493. "In high-ambient temperatures, dopaminergic manipulations clearly improve performance. The distribution of the power output reveals that after dopamine reuptake inhibition, subjects are able to maintain a higher power output compared with placebo. ... Dopaminergic drugs appear to override a safety switch and allow athletes to use a reserve capacity that is 'off-limits' in a normal (placebo) situation.".
- ↑ "Executive dysfunction in Parkinson's disease and timing deficits". Frontiers in Integrative Neuroscience 7: 75. October 2013. doi:10.3389/fnint.2013.00075. PMID 24198770. "Manipulations of dopaminergic signaling profoundly influence interval timing, leading to the hypothesis that dopamine influences internal pacemaker, or "clock," activity. For instance, amphetamine, which increases concentrations of dopamine at the synaptic cleft advances the start of responding during interval timing, whereas antagonists of D2 type dopamine receptors typically slow timing;... Depletion of dopamine in healthy volunteers impairs timing, while amphetamine releases synaptic dopamine and speeds up timing.".
- ↑ "Is it time to turn our attention toward central mechanisms for post-exertional recovery strategies and performance?". Frontiers in Physiology 6: 79. March 2015. doi:10.3389/fphys.2015.00079. PMID 25852568. "Aside from accounting for the reduced performance of mentally fatigued participants, this model rationalizes the reduced RPE and hence improved cycling time trial performance of athletes using a glucose mouthwash (Chambers et al., 2009) and the greater power output during a RPE matched cycling time trial following amphetamine ingestion (Swart, 2009). ... Dopamine stimulating drugs are known to enhance aspects of exercise performance (Roelands et al., 2008)".
- ↑ "Neurophysiological effects of exercise in the heat". Scandinavian Journal of Medicine & Science in Sports 25 (Suppl 1): 65–78. June 2015. doi:10.1111/sms.12350. PMID 25943657. "This indicates that subjects did not feel they were producing more power and consequently more heat. The authors concluded that the "safety switch" or the mechanisms existing in the body to prevent harmful effects are overridden by the drug administration (Roelands et al., 2008b). Taken together, these data indicate strong ergogenic effects of an increased DA concentration in the brain, without any change in the perception of effort.".
- ↑ "Commonly Abused Prescription Drugs Chart". National Institute on Drug Abuse. http://www.drugabuse.gov/drugs-abuse/commonly-abused-drugs/commonly-abused-prescription-drugs-chart.
- ↑ "Stimulant ADHD Medications – Methylphenidate and Amphetamines". National Institute on Drug Abuse. http://www.drugabuse.gov/publications/infofacts/stimulant-adhd-medications-methylphenidate-amphetamines.
- ↑ 97.0 97.1 97.2 97.3 97.4 "National Institute on Drug Abuse. 2009. Stimulant ADHD Medications – Methylphenidate and Amphetamines". National Institute on Drug Abuse. https://nida.nih.gov/publications/research-reports/misuse-prescription-drugs/overview.
- ↑ 98.0 98.1 98.2 "Mixing the Medicine: The Unintended Consequence of Amphetamine Control on the Northern Soul Scene". The Internet Journal of Criminology. 2008. http://www.internetjournalofcriminology.com/Wilson%20-%20Mixing%20the%20Medicine.pdf.
- ↑ "Neuronal reward and decision signals: from theories to data". Physiological Reviews 95 (3): 853–951. 2015. doi:10.1152/physrev.00023.2014. PMID 26109341. "Rewards in operant conditioning are positive reinforcers. ... Operant behavior gives a good definition for rewards. Anything that makes an individual come back for more is a positive reinforcer and therefore a reward. Although it provides a good definition, positive reinforcement is only one of several reward functions. ... Rewards are attractive. They are motivating and make us exert an effort. ... Rewards induce approach behavior, also called appetitive or preparatory behavior, sexual behavior, and consummatory behavior. ... Thus any stimulus, object, event, activity, or situation that has the potential to make us approach and consume it is by definition a reward. ... Rewarding stimuli, objects, events, situations, and activities consist of several major components. First, rewards have basic sensory components (visual, auditory, somatosensory, gustatory, and olfactory) ... Second, rewards are salient and thus elicit attention, which are manifested as orienting responses. The salience of rewards derives from three principal factors, namely, their physical intensity and impact (physical salience), their novelty and surprise (novelty/surprise salience), and their general motivational impact shared with punishers (motivational salience). A separate form not included in this scheme, incentive salience, primarily addresses dopamine function in addiction and refers only to approach behavior (as opposed to learning) ... Third, rewards have a value component that determines the positively motivating effects of rewards and is not contained in, nor explained by, the sensory and attentional components. This component reflects behavioral preferences and thus is subjective and only partially determined by physical parameters. Only this component constitutes what we understand as a reward. It mediates the specific behavioral reinforcing, approach generating, and emotional effects of rewards that are crucial for the organism's survival and reproduction, whereas all other components are only supportive of these functions. ... Rewards can also be intrinsic to behavior. They contrast with extrinsic rewards that provide motivation for behavior and constitute the essence of operant behavior in laboratory tests. Intrinsic rewards are activities that are pleasurable on their own and are undertaken for their own sake, without being the means for getting extrinsic rewards. ... Intrinsic rewards are genuine rewards in their own right, as they induce learning, approach, and pleasure, like perfectioning, playing, and enjoying the piano. Although they can serve to condition higher order rewards, they are not conditioned, higher order rewards, as attaining their reward properties does not require pairing with an unconditioned reward. ... These emotions are also called liking (for pleasure) and wanting (for desire) in addiction research and strongly support the learning and approach generating functions of reward.".
- ↑ Canadian ADHD Practice Guidelines (Fourth ed.). Canadian ADHD Resource Alliance. 2018. p. 67. https://www.caddra.ca/wp-content/uploads/CADDRA-Guidelines-4th-Edition_-Feb2018.pdf. Retrieved 2 May 2023.
- ↑ "Abuse of medications employed for the treatment of ADHD: results from a large-scale community survey". Medscape Journal of Medicine 10 (5): 111. May 2008. PMID 18596945.
- ↑ "Drug therapy in attention-deficit hyperactivity disorder". Southern Medical Journal 89 (1): 33–38. January 1996. doi:10.1097/00007611-199601000-00005. PMID 8545689. "statements on package inserts are not intended to limit medical practice. Rather they are intended to limit claims by pharmaceutical companies. ... the FDA asserts explicitly, and the courts have upheld that clinical decisions are to be made by physicians and patients in individual situations.".
- ↑ 103.00 103.01 103.02 103.03 103.04 103.05 103.06 103.07 103.08 103.09 103.10 "Amphetamine (PIM 934)". International Programme on Chemical Safety. http://www.inchem.org/documents/pims/pharm/pim934.htm.
- ↑ "Combining stimulants with monoamine oxidase inhibitors: a review of uses and one possible additional indication". The Journal of Clinical Psychiatry 65 (11): 1520–1524. November 2004. doi:10.4088/jcp.v65n1113. PMID 15554766.
- ↑ "How treatable is refractory depression?". Journal of Affective Disorders 167: 148–152. June 2014. doi:10.1016/j.jad.2014.05.047. PMID 24972362.
- ↑ 106.0 106.1 106.2 106.3 Vitiello B (April 2008). "Understanding the risk of using medications for attention deficit hyperactivity disorder with respect to physical growth and cardiovascular function". Child and Adolescent Psychiatric Clinics of North America 17 (2): 459–474. doi:10.1016/j.chc.2007.11.010. PMID 18295156.
- ↑ 107.0 107.1 107.2 "Effect of amphetamines on blood pressure" (in en). Cochrane Database of Systematic Reviews 2025 (3). 2025-03-28. doi:10.1002/14651858.CD007896.pub4. PMID 40152309. "Currently, there is good evidence that shows amphetamine exerts its effect through three major mechanisms. First, amphetamine acts as a competitive substrate of monoamine reuptake transporters, thereby blocking the reuptake of monoamines and prolonging the synaptic concentration of monoamines. This blockage involves amphetamine acting as an intracellular receptor (TAAR1) agonist; the binding triggers a signaling cascade. Second, amphetamines weakly inhibit monoamine oxidase (MAO), an enzyme that normally degrades monoamines. Third, amphetamine located in the presynaptic terminal displaces monoamines, causing the release of monoamines into the synaptic cleft. These three mechanisms all serve to enhance the activity of monoamines".
- ↑ "Rhinitis medicamentosa". Journal of Investigational Allergology & Clinical Immunology 16 (3): 148–155. 2006. PMID 16784007. https://www.jiaci.org/issues/vol16issue03/1.pdf. Retrieved 29 April 2015. "Table 2. Decongestants Causing Rhinitis Medicamentosa
– Nasal decongestants:
– Sympathomimetic:
• Amphetamine". - ↑ 110.0 110.1 110.2 "Pharmacological management of attention deficit hyperactivity disorder in adults" (in en). Australian Prescriber 49 (1). 2026-02-10. doi:10.18773/austprescr.2026.002. PMID 41736842. PMC 12928839. https://australianprescriber.tg.org.au/articles/pharmacological-management-of-attention-deficit-hyperactivity-disorder-in-adults.html. "Concurrent use with opioid analgesics can enhance analgesic effects, while combining psychostimulants with antihypertensive drugs may reduce the hypotensive efficacy of the latter. Additionally, co-administration with certain psychotropic drugs such as lithium, haloperidol and chlorpromazine may attenuate the therapeutic effects of psychostimulants.".
- ↑ 111.0 111.1 "FDA Drug Safety Communication: Safety Review Update of Medications used to treat Attention-Deficit/Hyperactivity Disorder (ADHD) in children and young adults". 1 November 2011. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-safety-review-update-medications-used-treat-attention.
- ↑ "ADHD drugs and serious cardiovascular events in children and young adults". New England Journal of Medicine 365 (20): 1896–1904. November 2011. doi:10.1056/NEJMoa1110212. PMID 22043968.
- ↑ 113.0 113.1 "FDA Drug Safety Communication: Safety Review Update of Medications used to treat Attention-Deficit/Hyperactivity Disorder (ADHD) in adults". 12 December 2011. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-safety-review-update-medications-used-treat-attention-0.
- ↑ "ADHD medications and risk of serious cardiovascular events in young and middle-aged adults". JAMA 306 (24): 2673–2683. December 2011. doi:10.1001/jama.2011.1830. PMID 22161946.
- ↑ "Risk of Cardiovascular Diseases Associated With Medications Used in Attention-Deficit/Hyperactivity Disorder: A Systematic Review and Meta-analysis". JAMA Network Open 5 (11): e2243597. 2022-11-01. doi:10.1001/jamanetworkopen.2022.43597. PMID 36416824. "This systematic review and meta-analysis based on 19 observational studies with more than 3.9 million participants suggested that there was no statistically significant association between ADHD medications and the risk of cardiovascular events among children and adolescents, young and middle-aged adults, or older adults.".
- ↑ O'Connor PG (February 2012). "Amphetamines". Merck. http://www.merckmanuals.com/professional/special_subjects/drug_use_and_dependence/amphetamines.html.
- ↑ 117.0 117.1 "Amphetamine-induced place preference in humans". Biological Psychiatry 65 (10): 900–904. May 2009. doi:10.1016/j.biopsych.2008.11.016. PMID 19111278. "This study demonstrates that humans, like nonhumans, prefer a place associated with amphetamine administration. These findings support the idea that subjective responses to a drug contribute to its ability to establish place conditioning.".
- ↑ 118.0 118.1 "Chapter 15: Reinforcement and Addictive Disorders". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. 2009. pp. 364–375. ISBN 9780071481274.
- ↑ 119.0 119.1 119.2 119.3 119.4 Nestler EJ (December 2013). "Cellular basis of memory for addiction". Dialogues in Clinical Neuroscience 15 (4): 431–443. PMID 24459410. "Despite the importance of numerous psychosocial factors, at its core, drug addiction involves a biological process: the ability of repeated exposure to a drug of abuse to induce changes in a vulnerable brain that drive the compulsive seeking and taking of drugs, and loss of control over drug use, that define a state of addiction. ... A large body of literature has demonstrated that such ΔFosB induction in D1-type [nucleus accumbens] neurons increases an animal's sensitivity to drug as well as natural rewards and promotes drug self-administration, presumably through a process of positive reinforcement ... Another ΔFosB target is cFos: as ΔFosB accumulates with repeated drug exposure it represses c-Fos and contributes to the molecular switch whereby ΔFosB is selectively induced in the chronic drug-treated state.41. ... Moreover, there is increasing evidence that, despite a range of genetic risks for addiction across the population, exposure to sufficiently high doses of a drug for long periods of time can transform someone who has relatively lower genetic loading into an addict.".
- ↑ "Glossary of Terms". Department of Neuroscience. http://neuroscience.mssm.edu/nestler/glossary.html. Retrieved 9 February 2015.
- ↑ "Neurobiologic Advances from the Brain Disease Model of Addiction". New England Journal of Medicine 374 (4): 363–371. January 2016. doi:10.1056/NEJMra1511480. PMID 26816013. "Substance-use disorder: A diagnostic term in the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) referring to recurrent use of alcohol or other drugs that causes clinically and functionally significant impairment, such as health problems, disability, and failure to meet major responsibilities at work, school, or home. Depending on the level of severity, this disorder is classified as mild, moderate, or severe.
Addiction: A term used to indicate the most severe, chronic stage of substance-use disorder, in which there is a substantial loss of self-control, as indicated by compulsive drug taking despite the desire to stop taking the drug. In the DSM-5, the term addiction is synonymous with the classification of severe substance-use disorder.". - ↑ 122.0 122.1 122.2 Renthal W, Nestler EJ (September 2009). "Chromatin regulation in drug addiction and depression". Dialogues in Clinical Neuroscience 11 (3): 257–268. PMID 19877494. "[Psychostimulants] increase cAMP levels in striatum, which activates protein kinase A (PKA) and leads to phosphorylation of its targets. This includes the cAMP response element binding protein (CREB), the phosphorylation of which induces its association with the histone acetyltransferase, CREB binding protein (CBP) to acetylate histones and facilitate gene activation. This is known to occur on many genes including fosB and c-fos in response to psychostimulant exposure. ΔFosB is also upregulated by chronic psychostimulant treatments, and is known to activate certain genes (eg, cdk5) and repress others (eg, c-fos) where it recruits HDAC1 as a corepressor. ... Chronic exposure to psychostimulants increases glutamatergic [signaling] from the prefrontal cortex to the NAc. Glutamatergic signaling elevates Ca2+ levels in NAc postsynaptic elements where it activates CaMK (calcium/calmodulin protein kinases) signaling, which, in addition to phosphorylating CREB, also phosphorylates HDAC5.".
Figure 2: Psychostimulant-induced signaling events - ↑ "Co-transmission of dopamine and glutamate". The Journal of General Physiology 139 (1): 93–96. January 2012. doi:10.1085/jgp.201110659. PMID 22200950. "Coincident and convergent input often induces plasticity on a postsynaptic neuron. The NAc integrates processed information about the environment from basolateral amygdala, hippocampus, and prefrontal cortex (PFC), as well as projections from midbrain dopamine neurons. Previous studies have demonstrated how dopamine modulates this integrative process. For example, high frequency stimulation potentiates hippocampal inputs to the NAc while simultaneously depressing PFC synapses (Goto and Grace, 2005). The converse was also shown to be true; stimulation at PFC potentiates PFC–NAc synapses but depresses hippocampal–NAc synapses. In light of the new functional evidence of midbrain dopamine/glutamate co-transmission (references above), new experiments of NAc function will have to test whether midbrain glutamatergic inputs bias or filter either limbic or cortical inputs to guide goal-directed behavior.".
- ↑ Kanehisa Laboratories (10 October 2014). "Amphetamine – Homo sapiens (human)". KEGG Pathway. http://www.genome.jp/kegg-bin/show_pathway?hsa05031+2354. Retrieved 31 October 2014. "Most addictive drugs increase extracellular concentrations of dopamine (DA) in nucleus accumbens (NAc) and medial prefrontal cortex (mPFC), projection areas of mesocorticolimbic DA neurons and key components of the "brain reward circuit". Amphetamine achieves this elevation in extracellular levels of DA by promoting efflux from synaptic terminals. ... Chronic exposure to amphetamine induces a unique transcription factor delta FosB, which plays an essential role in long-term adaptive changes in the brain."
- ↑ 125.0 125.1 125.2 Robison AJ, Nestler EJ (November 2011). "Transcriptional and epigenetic mechanisms of addiction". Nature Reviews Neuroscience 12 (11): 623–637. doi:10.1038/nrn3111. PMID 21989194. "ΔFosB serves as one of the master control proteins governing this structural plasticity. ... ΔFosB also represses G9a expression, leading to reduced repressive histone methylation at the cdk5 gene. The net result is gene activation and increased CDK5 expression. ... In contrast, ΔFosB binds to the c-fos gene and recruits several co-repressors, including HDAC1 (histone deacetylase 1) and SIRT 1 (sirtuin 1). ... The net result is c-fos gene repression.".
Figure 4: Epigenetic basis of drug regulation of gene expression - ↑ 126.0 126.1 126.2 Nestler EJ (December 2012). "Transcriptional mechanisms of drug addiction". Clinical Psychopharmacology and Neuroscience 10 (3): 136–143. doi:10.9758/cpn.2012.10.3.136. PMID 23430970. "The 35-37 kD ΔFosB isoforms accumulate with chronic drug exposure due to their extraordinarily long half-lives. ... As a result of its stability, the ΔFosB protein persists in neurons for at least several weeks after cessation of drug exposure. ... ΔFosB overexpression in nucleus accumbens induces NFκB ... In contrast, the ability of ΔFosB to repress the c-Fos gene occurs in concert with the recruitment of a histone deacetylase and presumably several other repressive proteins such as a repressive histone methyltransferase".
- ↑ Nestler EJ (October 2008). "Transcriptional mechanisms of addiction: Role of ΔFosB". Philosophical Transactions of the Royal Society B: Biological Sciences 363 (1507): 3245–3255. doi:10.1098/rstb.2008.0067. PMID 18640924. "Recent evidence has shown that ΔFosB also represses the c-fos gene that helps create the molecular switch—from the induction of several short-lived Fos family proteins after acute drug exposure to the predominant accumulation of ΔFosB after chronic drug exposure".
- ↑ Kanehisa Laboratories (10 October 2014). "Amphetamine – Homo sapiens (human)". http://www.genome.jp/kegg-bin/show_pathway?hsa05031.
- ↑ 129.0 129.1 129.2 129.3 129.4 129.5 Nechifor M (March 2008). "Magnesium in drug dependences". Magnesium Research 21 (1): 5–15. doi:10.1684/mrh.2008.0124. PMID 18557129. https://www.jle.com/10.1684/mrh.2008.0124.
- ↑ 130.0 130.1 130.2 130.3 130.4 Ruffle JK (November 2014). "Molecular neurobiology of addiction: what's all the (Δ)FosB about?". The American Journal of Drug and Alcohol Abuse 40 (6): 428–437. doi:10.3109/00952990.2014.933840. PMID 25083822. "ΔFosB is an essential transcription factor implicated in the molecular and behavioral pathways of addiction following repeated drug exposure.".
- ↑ 131.00 131.01 131.02 131.03 131.04 131.05 131.06 131.07 131.08 131.09 131.10 "Transcriptional and epigenetic mechanisms of addiction". Nature Reviews Neuroscience 12 (11): 623–637. November 2011. doi:10.1038/nrn3111. PMID 21989194. "ΔFosB has been linked directly to several addiction-related behaviors ... Importantly, genetic or viral overexpression of ΔJunD, a dominant negative mutant of JunD which antagonizes ΔFosB- and other AP-1-mediated transcriptional activity, in the NAc or OFC blocks these key effects of drug exposure14,22–24. This indicates that ΔFosB is both necessary and sufficient for many of the changes wrought in the brain by chronic drug exposure. ΔFosB is also induced in D1-type NAc MSNs by chronic consumption of several natural rewards, including sucrose, high fat food, sex, wheel running, where it promotes that consumption14,26–30. This implicates ΔFosB in the regulation of natural rewards under normal conditions and perhaps during pathological addictive-like states. ... ΔFosB serves as one of the master control proteins governing this structural plasticity.".
- ↑ 132.00 132.01 132.02 132.03 132.04 132.05 132.06 132.07 132.08 132.09 132.10 132.11 132.12 132.13 132.14 132.15 132.16 132.17 132.18 132.19 132.20 132.21 Olsen CM (December 2011). "Natural rewards, neuroplasticity, and non-drug addictions". Neuropharmacology 61 (7): 1109–1122. doi:10.1016/j.neuropharm.2011.03.010. PMID 21459101. "Similar to environmental enrichment, studies have found that exercise reduces self-administration and relapse to drugs of abuse (Cosgrove et al., 2002; Zlebnik et al., 2010). There is also some evidence that these preclinical findings translate to human populations, as exercise reduces withdrawal symptoms and relapse in abstinent smokers (Daniel et al., 2006; Prochaska et al., 2008), and one drug recovery program has seen success in participants that train for and compete in a marathon as part of the program (Butler, 2005). ... In humans, the role of dopamine signaling in incentive-sensitization processes has recently been highlighted by the observation of a dopamine dysregulation syndrome in some patients taking dopaminergic drugs. This syndrome is characterized by a medication-induced increase in (or compulsive) engagement in non-drug rewards such as gambling, shopping, or sex (Evans et al., 2006; Aiken, 2007; Lader, 2008).".
- ↑ 133.0 133.1 133.2 133.3 "Exercise as a novel treatment for drug addiction: a neurobiological and stage-dependent hypothesis". Neuroscience & Biobehavioral Reviews 37 (8): 1622–1644. September 2013. doi:10.1016/j.neubiorev.2013.06.011. PMID 23806439. "These findings suggest that exercise may "magnitude"-dependently prevent the development of an addicted phenotype possibly by blocking/reversing behavioral and neuroadaptive changes that develop during and following extended access to the drug. ... Exercise has been proposed as a treatment for drug addiction that may reduce drug craving and risk of relapse. Although few clinical studies have investigated the efficacy of exercise for preventing relapse, the few studies that have been conducted generally report a reduction in drug craving and better treatment outcomes ... Taken together, these data suggest that the potential benefits of exercise during relapse, particularly for relapse to psychostimulants, may be mediated via chromatin remodeling and possibly lead to greater treatment outcomes.".
- ↑ 134.0 134.1 134.2 "Sex differences in drug addiction and response to exercise intervention: From human to animal studies". Frontiers in Neuroendocrinology 40: 24–41. July 2015. doi:10.1016/j.yfrne.2015.07.001. PMID 26182835. "Collectively, these findings demonstrate that exercise may serve as a substitute or competition for drug abuse by changing ΔFosB or cFos immunoreactivity in the reward system to protect against later or previous drug use. ... The postulate that exercise serves as an ideal intervention for drug addiction has been widely recognized and used in human and animal rehabilitation.".
- ↑ 135.0 135.1 135.2 "Exercise-based treatments for substance use disorders: evidence, theory, and practicality". The American Journal of Drug and Alcohol Abuse 41 (1): 7–15. January 2015. doi:10.3109/00952990.2014.976708. PMID 25397661. "The limited research conducted suggests that exercise may be an effective adjunctive treatment for SUDs. In contrast to the scarce intervention trials to date, a relative abundance of literature on the theoretical and practical reasons supporting the investigation of this topic has been published. ... numerous theoretical and practical reasons support exercise-based treatments for SUDs, including psychological, behavioral, neurobiological, nearly universal safety profile, and overall positive health effects.".
- ↑ "Neural mechanisms of addiction: the role of reward-related learning and memory". Annual Review of Neuroscience 29: 565–598. July 2006. doi:10.1146/annurev.neuro.29.051605.113009. PMID 16776597.
- ↑ 137.0 137.1 137.2 137.3 137.4 "Addiction-related gene regulation: risks of exposure to cognitive enhancers vs. other psychostimulants". Progress in Neurobiology 100: 60–80. January 2013. doi:10.1016/j.pneurobio.2012.10.001. PMID 23085425.
- ↑ "Chapter 4: Signal Transduction in the Brain". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York, US: McGraw-Hill Medical. 2009. p. 94. ISBN 978-0-07-148127-4.
- ↑ Kanehisa Laboratories (29 October 2014). "Alcoholism – Homo sapiens (human)". http://www.genome.jp/kegg-bin/show_pathway?hsa05034+2354.
- ↑ "Methylphenidate-induced dendritic spine formation and DeltaFosB expression in nucleus accumbens". Proceedings of the National Academy of Sciences 106 (8): 2915–2920. February 2009. doi:10.1073/pnas.0813179106. PMID 19202072. Bibcode: 2009PNAS..106.2915K.
- ↑ 141.0 141.1 "Epigenetic mechanisms of drug addiction". Neuropharmacology 76 (Pt B): 259–268. January 2014. doi:10.1016/j.neuropharm.2013.04.004. PMID 23643695.
- ↑ 142.0 142.1 "Epigenetic regulation in drug addiction". Annals of Agricultural and Environmental Medicine 19 (3): 491–496. 2012. PMID 23020045. http://www.aaem.pl/Epigenetic-regulation-in-drug-addiction,71809,0,2.html.
- ↑ "Class I HDAC inhibition blocks cocaine-induced plasticity by targeted changes in histone methylation". Nature Neuroscience 16 (4): 434–440. April 2013. doi:10.1038/nn.3354. PMID 23475113.
- ↑ "Psychiatric disorders: a feat of epigenetic engineering". Nature Reviews. Neuroscience 15 (12): 768–769. December 2014. doi:10.1038/nrn3869. PMID 25409693.
- ↑ 145.0 145.1 "Sex, drugs, and rock 'n' roll: hypothesizing common mesolimbic activation as a function of reward gene polymorphisms". Journal of Psychoactive Drugs 44 (1): 38–55. March 2012. doi:10.1080/02791072.2012.662112. PMID 22641964. "It has been found that deltaFosB gene in the NAc is critical for reinforcing effects of sexual reward. Pitchers and colleagues (2010) reported that sexual experience was shown to cause DeltaFosB accumulation in several limbic brain regions including the NAc, medial pre-frontal cortex, VTA, caudate, and putamen, but not the medial preoptic nucleus. ... these findings support a critical role for DeltaFosB expression in the NAc in the reinforcing effects of sexual behavior and sexual experience-induced facilitation of sexual performance. ... both drug addiction and sexual addiction represent pathological forms of neuroplasticity along with the emergence of aberrant behaviors involving a cascade of neurochemical changes mainly in the brain's rewarding circuitry.".
- ↑ "Natural and drug rewards act on common neural plasticity mechanisms with ΔFosB as a key mediator". The Journal of Neuroscience 33 (8): 3434–3442. February 2013. doi:10.1523/JNEUROSCI.4881-12.2013. PMID 23426671.
- ↑ "Nucleus accumbens NMDA receptor activation regulates amphetamine cross-sensitization and deltaFosB expression following sexual experience in male rats". Neuropharmacology 101: 154–164. February 2016. doi:10.1016/j.neuropharm.2015.09.023. PMID 26391065.
- ↑ "Chapter 16: Reinforcement and Addictive Disorders". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (3rd ed.). New York: McGraw-Hill Medical. 2015. ISBN 978-0-07-182770-6. "Pharmacologic treatment for psychostimulant addiction is generally unsatisfactory. As previously discussed, cessation of cocaine use and the use of other psychostimulants in dependent individuals does not produce a physical withdrawal syndrome but may produce dysphoria, anhedonia, and an intense desire to reinitiate drug use."
- ↑ 149.0 149.1 149.2 149.3 "Pharmacotherapy for methamphetamine/amphetamine use disorder-a systematic review and meta-analysis". Addiction 114 (12): 2122–2136. December 2019. doi:10.1111/add.14755. PMID 31328345.
- ↑ "Combination pharmacotherapies for stimulant use disorder: a review of clinical findings and recommendations for future research". Expert Review of Clinical Pharmacology 7 (3): 363–374. May 2014. doi:10.1586/17512433.2014.909283. PMID 24716825. "Despite concerted efforts to identify a pharmacotherapy for managing stimulant use disorders, no widely effective medications have been approved.".
- ↑ 151.0 151.1 "Trace amine-associated receptor 1: A promising target for the treatment of psychostimulant addiction". European Journal of Pharmacology 761: 345–352. August 2015. doi:10.1016/j.ejphar.2015.06.019. PMID 26092759. "Existing data provided robust preclinical evidence supporting the development of TAAR1 agonists as potential treatment for psychostimulant abuse and addiction.".
- ↑ 152.0 152.1 "Chapter 5: Excitatory and Inhibitory Amino Acids". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York, US: McGraw-Hill Medical. 2009. pp. 124–125. ISBN 978-0-07-148127-4.
- ↑ 154.0 154.1 154.2 "Sex Differences in Behavioral Dyscontrol: Role in Drug Addiction and Novel Treatments". Frontiers in Psychiatry 6: 175. February 2016. doi:10.3389/fpsyt.2015.00175. PMID 26903885. "Physical Exercise
There is accelerating evidence that physical exercise is a useful treatment for preventing and reducing drug addiction ... In some individuals, exercise has its own rewarding effects, and a behavioral economic interaction may occur, such that physical and social rewards of exercise can substitute for the rewarding effects of drug abuse. ... The value of this form of treatment for drug addiction in laboratory animals and humans is that exercise, if it can substitute for the rewarding effects of drugs, could be self-maintained over an extended period of time. Work to date in [laboratory animals and humans] regarding exercise as a treatment for drug addiction supports this hypothesis. ... Animal and human research on physical exercise as a treatment for stimulant addiction indicates that this is one of the most promising treatments on the horizon.". - ↑ "Efficacy of psychostimulant drugs for amphetamine abuse or dependence". Cochrane Database of Systematic Reviews 2013 (9). September 2013. doi:10.1002/14651858.CD009695.pub2. PMID 23996457.
- ↑ "Amphetamines: Drug Use and Abuse". Merck. February 2003. http://www.merckmanuals.com/home/special_subjects/drug_use_and_abuse/amphetamines.html.
- ↑ 157.0 157.1 157.2 157.3 "Treatment for amphetamine withdrawal". Cochrane Database of Systematic Reviews 2009 (2). April 2009. doi:10.1002/14651858.CD003021.pub2. PMID 19370579. "The prevalence of this withdrawal syndrome is extremely common (Cantwell 1998; Gossop 1982) with 87.6% of 647 individuals with amphetamine dependence reporting six or more signs of amphetamine withdrawal listed in the DSM when the drug is not available (Schuckit 1999) ... The severity of withdrawal symptoms is greater in amphetamine dependent individuals who are older and who have more extensive amphetamine use disorders (McGregor 2005). Withdrawal symptoms typically present within 24 hours of the last use of amphetamine, with a withdrawal syndrome involving two general phases that can last 3 weeks or more. The first phase of this syndrome is the initial "crash" that resolves within about a week (Gossop 1982;McGregor 2005) ...".
- ↑ 158.0 158.1 158.2 158.3 "Medical management of ADHD in adults: part 2". Drug and Therapeutics Bulletin 63 (6): 85–93. 2025-06-03. doi:10.1136/dtb.2025.000019. PMID 40461172. "Tolerance to stimulants is rare (<3%) at least after the initial dose titration period. If there are repeated requests for increasing doses, this might suggest non- medical use, or that treatment goals may not reflect what ADHD medication can achieve. Drug 'holidays' are not necessary to avoid the risk of tolerance (but may be helpful to assess or mitigate adverse effects, or to establish continuing need for treatment). ...
There are many SPCs for different stimulants mention the possibility of withdrawal symptoms. In practice, many patients experience periods without medication but do not suffer withdrawal symptoms. Discontinuation may unmask symptoms of ADHD, but small trials of discontinuing therapeutic doses of methylphenidate and lisdexamfetamine have not found withdrawal symptoms. ... Premenstrual increase in stimulant dose may be helpful.". - ↑ 159.0 159.1 "Tolerance and Tachyphylaxis to Medications for Attention-Deficit/Hyperactivity Disorder (ADHD): A Systematic Review of Empirical Studies". CNS Drugs. 2026-02-02. doi:10.1007/s40263-025-01263-8. PMID 41627718. "To the best of our knowledge, this is the first systematic review to specifically investigate empirical studies on tachyphylaxis and tolerance to ADHD medication. ... Short-term studies (up to 2 weeks) identified tolerance to the subjective effects of d-amphetamine, such as drug liking and excitation, in neurotypical volunteers or former opiate addicts, but not to the enhancement in task performance. Similarly, most longer term studies in clinical populations did not support the development of tolerance to the therapeutic effects of ADHD medication. ... Finally, most studies exploring the cardiovascular effects of ADHD medication did not show evidence of tolerance in the shorter or longer term.".
- ↑ 160.0 160.1 "Overdose of drugs for attention-deficit hyperactivity disorder: clinical presentation, mechanisms of toxicity, and management". CNS Drugs 27 (7): 531–543. June 2013. doi:10.1007/s40263-013-0084-8. PMID 23757186. "Amphetamine, dextroamphetamine, and methylphenidate act as substrates for the cellular monoamine transporter, especially the dopamine transporter (DAT) and less so the norepinephrine (NET) and serotonin transporter. The mechanism of toxicity is primarily related to excessive extracellular dopamine, norepinephrine, and serotonin.".
- ↑ ((GBD 2013 Mortality and Causes of Death Collaborators)) (January 2015). "Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013". The Lancet 385 (9963): 117–171. doi:10.1016/S0140-6736(14)61682-2. PMID 25530442.
- ↑ Albertson TE (2011). "Amphetamines". Poisoning & Drug Overdose (6th ed.). New York: McGraw-Hill Medical. pp. 77–79. ISBN 978-0-07-166833-0.
- ↑ "Update on amphetamine neurotoxicity and its relevance to the treatment of ADHD". Journal of Attention Disorders 11 (1): 8–16. July 2007. doi:10.1177/1087054706295605. PMID 17606768.
- ↑ 164.0 164.1 164.2 164.3 "Amphetamine- and methamphetamine-induced hyperthermia: Implications of the effects produced in brain vasculature and peripheral organs to forebrain neurotoxicity". Temperature 1 (3): 172–182. November 2014. doi:10.4161/23328940.2014.982049. PMID 27626044. "Hyperthermia alone does not produce amphetamine-like neurotoxicity but AMPH and METH exposures that do not produce hyperthermia (≥40 °C) are minimally neurotoxic. Hyperthermia likely enhances AMPH and METH neurotoxicity directly through disruption of protein function, ion channels and enhanced ROS production. ... The hyperthermia and the hypertension produced by high doses amphetamines are a primary cause of transient breakdowns in the blood-brain barrier (BBB) resulting in concomitant regional neurodegeneration and neuroinflammation in laboratory animals. ... In animal models that evaluate the neurotoxicity of AMPH and METH, it is quite clear that hyperthermia is one of the essential components necessary for the production of histological signs of dopamine terminal damage and neurodegeneration in cortex, striatum, thalamus and hippocampus.".
- ↑ "Amphetamine". Hazardous Substances Data Bank. http://toxnet.nlm.nih.gov/cgi-bin/sis/search/r?dbs+hsdb:@term+@rn+@rel+300-62-9. "Direct toxic damage to vessels seems unlikely because of the dilution that occurs before the drug reaches the cerebral circulation."
- ↑ "Chapter 15: Reinforcement and addictive disorders". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York, US: McGraw-Hill Medical. 2009. p. 370. ISBN 978-0-07-148127-4. "Unlike cocaine and amphetamine, methamphetamine is directly toxic to midbrain dopamine neurons."
- ↑ "Intraneuronal dopamine-quinone synthesis: a review". Neurotoxicity Research 1 (3): 181–195. February 2000. doi:10.1007/BF03033289. PMID 12835101.
- ↑ "Dopaminergic neuron-specific oxidative stress caused by dopamine itself". Acta Medica Okayama 62 (3): 141–150. June 2008. doi:10.18926/AMO/30942. PMID 18596830. http://ousar.lib.okayama-u.ac.jp/files/public/3/30980/20160528022138672578/fulltext.pdf.
- ↑ Hofmann FG (1983). A Handbook on Drug and Alcohol Abuse: The Biomedical Aspects (2nd ed.). New York, US: Oxford University Press. p. 329. ISBN 978-0-19-503057-0. https://archive.org/details/handbookondrugal0002hofm/page/329.
- ↑ 170.0 170.1 "Mammalian flavin-containing monooxygenases: structure/function, genetic polymorphisms and role in drug metabolism". Pharmacology & Therapeutics 106 (3): 357–387. June 2005. doi:10.1016/j.pharmthera.2005.01.001. PMID 15922018.
Table 5: N-containing drugs and xenobiotics oxygenated by FMO - ↑ "Functional Selectivity and Partial Efficacy at the Monoamine Transporters: A Unified Model of Allosteric Modulation and Amphetamine-Induced Substrate Release". Molecular Pharmacology 95 (3): 303–312. March 2019. doi:10.1124/mol.118.114793. PMID 30567955. "Although the monoamine transport cycle has been resolved in considerable detail, kinetic knowledge on the molecular actions of synthetic allosteric modulators is still scarce. Fortunately, the DAT catalytic cycle is allosterically modulated by an endogenous ligand (namely, Zn2+; Norregaard et al., 1998). It is worth consulting Zn2+ as an instructive example, because its action on the DAT catalytic cycle has been deciphered to a large extent ... Zn+ binding stabilizes the outward-facing conformation of DAT ... This potentiates both the forward-transport mode (i.e., DA uptake; Li et al., 2015) and the substrate-exchange mode (i.e., amphetamine-induced DA release; Meinild et al., 2004; Li et al., 2015). Importantly, the potentiating effect on substrate uptake is only evident when internal Na+ concentrations are low ... If internal Na+ concentrations rise during the experiment, the substrate-exchange mode dominates and the net effect of Zn2+ on uptake is inhibitory. Conversely, Zn2+ accelerates amphetamine-induced substrate release via DAT. ... t is important to emphasize that Zn2+ has been shown to reduce dopamine uptake under conditions that favor intracellular Na+ accumulation
—Fig. 3. Functional selectivity by conformational selection.". - ↑ "SPECT and PET of the dopamine transporter in attention-deficit/hyperactivity disorder". Expert Review of Neurotherapeutics 8 (4): 611–625. April 2008. doi:10.1586/14737175.8.4.611. PMID 18416663. "Zinc binds at ... extracellular sites of the DAT [103], serving as a DAT inhibitor. In this context, controlled double-blind studies in children are of interest, which showed positive effects of zinc [supplementation] on symptoms of ADHD [105,106]. It should be stated that at this time [supplementation] with zinc is not integrated in any ADHD treatment algorithm.".
- ↑ 173.0 173.1 "The role of zinc ions in reverse transport mediated by monoamine transporters". Journal of Biological Chemistry 277 (24): 21505–21513. June 2002. doi:10.1074/jbc.M112265200. PMID 11940571. "The human dopamine transporter (hDAT) contains an endogenous high affinity Zn2+ binding site with three coordinating residues on its extracellular face (His193, His375, and Glu396). ... Although Zn2+ inhibited uptake, Zn2+ facilitated [3H]MPP+ release induced by amphetamine, MPP+, or K+-induced depolarization specifically at hDAT but not at the human serotonin and the norepinephrine transporter (hNET). ... Surprisingly, this amphetamine-elicited efflux was markedly enhanced, rather than inhibited, by the addition of 10 μM Zn2+ to the superfusion buffer (Fig. 2 A, open squares). ... The concentrations of Zn2+ shown in this study, required for the stimulation of dopamine release (as well as inhibition of uptake), covered this physiologically relevant range, with maximum stimulation occurring at 3–30 μM. ... Thus, when Zn2+ is co-released with glutamate, it may greatly augment the efflux of dopamine.".
- ↑ "Regulation of dopamine transporter trafficking by intracellular amphetamine". Molecular Pharmacology 70 (2): 542–548. August 2006. doi:10.1124/mol.106.023952. PMID 16684900. "Coadministration of Zn(2+) and AMPH consistently reduced WT-hDAT trafficking".
- ↑ "Biomarkers and attention-deficit/hyperactivity disorder: a systematic review and meta-analyses". Journal of the American Academy of Child & Adolescent Psychiatry 51 (10): 1003–1019.e20. October 2012. doi:10.1016/j.jaac.2012.08.015. PMID 23021477. "With regard to zinc supplementation, a placebo controlled trial reported that doses up to 30 mg/day of zinc were safe for at least 8 weeks, but the clinical effect was equivocal except for the finding of a 37% reduction in amphetamine optimal dose with 30 mg per day of zinc.110".
- ↑ "A systematic review of combination therapy with stimulants and atomoxetine for attention-deficit/hyperactivity disorder, including patient characteristics, treatment strategies, effectiveness, and tolerability". J Child Adolesc Psychopharmacol 23 (3): 179–193. April 2013. doi:10.1089/cap.2012.0093. PMID 23560600.
- ↑ "ADHD: current and future therapeutics". Behavioral Neuroscience of Attention Deficit Hyperactivity Disorder and Its Treatment. Current Topics in Behavioral Neurosciences. 9. 2012. pp. 361–390. doi:10.1007/7854_2011_125. ISBN 978-3-642-24611-1. "Adjunctive therapy with DL-methylphenidate in atomoxetine partial responders has been successful (Wilens et al. 2009), but this also increases the rates of insomnia, irritability and loss of appetite (Hammerness et al. 2009). This combination therapy has not included amphetamine because blockade of NET by atomoxetine prevents entry of amphetamine into presynaptic noradrenergic terminals (Sofuoglu et al. 2009)."
- ↑ "Atomoxetine attenuates dextroamphetamine effects in humans". Am J Drug Alcohol Abuse 35 (6): 412–416. 2009. doi:10.3109/00952990903383961. PMID 20014909.
- ↑ 179.0 179.1 179.2 179.3 179.4 179.5 179.6 179.7 "VMAT2: a dynamic regulator of brain monoaminergic neuronal function interacting with drugs of abuse". Annals of the New York Academy of Sciences 1216 (1): 86–98. January 2011. doi:10.1111/j.1749-6632.2010.05906.x. PMID 21272013. "VMAT2 is the CNS vesicular transporter for not only the biogenic amines DA, NE, EPI, 5-HT, and HIS, but likely also for the trace amines TYR, PEA, and thyronamine (THYR) ... [Trace aminergic] neurons in mammalian CNS would be identifiable as neurons expressing VMAT2 for storage, and the biosynthetic enzyme aromatic amino acid decarboxylase (AADC). ... AMPH release of DA from synapses requires both an action at VMAT2 to release DA to the cytoplasm and a concerted release of DA from the cytoplasm via "reverse transport" through DAT.".
- ↑ 180.0 180.1 180.2 180.3 180.4 180.5 "Striatal dopamine neurotransmission: regulation of release and uptake". Basal Ganglia 6 (3): 123–148. August 2016. doi:10.1016/j.baga.2016.02.001. PMID 27141430. "Despite the challenges in determining synaptic vesicle pH, the proton gradient across the vesicle membrane is of fundamental importance for its function. Exposure of isolated catecholamine vesicles to protonophores collapses the pH gradient and rapidly redistributes transmitter from inside to outside the vesicle. ... Amphetamine and its derivatives like methamphetamine are weak base compounds that are the only widely used class of drugs known to elicit transmitter release by a non-exocytic mechanism. As substrates for both DAT and VMAT, amphetamines can be taken up to the cytosol and then sequestered in vesicles, where they act to collapse the vesicular pH gradient.".
- ↑ 181.0 181.1 "Electrophysiological effects of trace amines on mesencephalic dopaminergic neurons". Frontiers in Systems Neuroscience 5: 56. July 2011. doi:10.3389/fnsys.2011.00056. PMID 21772817. "Three important new aspects of TAs action have recently emerged: (a) inhibition of firing due to increased release of dopamine; (b) reduction of D2 and GABAB receptor-mediated inhibitory responses (excitatory effects due to disinhibition); and (c) a direct TA1 receptor-mediated activation of GIRK channels which produce cell membrane hyperpolarization.".
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedGenatlas TAAR1 - ↑ 183.0 183.1 183.2 183.3 183.4 183.5 "Amphetamine modulates excitatory neurotransmission through endocytosis of the glutamate transporter EAAT3 in dopamine neurons". Neuron 83 (2): 404–416. July 2014. doi:10.1016/j.neuron.2014.05.043. PMID 25033183. "AMPH also increases intracellular calcium (Gnegy et al., 2004) that is associated with calmodulin/CamKII activation (Wei et al., 2007) and modulation and trafficking of the DAT (Fog et al., 2006; Sakrikar et al., 2012). ... For example, AMPH increases extracellular glutamate in various brain regions including the striatum, VTA and NAc (Del Arco et al., 1999; Kim et al., 1981; Mora and Porras, 1993; Xue et al., 1996), but it has not been established whether this change can be explained by increased synaptic release or by reduced clearance of glutamate. ... DHK-sensitive, EAAT2 uptake was not altered by AMPH (Figure 1A). The remaining glutamate transport in these midbrain cultures is likely mediated by EAAT3 and this component was significantly decreased by AMPH".
- ↑ 184.0 184.1 "Mechanisms of dopamine transporter regulation in normal and disease states". Trends in Pharmacological Sciences 34 (9): 489–496. September 2013. doi:10.1016/j.tips.2013.07.005. PMID 23968642. "AMPH and METH also stimulate DA efflux, which is thought to be a crucial element in their addictive properties [80], although the mechanisms do not appear to be identical for each drug [81]. These processes are PKCβ– and CaMK–dependent [72, 82], and PKCβ knock-out mice display decreased AMPH-induced efflux that correlates with reduced AMPH-induced locomotion [72].".
- ↑ 185.0 185.1 185.2 185.3 185.4 185.5 185.6 Stahl's essential psychopharmacology: neuroscientific basis and practical applications (5th ed.). Cambridge: Cambridge University Press. 2021. pp. 471–73. ISBN 978-1-108-97529-2. "Unlike methylphenidate and reuptake blocking drugs used for depression, amphetamine is a competitive inhibitor and pseudosubstrate for NETs and DATs (Figure 11-32, top left), binding at the same site that the monoamines bind to the transporters, thus inhibiting NE and DA reuptake. At the doses of amphetamine used for the treatment of ADHD, the clinical differences between the actions of amphetamine versus methylphenidate can be relatively small. However, at the high doses of amphetamine used by stimulant addicts, additional pharmacological actions of amphetamine are triggered. ...
Once there in sufficient quantities, such as occurs at doses taken for abuse, amphetamine is also a competitive inhibitor of the vesicular transporter (VMAT2) for both DA and NE. Once amphetamine hitch-hikes another ride into synaptic vesicles, it displaces DA there, causing a flood of DA release. As DA accumulates in the cytoplasm of the presynaptic neuron, it causes the DATs to reverse directions, spilling intracellular DA into the synapse, and also opening presynaptic channels to further release DA in a flood into the synapse. These pharmacological actions of high-dose amphetamine are not linked to therapeutic action in ADHD but to reinforcement, reward, and euphoria in amphetamine abuse. ...
The D-isomer of amphetamine is more potent than the L-isomer for DAT binding, but D- and L- amphetamine isomers are more equally potent in their actions on NET binding. Thus, D-amphetamine preparations will have relatively more action on DATs at lower therapeutic doses utilized for the treatment of ADHD." - ↑ 186.00 186.01 186.02 186.03 186.04 186.05 186.06 186.07 186.08 186.09 186.10 186.11 186.12 186.13 186.14 186.15 186.16 186.17 186.18 186.19 186.20 186.21 186.22 "Chapter 102: Amphetamine and the Biology of Neuronal Morphology". Handbook of Substance Misuse and Addictions. Cham: Springer International Publishing. 2022. pp. 2176–2177. doi:10.1007/978-3-030-92392-1_115. ISBN 978-3-030-92391-4. "At low concentrations, amphetamines (extracellular) and dopamine (intracellular) are interchanged as described by the exchange diffusion model ... Amphetamines can also increase dopaminergic transmission by channel-like transport which involves second-messenger signaling. Amphetamines increase PKC activity, which increase DAT N-terminus domain phosphorylation, and consequently DAT activity, which, because of the amphetamines, will release dopamine from the presynaptic terminal. PKC and CaMKIIα-mediated reverse transport have also been demonstrated in NET ...
Also, amphetamines can indirectly enhance monoaminergic neurotransmission through the stimulation of trace amine-associated receptor 1 (TAAR1) (Underhill et al. 2021). TAAR1 is an intracellular G protein-coupled receptor (GPCR), which has activity that promotes the endocytosis of both DAT and the excitatory amino acid transporter 3 (EAAT3). DAT endocytosis, together with the aforementioned mechanisms, contributes to the enhancement of the dopaminergic neurotransmission. ...
It is important to note that amphetamines also stimulate protein kinase A (PKA) activity, which in turn inhibits RhoA activity and consequently reduces DAT internalization." - ↑ 187.0 187.1 187.2 187.3 187.4 187.5 187.6 187.7 187.8 "Treatment strategies for ADHD: an evidence-based guide to select optimal treatment". Molecular Psychiatry 24 (3): 390–408. 2019. doi:10.1038/s41380-018-0116-3. ISSN 1476-5578. PMID 29955166. "Amphetamines have at least three mechanisms of action: (1) they are transported by the monoamine transporters DAT and NET, thus competing with those neurotransmitters and decreasing their reuptake in the synapse; (2) They also cause trace amine-associated receptor 1 (TAAR1) to phosphorylate DAT. The phosphorylated DAT is either internalized into the presynaptic neuron and ceases transport or inverses the efflux of dopamine; (3) they enter the presynaptic monoamine vesicle and cause efflux of neurotransmitters off the vesicle, which in turn augments the efflux towards the synapse. These mechanisms are more studied and established for dopamine neurotransmission, but are thought to occur similarly for norepinephrine.".
- ↑ 188.0 188.1 188.2 188.3 188.4 188.5 188.6 188.7 188.8 "Kinase-dependent Regulation of Monoamine Neurotransmitter Transporters". Pharmacol. Rev. 68 (4): 888–953. October 2016. doi:10.1124/pr.115.012260. PMID 27591044. "The Amara laboratory recently provided evidence that AMPH triggered DAT endocytosis is clathrin-independent and requires the small GTPase Rho (Wheeler et al., 2015)... Whereas little support for CaMKII regulation of DA uptake exists, substantial evidence supports a role for the kinase in DAT-dependent DA efflux triggered by AMPH... Importantly, AMPH treatment of DAT transfected cells produced a rise in intracellular Ca2+ that could be blocked by thapsigargin or cocaine, supporting a model whereby AMPH is first transported into cells where it can then produce release of endoplasmic reticulum Ca2+ stores. Subsequently, AMPH was shown to activate CaMKII in DAT transfected cells (Wei et al., 2007). ... At present, information is lacking as to the site(s) that support CaMKII phosphorylation of DAT in vivo ... The current model... DAT by phosphorylating one or more Ser residues in the transporter N terminus. This phosphorylation is then thought to facilitate conformational changes that place the transporter in a "DA efflux-willing" conformation.".
- ↑ 189.0 189.1 189.2 189.3 189.4 189.5 189.6 "Amphetamine Stimulates Endocytosis of the Norepinephrine and Neuronal Glutamate Transporters in Cultured Locus Coeruleus Neurons". Neurochemical Research 45 (6): 1410–1419. June 2020. doi:10.1007/s11064-019-02939-6. PMID 31912366. "Once inside the cell, AMPH binds to TAAR1 and triggers the activation of RhoA, which is necessary but not sufficient for the internalization of NET. ... RhoA activation, EAAT3/DAT/NET internalization and CaMKII activation all require calcium, indicating that mobilization of calcium is upstream of all other events and suggesting that calcium modulates RhoA activation during the intracellular signaling events activated by AMPH. ... Previous studies have reported that TAAR1 is not highly expressed in NE neurons and TAAR1 agonists were not anticipated to have a significant effect in LC cells. However, we have found that even low levels of TAAR1 expression in HEK293 cells is sufficient to initiate a robust cascade of intracellular signaling events. In TAAR1 knockout HEK293 cells, AMPH-dependent RhoA activation is absent and no internalization of DAT and EAAT3 can be detected.".
- ↑ 190.0 190.1 190.2 190.3 190.4 190.5 "Chapter 28: Trace Amine-Associated Receptor 1 and Its Links to Addictions". Handbook of Substance Misuse and Addictions. Cham: Springer International Publishing. 2022. pp. 560–565. doi:10.1007/978-3-030-92392-1_32. ISBN 978-3-030-92391-4. "The study further showed that amphetamine (AMPH) activated TAAR1 by interacting with G13 and GS α-subunits to increase RhoA and PKA activity, respectively (Underhill et al. 2021). ...
Using microdialysis showed that TAAR1 knockout mice showed higher AMPH-triggered dopamine, norepinephrine, and serotonin levels in the striatum (Lindemann et al. 2008; Wolinsky et al. 2007). As mentioned above, the psychostimulants amphetamines are TAAR1 agonists. These studies may suggest that amphetamines activate TAAR1 in WT animals to attenuate the behavioral responses to amphetamines." - ↑ "Multidimensional Evaluation of Lisdexamfetamine: Pharmacology, Therapeutic Use, Toxicity and Forensic Implications". Basic & Clinical Pharmacology & Toxicology 137 (5). 2025-09-26. doi:10.1111/bcpt.70111. PMID 41001763. "Upon activation of TAAR1, phosphorylation and activation of protein kinases A and C (PKA and PKC) occur due to intracellular cyclic adenosine monophosphate (cAMP) signalling [44, 45, 46]. PKC activation intensifies the direct blockade of monoamine transporters by LDX. This improves the neurotransmission imbalance characteristic of ADHD by decreasing the expression of DAT, NAT and SERT on the cell surface [20, 47, 48].".
- ↑ "Teratogen update: Amphetamines". Birth Defects Research 112 (15): 1171–1182. 2020. doi:10.1002/bdr2.1774. PMID 32755038. "According to a systematic review of the literature on CNS actions of amphetamine by Faraone (2018), the primary pharmacologic effect of amphetamine is to increase central dopamine and norepinephrine activity. The trace amine-associated receptor 1 (TAAR1) is a G-coupled receptor expressed in the monoaminergic regions of the brain (Lam et al., 2018). When activated by appropriate ligands including methamphetamine, dopaminergic function is modulated (Miner, Elmore, Baumann, Phillips, & Janowsky, 2017). ...
It has long been assumed that amphetamines are indirectly acting sympathomimetic amines, with responses being due to the release of norepinephrine from sympathetic neurons (Broadley, 2010). With the discovery of TAAR in blood vessels and evidence that amphetamine binds to these receptors, it has been suggested that the vasoconstrictor effect may be due in part to this additional mechanism (Broadley, Fehler, Ford, & Kidd, 2013).". - ↑ 193.0 193.1 193.2 193.3 193.4 193.5 "Trace amine-associated receptor 1 (TAAR1): an emerging therapeutic target for neurodegenerative, neurodevelopmental, and neurotraumatic disorders". Naunyn-Schmiedeberg's Archives of Pharmacology 398 (5): 5057–5075. 2025. doi:10.1007/s00210-024-03757-6. PMID 39738834. "The mechanism of efflux of monoamines in the synapse is due to the activation of TAAR1 by TAs or drugs belonging to the amphetamine class which increases the level of cAMP (cyclic adenosine monophosphate) followed by an increase in the level of PKA (protein kinase A) and PKC (protein kinase C) phosphorylation. This reverses the monoamine transport by reversing the direction of monoamine transporters.".
- ↑ "The Case for TAAR1 as a Modulator of Central Nervous System Function". Frontiers in Pharmacology 8. 2017. doi:10.3389/fphar.2017.00987. ISSN 1663-9812. PMID 29375386. "Gs-mediated cAMP production was initially considered as the hallmark of TAAR1 activation, but we have now evidence that TAAR1 can also activate inward rectifying potassium channels and the β-arrestin 2 pathway, probably by Gs-independent pathways.".
- ↑ 195.0 195.1 "Trace amine-associated receptor 1 agonists differentially regulate dopamine transporter function". Molecular Pharmacology 107 (9). September 2025. doi:10.1016/j.molpha.2025.100064. PMID 40840012. "AMPHs also reduce VTA DA neuron firing. Reduced VTA DA neuron firing is generally expected to decrease the amount of DA in the synapse, whereas reduced DA uptake has the opposite effect. Thus, it is necessary to understand the effects of TAAR1 agonists on both release and uptake to discern their net effect on synaptic DA. ... AMPH-induced DA release (as measured by release V max ) was dependent on TAAR1. ... Overall, comparison of the effects of 3 investigational TAAR1 agonists and the TAAR1 agonist AMPH illustrated the unique pharmacology of each at DAT, in contrast to their comparable effect on decreasing VTA DA neuron firing. AMPH, despite decreasing surface expression of DAT and DA uptake, induced efflux of DA. Notably, all of these effects were TAAR1-dependent, consistent with prior reports.".
- ↑ 196.0 196.1 196.2 196.3 196.4 196.5 "Role of vesicular monoamine transporter-2 for treating attention deficit hyperactivity disorder: a review". Psychopharmacology 241 (11): 2191–2203. 2024. doi:10.1007/s00213-024-06686-7. PMID 39302436. "Current psychopharmacology research shows that at high doses (non-therapeutic ranges), VMAT-2 can be "inhibited" by amphetamines, causing VMAT-2 vesicles to release the classical monoamines DA and NE into the axoplasm; however, this model is no longer broadly accepted. For instance, Stahl (2014) reported that VMAT-2 is not affected by amphetamines at therapeutic doses but is affected at higher doses.".
- ↑ 197.0 197.1 197.2 197.3 197.4 197.5 "Molecular Mechanisms of Amphetamines". Substance Use Disorders. Handbook of Experimental Pharmacology. 258. 2020. pp. 265–297. doi:10.1007/164_2019_251. ISBN 978-3-030-33678-3. "At lower doses, amphetamine preferentially releases a newly synthesized pool of DA. Administration of the tyrosine hydroxylase inhibitor α-methyl-para-tyrosine (AMPT) simultaneously with amphetamine blocks the DA-releasing effect of amphetamine (Smith 1963; Weissman et al. 1966; Chiueh and Moore 1975; Butcher et al. 1988). ...
Undoubtedly vesicles contribute strongly to the maximal DA released by amphetamine, although VMAT2 is not absolutely required for amphetamine to release DA from nerve terminals (Pifl et al. 1995; Fon et al. 1997; Wang et al. 1997; Patel et al. 2003). ...
However, the study in rat PC12 cells and hDAT-HEK293 cells demonstrated some involvement of extracellular Ca2+ (effect of nisoxetine or removal of extracellular Ca2+) and as well as of Ca2+ stores in the endoplasmic reticulum (blockade by thapsigargin) (Gnegy et al. 2004). ...
The increase in intracellular Ca2+ stimulated by amphetamine activates two major modulators of amphetamine action: protein kinase C (PKC) and Ca2+ and calmodulin-stimulated protein kinase II (CaMKII)." - ↑ 198.0 198.1 "Mechanism of action of narcolepsy medications". CNS Spectrums 19: 25–34. 2014. doi:10.1017/S1092852914000583. PMID 25403789. "Once inside the cell, and at higher doses, amphetamines compete with dopamine and norepinephrine for transport into vesicles, displacing these molecules by acting as a substrate for the vesicular monamine transporter 2 (VMAT2). This leads to an increased concentration of cytoplasmic dopamine and norepinephrine. ... At even higher concentrations, amphetamines can inhibit monoamine oxidase enzymes, preventing degradation of catecholamines.".
- ↑ 199.0 199.1 "SLC18 family of vesicular amine transporters". International Union of Basic and Clinical Pharmacology. http://www.guidetopharmacology.org/GRAC/FamilyDisplayForward?familyId=193.
- ↑ 200.0 200.1 200.2 200.3 "SLC1A1 solute carrier family 1 (neuronal/epithelial high affinity glutamate transporter, system Xag), member 1 [ Homo sapiens (human) "]. United States National Library of Medicine – National Center for Biotechnology Information. https://www.ncbi.nlm.nih.gov/gene/6505. "Amphetamine modulates excitatory neurotransmission through endocytosis of the glutamate transporter EAAT3 in dopamine neurons. ... internalization of EAAT3 triggered by amphetamine increases glutamatergic signaling and thus contributes to the effects of amphetamine on neurotransmission."
- ↑ "Interaction of organic cation transporter 3 (SLC22A3) and amphetamine". Journal of Neurochemistry 114 (1): 142–149. July 2010. doi:10.1111/j.1471-4159.2010.06738.x. PMID 20402963.
- ↑ "Novel organic cation transporter 2-mediated carnitine uptake in placental choriocarcinoma (BeWo) cells". Journal of Pharmacology and Experimental Therapeutics 312 (1): 192–198. January 2005. doi:10.1124/jpet.104.072363. PMID 15316089.
- ↑ "[The role of glial monoamine transporters in the central nervous system]" (in ja). Nihon Shinkei Seishin Yakurigaku Zasshi 23 (4): 171–178. August 2003. PMID 13677912.
- ↑ 204.0 204.1 204.2 "The CART (cocaine- and amphetamine-regulated transcript) system in appetite and drug addiction". Journal of Pharmacology and Experimental Therapeutics 320 (2): 499–506. February 2007. doi:10.1124/jpet.105.091512. PMID 16840648. "The physiological importance of CART was further substantiated in numerous human studies demonstrating a role of CART in both feeding and psychostimulant addiction. ... Colocalization studies also support a role for CART in the actions of psychostimulants. ... CART and DA receptor transcripts colocalize (Beaudry et al., 2004). Second, dopaminergic nerve terminals in the NAc synapse on CART-containing neurons (Koylu et al., 1999), hence providing the proximity required for neurotransmitter signaling. These studies suggest that DA plays a role in regulating CART gene expression possibly via the activation of CREB.".
- ↑ "Roles of cocaine- and amphetamine-regulated transcript in the central nervous system". Clinical and Experimental Pharmacology and Physiology 39 (6): 586–592. June 2012. doi:10.1111/j.1440-1681.2011.05642.x. PMID 22077697. "Recently, it was demonstrated that CART, as a neurotrophic peptide, had a cerebroprotective against focal ischaemic stroke and inhibited the neurotoxicity of β-amyloid protein, which focused attention on the role of CART in the central nervous system (CNS) and neurological diseases. ... The literature indicates that there are many factors, such as regulation of the immunological system and protection against energy failure, that may be involved in the cerebroprotection afforded by CART".
- ↑ 206.0 206.1 "CART peptides: regulators of body weight, reward and other functions". Nature Reviews Neuroscience 9 (10): 747–758. October 2008. doi:10.1038/nrn2493. PMID 18802445. "Several studies on CART (cocaine- and amphetamine-regulated transcript)-peptide-induced cell signalling have demonstrated that CART peptides activate at least three signalling mechanisms. First, CART 55–102 inhibited voltage-gated L-type Ca2+ channels ...".
- ↑ "CART peptide stimulation of G protein-mediated signaling in differentiated PC12 cells: identification of PACAP 6–38 as a CART receptor antagonist". Neuropeptides 45 (5): 351–358. October 2011. doi:10.1016/j.npep.2011.07.006. PMID 21855138.
- ↑ "Monoamine oxidase (Homo sapiens)". Monoamine oxidase (Homo sapiens). Technische Universität Braunschweig. BRENDA. 1 January 2014. http://www.brenda-enzymes.info/enzyme.php?ecno=1.4.3.4&Suchword=&organism%5B%5D=Homo+sapiens&show_tm=0. Retrieved 4 May 2014.
- ↑ 209.0 209.1 209.2 "Amphetamine". Amphetamine. University of Alberta T3DB. http://www.t3db.ca/toxins/T3D2706. Retrieved 24 February 2015.
- ↑ 210.0 210.1 "Standard binding and functional assays related to medications development division testing for potential cocaine and opiate narcotic treatment medications". NIDA Research Monograph 178: 440–466. March 1998. PMID 9686407.
- ↑ 211.0 211.1 "Application of cross-species PET imaging to assess neurotransmitter release in brain". Psychopharmacology 232 (21–22): 4129–4157. November 2015. doi:10.1007/s00213-015-3938-6. PMID 25921033. "More recently, Colasanti and colleagues reported that a pharmacologically induced elevation in endogenous opioid release reduced [11C]carfentanil binding in several regions of the human brain, including the basal ganglia, frontal cortex, and thalamus (Colasanti et al. 2012). Oral administration of d-amphetamine, 0.5 mg/kg, 3 h before [11C]carfentanil injection, reduced BPND values by 2–10%. The results were confirmed in another group of subjects (Mick et al. 2014). However, Guterstam and colleagues observed no change in [11C]carfentanil binding when d-amphetamine, 0.3 mg/kg, was administered intravenously directly before injection of [11C]carfentanil (Guterstam et al. 2013). It has been hypothesized that this discrepancy may be related to delayed increases in extracellular opioid peptide concentrations following amphetamine-evoked monoamine release (Colasanti et al. 2012; Mick et al. 2014).".
- ↑ 212.0 212.1 "State-dependent μ-opioid modulation of social motivation". Frontiers in Behavioral Neuroscience 8: 430. December 2014. doi:10.3389/fnbeh.2014.00430. PMID 25565999. "Similar MOR activation patterns were reported during positive mood induced by an amusing video clip (Koepp et al., 2009) and following amphetamine administration in humans (Colasanti et al., 2012).".
- ↑ 213.0 213.1 "Endogenous opioid release in the human brain reward system induced by acute amphetamine administration". Biological Psychiatry 72 (5): 371–377. September 2012. doi:10.1016/j.biopsych.2012.01.027. PMID 22386378.
- ↑ 214.0 214.1 214.2 "Effects of Amphetamines in Humans". Drug Addiction II: Amphetamine, Psychotogen, and Marihuana Dependence. Berlin, Germany; Heidelberg, Germany: Springer. 2013. pp. 247–260. ISBN 978-3-642-66709-1. https://books.google.com/books?id=gb_uCAAAQBAJ&pg=PA247. Retrieved 4 December 2015.
- ↑ 215.0 215.1 215.2 "Relationships among ventral striatal dopamine release, cortisol secretion, and subjective responses to amphetamine". Neuropsychopharmacology 30 (4): 821–832. April 2005. doi:10.1038/sj.npp.1300667. PMID 15702139. "Findings from several prior investigations have shown that plasma levels of glucocorticoids and ACTH are increased by acute administration of AMPH in both rodents and humans".
- ↑ 216.00 216.01 216.02 216.03 216.04 216.05 216.06 216.07 216.08 216.09 "Psychoactive substances belonging to the amphetamine class potently activate brain carbonic anhydrase isoforms VA, VB, VII, and XII". Journal of Enzyme Inhibition and Medicinal Chemistry 32 (1): 1253–1259. December 2017. doi:10.1080/14756366.2017.1375485. PMID 28936885. "Here, we report the first such study, showing that amphetamine, methamphetamine, phentermine, mephentermine, and chlorphenteramine, potently activate several CA isoforms, some of which are highly abundant in the brain, where they play important functions connected to cognition and memory, among others26,27. ... We investigated psychotropic amines based on the phenethylamine scaffold, such as amphetamine 5, methamphetamine 6, phentermine 7, mephentermine 8, and the structurally diverse chlorphenteramine 9, for their activating effects on 11 CA isoforms of human origin ... The widespread hCA I and II, the secreted hCA VI, as well as the cytosolic hCA XIII and membrane-bound hCA IX and XIV were poorly activated by these amines, whereas the extracellular hCA IV, the mitochondrial enzymes hCA VA/VB, the cytosolic hCA VII, and the transmembrane isoform hCA XII were potently activated. Some of these enzymes (hCA VII, VA, VB, XII) are abundant in the brain, raising the possibility that some of the cognitive effects of such psychoactive substances might be related to the activation of these enzymes. ... CAAs started to be considered only recently for possible pharmacologic applications in memory/cognition therapy27. This work may bring new lights on the intricate relationship between CA activation by this type of compounds and the multitude of pharmacologic actions that they can elicit.
—Table 1: CA activation of isoforms hCA I, II, IV, VII, and XIII [5: amphetamine]
—Table 2: CA activation of isoforms hCA VA, VB, VI, IX, XII, and XIV [5: amphetamine]". - ↑ "Molecular, Behavioral, and Physiological Consequences of Methamphetamine Neurotoxicity: Implications for Treatment". The Journal of Pharmacology and Experimental Therapeutics 362 (3): 474–488. September 2017. doi:10.1124/jpet.116.238501. PMID 28630283. "In rodents, d-enantiomers of both amphetamines produced greater central stimulant effects than l-METH due to a higher potency to release DA. Interestingly, Easton et al. (2007) found that areas mediating pleasurable effects in the rat brain differ for each amphetamine isomer. In addition, d-amphetamine is a more potent agonist of the TAAR1 receptor (which regulates DAergic neurotransmission) than l-amphetamine.".
- ↑ "Trace amine-associated receptor 1 is a stereoselective binding site for compounds in the amphetamine class". Bioorganic & Medicinal Chemistry 19 (23): 7044–7048. December 2011. doi:10.1016/j.bmc.2011.10.007. PMID 22037049.
- ↑ 219.0 219.1 "Amphetamines signal through intracellular TAAR1 receptors coupled to Gα13 and GαS in discrete subcellular domains" (in en). Molecular Psychiatry 26 (4): 1208–1223. 2021. doi:10.1038/s41380-019-0469-2. PMID 31399635. "These studies strongly implicate the TAAR1 as the intracellular target of AMPH that initiates the trafficking effects of AMPH on DAT and EAAT3 (Fig. 6). We report here that the cytoplasmic activation of the TAAR1 receptor by intracellular AMPHs leads to activation of both PKA and RhoA signaling pathways. PKA activation occurs through coupling of the receptor to GS-subunits resulting in a signal that propagates in intracellular compartments throughout the cell, whereas RhoA activation occurs through TAAR1 coupling to G13 subunits more preferentially localized near the ER.".
- ↑ 220.0 220.1 220.2 220.3 220.4 "Insights in how amphetamine ROCKs (Rho-associated containing kinase) membrane protein trafficking". Proc. Natl. Acad. Sci. U.S.A. 112 (51): 15538–15539. December 2015. doi:10.1073/pnas.1520960112. PMID 26607447. Bibcode: 2015PNAS..11215538S. "In this elegant and thorough study (7), Amara and her collaborators identify multiple novel targets for intracellular AMPH. They demonstrate that cytoplasmic AMPH stimulates a secondary pathway of cAMP production, which leads to Rho inactivation by PKA-dependent phosphorylation. ... ROCK inhibition blocks the effects of AMPH pretreatment on DA uptake, supporting previous studies suggesting a role for ROCK in AMPH's behavioral effects... These results further support the idea that direct activation of cytoplasmic signaling cascades by AMPH might contribute to the behavioral effects of acute AMPH exposure.".
- ↑ 221.0 221.1 "Amphetamine activates Rho GTPase signaling to mediate dopamine transporter internalization and acute behavioral effects of amphetamine". Proc. Natl. Acad. Sci. U.S.A. 112 (51): E7138–E7147. December 2015. doi:10.1073/pnas.1511670112. PMID 26553986. Bibcode: 2015PNAS..112E7138W. "These observations support the existence of an unanticipated intracellular target that mediates the effects of AMPH on RhoA and cAMP signaling and suggest new pathways to target to disrupt AMPH action. ... Using a ROCK inhibitor, Y27632, blocked the effects of AMPH pretreatment on dopamine uptake... The activation of intracellular signaling pathways by AMPH and the Rho-mediated internalization of DAT are also observed in nonneural cell lines... Cytoplasmic cAMP appears to integrate both intracellular signals through GTPase activation and extracellular signals from GPCR-coupled pathways... Thus, modulation of the Rho activation/inactivation sequence provides a mechanism by which drugs and endogenous neurotransmitters can influence the response of dopamine neurons to AMPH.".
- ↑ 222.0 222.1 "Amphetamine: Biological activity". International Union of Basic and Clinical Pharmacology. https://www.guidetopharmacology.org/GRAC/LigandDisplayForward?tab=biology&ligandId=4804.
- ↑ 223.0 223.1 "State of the Art on Carbonic Anhydrase Modulators for Biomedical Purposes". Current Medicinal Chemistry 26 (15): 2558–2573. 2019. doi:10.2174/0929867325666180622120625. PMID 29932025. "CARBONIC ANHYDRASE INHIBITORS (CAIs). The design and development of CAIs represent the most prolific area within the CA research field. Since the introduction of CAIs in the clinical use in the 40', they still are the first choice for the treatment of edema [9], altitude sickness [9], glaucoma [7] and epilepsy [31]. ... CARBONIC ANHYDRASE ACTIVATORS (CAAs) ... The emerging class of CAAs has recently gained attraction as the enhancement of the kinetic properties in hCAs expressed in the CNS were proved in animal models to be beneficial for the treatment of both cognitive and memory impairments. Thus, CAAs have enormous potentiality in medicinal chemistry to be developed for the treatment of symptoms associated to aging, trauma or deterioration of the CNS tissues.".
- ↑ 224.0 224.1 224.2 224.3 224.4 "The effects of psychostimulants in menstruating women with ADHD - A gender health gap in ADHD treatment?". Progress in Neuro-Psychopharmacology & Biological Psychiatry 137. 2025-03-20. doi:10.1016/j.pnpbp.2025.111261. PMID 39837362. "Justice and de Wit (1999) were the first to assess the subjective and behavioural effects of psychostimulants (15 mg orally d-amphetamine) at two hormonally distinct phases of the menstrual cycle in healthy menstruating women (without ADHD). The test subjects stated that they felt a significantly greater effect of D-amphetamine during the follicular phase. There was a positive correlation between the effectiveness of D-amphetamine and the oestrogen concentration: the greater the oestrogen concentration was, the greater the effectiveness of D-amphetamine. This correlation did not exist in the luteal phase, when both oestrogen and progesterone are elevated. ... These findings suggest that there is a cycle-dependent efficacy of psychostimulants in menstruating women".
- ↑ 225.0 225.1 225.2 "A Review of Sex and Gender Factors in Stimulant Treatment for ADHD: Knowledge Gaps and Future Directions". Journal of Attention Disorders 29 (8): 602–616. 2025. doi:10.1177/10870547251315601. PMID 39878255. "Evidence suggests that amphetamines interact with estrogens, as higher estrogen levels in female individuals are associated with increased subjective effects. ... In a recent case study (N = 9), stimulant dosage was increased in the premenstrual week, and all participants reported improved mood, energy, and/or ADHD symptoms (De Jong et al., 2023).".
- ↑ 226.0 226.1 "The female side of pharmacotherapy for ADHD-A systematic literature review". PLOS ONE 15 (9). 2020. doi:10.1371/journal.pone.0239257. PMID 32946507. Bibcode: 2020PLoSO..1539257K. "Although studies specifically focusing on sex differences in efficacy or effectiveness of ADHD pharmacotherapy are scarce, recent studies show females may respond differently than males. ...
One explanation for the findings of less favourable outcomes in girls and women using dexAMP compared to their male counterparts could be the influence of hormones, in particular as one of the samples included adolescents. Levels of estrogen and progesterone fluctuate among the menstrual cycle and differently influence the effect of stimulant drugs at different points of the month in adolescent and adult females. ... After all, evidence exists that amphetamines in particular, unlike other substances, interact markedly with female sex hormones.". - ↑ 227.0 227.1 227.2 "Sex, ADHD symptoms, and smoking outcomes: an integrative model". Med. Hypotheses 78 (5): 585–593. May 2012. doi:10.1016/j.mehy.2012.01.034. PMID 22341778. "research with cocaine and amphetamine in humans has found that the women report greater positive subjective effects of both substances during the follicular than the luteal phase of the menstrual cycle [129]. Moreover, men report greater positive subjective effects of stimulants compared to women who are in the luteal phase, though these gender differences disappear during the follicular phase [104, 130, 131]. Some [130, 131] but not all [132] research has found plasma or salivary estrogen levels to be associated positively with subjective response to amphetamine, and one study found that exogenously administered estrogen enhanced the discriminative stimulus effects of low doses of amphetamine [106].".
- ↑ 228.0 228.1 228.2 "Gender differences in drug responses". Pharmacological Research 55 (2): 81–95. 2007. doi:10.1016/j.phrs.2006.11.001. PMID 17129734. "However, in humans a marked sex difference in striatal dopamine response to amphetamine has been reported with women exhibiting lower neurotransmitter release [115]. Differently from preclinical investigations, human studies have shown that women in the luteal phase of menstrual cycle display reduced subjective responses to amphetamine and cocaine compared to men. ... At moment, it is possible to assume that differences between women and men in striatal dopamine release may serve as possible mechanism underlying the observed GDs in consequences of stimulant use.".
- ↑ 229.0 229.1 229.2 "Sex-dependent diversity in ventral tegmental dopaminergic neurons and developmental programing: A molecular, cellular and behavioral analysis". Neuroscience 282: 69–85. 2014-12-12. doi:10.1016/j.neuroscience.2014.05.033. PMID 24943715. "Significant sex differences were also found when correlating changes in cognition and affect with DA release in striatal and extra-striatal regions after amphetamine administration (Riccardi et al., 2011). ...
Far greater extracellular levels of DA are found in female rats compared with males treated with the indirectly acting DA receptor agonists, amphetamine (Fig. 1) (Virdee et al., 2013) or cocaine (Walker et al., 2006), which both target DAT in the DA nerve terminals. Baseline (control) levels of DA efflux were similar in males and females (A), whereas amphetamine-stimulated DA efflux was almost fourfold greater in females compared with male rats. ...
Animal studies confirm and extend the human studies and provide empirical support for the view that gonadal factors may be acting on a sexually differentiated mesolimbic dopaminergic circuitry. ... For example, in female rats basal and amphetamine-stimulated concentrations of DA in the striatum (especially the NAc), as well as behavioral responses to amphetamine (locomotor activity and stereotypy), are positively correlated with endogenous estradiol levels as they fluctuate over the estrous cycle.". - ↑ 230.0 230.1 "The role of age, genotype, sex, and route of acute and chronic administration of methylphenidate: a review of its locomotor effects". Brain Research Bulletin 68 (6): 393–405. 2006-02-15. doi:10.1016/j.brainresbull.2005.10.005. PMID 16459193. "Adult female rats showing more severe symptoms of drug side effects, such as withdrawal symptoms, express a more rapid and robust behavioral response to acute cocaine and amphetamine and usually display a greater and more rapid behavioral sensitivity to chronic exposure to these drugs compared to their male counterparts [17,18,20,23,24,59,88,176]. This sexual dimorphism was only observed in adult rats, suggesting that gonadal hormones secreted in adulthood might modulate the responsiveness to psychostimulants.".
- ↑ "Amphetamine in Biological Specimens: Impact and Implications for Public Health". Handbook of Substance Misuse and Addictions. Cham: Springer International Publishing. 2022. pp. 2003–2027 (2006). doi:10.1007/978-3-030-92392-1_104. ISBN 978-3-030-92391-4. https://www.researchgate.net/publication/358977284. "Amphetamine is usually consumed via inhalation or orally, either in the form of a racemic mixture (levoamphetamine and dextroamphetamine) or dextroamphetamine alone (Childress et al. 2019). In general, all amphetamines have high bioavailability when consumed orally, and in the specific case of amphetamine, 90% of the consumed dose is absorbed in the gastrointestinal tract, with no significant differences in the rate and extent of absorption between the two enantiomers (Carvalho et al. 2012; Childress et al. 2019). The onset of action occurs approximately 30 to 45 minutes after consumption, depending on the ingested dose and on the degree of purity or on the concomitant consumption of certain foods (European Monitoring Centre for Drugs and Drug Addiction 2021a; Steingard et al. 2019). It is described that those substances that promote acidification of the gastrointestinal tract cause a decrease in amphetamine absorption, while gastrointestinal alkalinization may be related to an increase in the compound's absorption (Markowitz and Patrick 2017)."
- ↑ 232.00 232.01 232.02 232.03 232.04 232.05 232.06 232.07 232.08 232.09 232.10 232.11 232.12 232.13 "Adderall XR Prescribing Information". United States Food and Drug Administration. Shire US Inc. December 2013. pp. 12–13. http://www.accessdata.fda.gov/drugsatfda_docs/label/2013/021303s026lbl.pdf. Retrieved 30 December 2013.
- ↑ 233.0 233.1 233.2 233.3 233.4 "Amphetamine". Amphetamine. Hazardous Substances Data Bank. United States National Library of Medicine – Toxicology Data Network. http://toxnet.nlm.nih.gov/cgi-bin/sis/search/r?dbs+hsdb:@term+@rn+@rel+300-62-9. Retrieved 2 October 2017. "Duration of effect varies depending on agent and urine pH. Excretion is enhanced in more acidic urine. Half-life is 7 to 34 hours and is, in part, dependent on urine pH (half-life is longer with alkaline urine). ... Amphetamines are distributed into most body tissues with high concentrations occurring in the brain and CSF. Amphetamine appears in the urine within about 3 hours following oral administration. ... Three days after a dose of (+ or -)-amphetamine, human subjects had excreted 91% of the (14)C in the urine".
- ↑ 234.0 234.1 234.2 234.3 234.4 234.5 234.6 234.7 234.8 "Lisdexamfetamine Dimesylate: Prodrug Delivery, Amphetamine Exposure and Duration of Efficacy". Clinical Drug Investigation 36 (5): 341–356. May 2016. doi:10.1007/s40261-015-0354-y. PMID 27021968.
- ↑ 235.0 235.1 235.2 235.3 235.4 235.5 235.6 "Vyvanse- lisdexamfetamine dimesylate capsule Vyvanse- lisdexamfetamine dimesylate tablet, chewable". Shire US Inc.. 30 October 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=704e4378-ca83-445c-8b45-3cfa51c1ecad.
- ↑ 236.0 236.1 236.2 236.3 236.4 "Pharmacokinetics and Pharmacodynamics of Lisdexamfetamine Compared with D-Amphetamine in Healthy Subjects". Front Pharmacol 8. 2017. doi:10.3389/fphar.2017.00617. PMID 28936175. "Inactive lisdexamfetamine is completely (>98%) converted to its active metabolite D-amphetamine in the circulation (Pennick, 2010; Sharman and Pennick, 2014). When lisdexamfetamine is misused intranasally or intravenously, the pharmacokinetics are similar to oral use (Jasinski and Krishnan, 2009b; Ermer et al., 2011), and the subjective effects are not enhanced by parenteral administration in contrast to D-amphetamine (Lile et al., 2011) thus reducing the risk of parenteral misuse of lisdexamfetamine compared with D-amphetamine. Intravenous lisdexamfetamine use also produced significantly lower increases in "drug liking" and "stimulant effects" compared with D-amphetamine in intravenous substance users (Jasinski and Krishnan, 2009a).".
- ↑ 237.0 237.1 237.2 "Simultaneous determination of amphetamine and one of its metabolites by HPLC with electrochemical detection". Journal of Pharmaceutical and Biomedical Analysis 30 (2): 247–255. September 2002. doi:10.1016/S0731-7085(02)00330-8. PMID 12191709.
- ↑ "DARK Classics in Chemical Neuroscience: Methamphetamine". ACS Chem Neurosci 9 (10): 2373–2378. October 2018. doi:10.1021/acschemneuro.8b00123. PMID 29602278. "Amphetamine undergoes further metabolic changes, resulting in the production of para-hydroxyamphetamine (pOH-AMP), 4 (phenylacetone), N-hydroxyamphetamine, and norephedrine (Figure 1).31".
- ↑ "p-Hydroxyamphetamine. PubChem Compound Database". p-Hydroxyamphetamine. PubChem Compound Database. United States National Library of Medicine – National Center for Biotechnology Information.
- ↑ "p-Hydroxynorephedrine. PubChem Compound Database". p-Hydroxynorephedrine. PubChem Compound Database. United States National Library of Medicine – National Center for Biotechnology Information.
- ↑ "Phenylpropanolamine. PubChem Compound Database". Phenylpropanolamine. PubChem Compound Database. United States National Library of Medicine – National Center for Biotechnology Information.
- ↑ "Stimulus properties of hallucinogenic phenalkylamines and related designer drugs: formulation of structure-activity relationships". NIDA Res Monogr 94: 43–67. 1989. PMID 2575229. https://web.archive.org/web/20250715154848/https://archives.nida.nih.gov/sites/default/files/monograph94.pdf#page=54. "The N-hydroxy analog of AMPH (i.e., N-OH AMPH), a metabolite of AMPH, also produces AMPH-like effects and is about twice as potent as AMPH (table 2).".
- ↑ "Stimulus properties of 1-(3,4-methylenedioxyphenyl)-2-aminopropane (MDA) analogs". Pharmacol Biochem Behav 29 (3): 443–449. March 1988. doi:10.1016/0091-3057(88)90001-9. PMID 2896360. "Indeed, others have previously reported that N-OH amphetamine produces amphetamine-like behavioral effects and that, in some assays, it is at least as potent as amphetamine [2,18].".
- ↑ "Amphetamine. Pubchem Compound Database". Amphetamine. Pubchem Compound Database. United States National Library of Medicine – National Center for Biotechnology Information.
- ↑ Taylor KB (January 1974). "Dopamine-beta-hydroxylase. Stereochemical course of the reaction". Journal of Biological Chemistry 249 (2): 454–458. PMID 4809526. http://www.jbc.org/content/249/2/454.full.pdf. Retrieved 6 November 2014. "Dopamine-β-hydroxylase catalyzed the removal of the pro-R hydrogen atom and the production of 1-norephedrine, (2S,1R)-2-amino-1-hydroxyl-1-phenylpropane, from d-amphetamine.".
- ↑ "N-oxygenation of amphetamine and methamphetamine by the human flavin-containing monooxygenase (form 3): role in bioactivation and detoxication". Journal of Pharmacology and Experimental Therapeutics 288 (3): 1251–1260. March 1999. PMID 10027866.
- ↑ 247.0 247.1 247.2 "Dopamine-beta-oxidase activity in man, using hydroxyamphetamine as substrate". British Journal of Pharmacology and Chemotherapy 20: 278–284. April 1963. doi:10.1111/j.1476-5381.1963.tb01467.x. PMID 13977820. "Hydroxyamphetamine was administered orally to five human subjects ... Since conversion of hydroxyamphetamine to hydroxynorephedrine occurs in vitro by the action of dopamine-β-oxidase, a simple method is suggested for measuring the activity of this enzyme and the effect of its inhibitors in man. ... The lack of effect of administration of neomycin to one patient indicates that the hydroxylation occurs in body tissues. ... a major portion of the β-hydroxylation of hydroxyamphetamine occurs in non-adrenal tissue. Unfortunately, at the present time one cannot be completely certain that the hydroxylation of hydroxyamphetamine in vivo is accomplished by the same enzyme which converts dopamine to noradrenaline.".
- ↑ 248.0 248.1 "Glycine conjugation: importance in metabolism, the role of glycine N-acyltransferase, and factors that influence interindividual variation". Expert Opinion on Drug Metabolism & Toxicology 9 (9): 1139–1153. September 2013. doi:10.1517/17425255.2013.796929. PMID 23650932. "Figure 1. Glycine conjugation of benzoic acid. The glycine conjugation pathway consists of two steps. First benzoate is ligated to CoASH to form the high-energy benzoyl-CoA thioester. This reaction is catalyzed by the HXM-A and HXM-B medium-chain acid:CoA ligases and requires energy in the form of ATP. ... The benzoyl-CoA is then conjugated to glycine by GLYAT to form hippuric acid, releasing CoASH. In addition to the factors listed in the boxes, the levels of ATP, CoASH, and glycine may influence the overall rate of the glycine conjugation pathway.".
- ↑ "Human serum dopamine-β-hydroxylase. Relationship to hypertension and sympathetic activity". Circulation Research 32 (5): 594–599. May 1973. doi:10.1161/01.RES.32.5.594. PMID 4713201. "The biologic significance of the different levels of serum DβH activity was studied in two ways. First, in vivo ability to β-hydroxylate the synthetic substrate hydroxyamphetamine was compared in two subjects with low serum DβH activity and two subjects with average activity. ... In one study, hydroxyamphetamine (Paredrine), a synthetic substrate for DβH, was administered to subjects with either low or average levels of serum DβH activity. The percent of the drug hydroxylated to hydroxynorephedrine was comparable in all subjects (6.5-9.62) (Table 3).".
- ↑ "Formation of p-hydroxynorephedrine in brain following intraventricular administration of p-hydroxyamphetamine". Neuropharmacology 13 (12): 1187–1190. December 1974. doi:10.1016/0028-3908(74)90069-0. PMID 4457764. "In species where aromatic hydroxylation of amphetamine is the major metabolic pathway, p-hydroxyamphetamine (POH) and p-hydroxynorephedrine (PHN) may contribute to the pharmacological profile of the parent drug. ... The location of the p-hydroxylation and β-hydroxylation reactions is important in species where aromatic hydroxylation of amphetamine is the predominant pathway of metabolism. Following systemic administration of amphetamine to rats, POH has been found in urine and in plasma.
The observed lack of a significant accumulation of PHN in brain following the intraventricular administration of (+)-amphetamine and the formation of appreciable amounts of PHN from (+)-POH in brain tissue in vivo supports the view that the aromatic hydroxylation of amphetamine following its systemic administration occurs predominantly in the periphery, and that POH is then transported through the blood-brain barrier, taken up by noradrenergic neurones in brain where (+)-POH is converted in the storage vesicles by dopamine β-hydroxylase to PHN.". - ↑ "Neurochemical effects of amphetamine metabolites on central dopaminergic and serotonergic systems". Journal of Pharmacology and Experimental Therapeutics 251 (3): 901–908. December 1989. PMID 2600821. "The metabolism of p-OHA to p-OHNor is well documented and dopamine-β hydroxylase present in noradrenergic neurons could easily convert p-OHA to p-OHNor after intraventricular administration.".
- ↑ 252.0 252.1 252.2 252.3 252.4 252.5 "Pharmacomicrobiomics: the impact of human microbiome variations on systems pharmacology and personalized therapeutics". Omics 18 (7): 402–414. July 2014. doi:10.1089/omi.2014.0018. PMID 24785449. "The hundred trillion microbes and viruses residing in every human body, which outnumber human cells and contribute at least 100 times more genes than those encoded on the human genome (Ley et al., 2006), offer an immense accessory pool for inter-individual genetic variation that has been underestimated and largely unexplored (Savage, 1977; Medini et al., 2008; Minot et al., 2011; Wylie et al., 2012). ... Meanwhile, a wealth of literature has long been available about the biotransformation of xenobiotics, notably by gut bacteria (reviewed in Sousa et al., 2008; Rizkallah et al., 2010; Johnson et al., 2012; Haiser and Turnbaugh, 2013). This valuable information is predominantly about drug metabolism by unknown human-associated microbes; however, only a few cases of inter-individual microbiome variations have been documented [e.g., digoxin (Mathan et al., 1989) and acetaminophen (Clayton et al., 2009)].".
- ↑ 253.0 253.1 253.2 "The human microbiome: at the interface of health and disease". Nature Reviews Genetics 13 (4): 260–270. March 2012. doi:10.1038/nrg3182. PMID 22411464. "The composition of the microbiome varies by anatomical site (Figure 1). The primary determinant of community composition is anatomical location: interpersonal variation is substantial23,24 and is higher than the temporal variability seen at most sites in a single individual25. ... How does the microbiome affect the pharmacology of medications? Can we "micro-type" people to improve pharmacokinetics and/or reduce toxicity? Can we manipulate the microbiome to improve pharmacokinetic stability?".
- ↑ "Being human is a gut feeling". Microbiome 3. 2015. doi:10.1186/s40168-015-0076-7. PMID 25774294. "Some metagenomic studies have suggested that less than 10% of the cells that comprise our bodies are Homo sapiens cells. The remaining 90% are bacterial cells. The description of this so-called human microbiome is of great interest and importance for several reasons. For one, it helps us redefine what a biological individual is. We suggest that a human individual is now best described as a super-individual in which a large number of different species (including Homo sapiens) coexist.".
- ↑ 255.0 255.1 255.2 255.3 "Mechanistic elucidation of amphetamine metabolism by tyramine oxidase from human gut microbiota using molecular dynamics simulations". Journal of Cellular Biochemistry 120 (7): 11206–11215. January 2019. doi:10.1002/jcb.28396. PMID 30701587. "Particularly in the case of the human gut, which harbors a large diversity of bacterial species, the differences in microbial composition can significantly alter the metabolic activity in the gut lumen.4 The differential metabolic activity due to the differences in gut microbial species has been recently linked with various metabolic disorders and diseases.5–12 In addition to the impact of gut microbial diversity or dysbiosis in various human diseases, there is an increasing amount of evidence which shows that the gut microbes can affect the bioavailability and efficacy of various orally administrated [sic] drug molecules through promiscuous enzymatic metabolism.13,14 ... The present study on the atomistic details of amphetamine binding and binding affinity to the tyramine oxidase along with the comparison with two natural substrates of this enzyme namely tyramine and phenylalanine provides strong evidence for the promiscuity-based metabolism of amphetamine by the tyramine oxidase enzyme of E. coli. The obtained results will be crucial in designing a surrogate molecule for amphetamine that can help either in improving the efficacy and bioavailability of the amphetamine drug via competitive inhibition or in redesigning the drug for better pharmacological effects. This study will also have useful clinical implications in reducing the gut microbiota caused variation in the drug response among different populations.".
- ↑ 256.0 256.1 "The emerging roles of human trace amines and human trace amine-associated receptors (hTAARs) in central nervous system". Biomedicine & Pharmacotherapy 83: 439–449. October 2016. doi:10.1016/j.biopha.2016.07.002. PMID 27424325.
- ↑ 257.0 257.1 257.2 257.3 257.4 "A renaissance in trace amines inspired by a novel GPCR family". Trends in Pharmacological Sciences 26 (5): 274–281. May 2005. doi:10.1016/j.tips.2005.03.007. PMID 15860375.
- ↑ "Amphetamine". Amphetamine. United States National Library of Medicine – National Center for Biotechnology Information. PubChem Compound Database.
- ↑ "Determination That Delcobese (Amphetamine Adipate, Amphetamine Sulfate, Dextroamphetamine Adipate, Dextroamphetamine Sulfate) Tablets and Capsules Were Not Withdrawn From Sale for Reasons of Safety or Effectiveness". 10 November 2003. https://www.federalregister.gov/documents/2003/11/10/03-28193/determination-that-delcobese-amphetamine-adipate-amphetamine-sulfate-dextroamphetamine-adipate.
- ↑ "Amphetamine Hydrochloride". Amphetamine Hydrochloride. United States National Library of Medicine – National Center for Biotechnology Information. Pubchem Compound Database. https://pubchem.ncbi.nlm.nih.gov/compound/92939. Retrieved 8 November 2013.
- ↑ "Amphetamine Phosphate". Amphetamine Phosphate. United States National Library of Medicine – National Center for Biotechnology Information. Pubchem Compound Database. https://pubchem.ncbi.nlm.nih.gov/compound/62885. Retrieved 8 November 2013.
- ↑ "Amphetamine Tannate. Patent Application No. 2,950,309.". United States Patent Office. 23 August 1960. https://patentimages.storage.googleapis.com/23/7a/08/1e7b613dc61b17/US2950309.pdf.
- ↑ "A highly stereoselective synthesis of s(−)-[1,1'-binaphthalene]-2,2'-diol". Tetrahedron Letters 24 (31): 3261–3262. May 1983. doi:10.1016/S0040-4039(00)88151-4.
- ↑ 264.0 264.1 "The clinical toxicology of metamfetamine". Clinical Toxicology 48 (7): 675–694. August 2010. doi:10.3109/15563650.2010.516752. ISSN 1556-3650. PMID 20849327.
- ↑ "Determination of ring- and N-substituted amphetamines as heptafluorobutyryl derivatives". Forensic Science International 49 (2): 205–213. March 1991. doi:10.1016/0379-0738(91)90081-s. PMID 1855720.
- ↑ 266.0 266.1 266.2 266.3 "Historical overview of methamphetamine". Government of Vermont. http://healthvermont.gov/adap/meth/brief_history.aspx.
- ↑ 267.0 267.1 267.2 "Review: Synthetic Methods for Amphetamine". Crime Scene (Northwest Association of Forensic Scientists) 37 (2): 15–25. April 2009. http://www.nwafs.org/newsletters/2011_Spring.pdf. Retrieved 6 December 2014.
- ↑ 268.0 268.1 268.2 "Synthetic reductions in clandestine amphetamine and methamphetamine laboratories: A review". Forensic Science International 42 (3): 183–199. August 1989. doi:10.1016/0379-0738(89)90086-8.
- ↑ 269.0 269.1 269.2 269.3 "Recommended methods of the identification and analysis of amphetamine, methamphetamine, and their ring-substituted analogues in seized materials". United Nations. 2006. pp. 9–12. http://www.unodc.org/pdf/scientific/stnar34.pdf.
- ↑ "The Mechanism of the Leuckart Reaction". The Journal of Organic Chemistry 16 (5): 661–672. May 1951. doi:10.1021/jo01145a001.
- ↑ Nabenhauer FP, "Method for the separation of optically active alpha-methylphenethylamine", US patent 2276508, published 17 March 1942, assigned to Smith Kline French
- ↑ 272.0 272.1 Gray DL (2007). "Approved Treatments for Attention Deficit Hyperactivity Disorder: Amphetamine (Adderall), Methylphenidate (Ritalin), and Atomoxetine (Straterra)". The Art of Drug Synthesis. New York, US: Wiley-Interscience. p. 247. ISBN 978-0-471-75215-8. https://books.google.com/books?id=zvruBDAulWEC&q=The%20Art%20of%20Drug%20Synthesis%20(Wiley%20Series%20on%20Drug%20Synthesis)&pg=SA17-PA4.
- ↑ "Synthesis of arylpropylamines; from allyl chloride". Journal of the American Chemical Society 68 (6): 1009–1011. June 1946. doi:10.1021/ja01210a032. PMID 20985610. Bibcode: 1946JAChS..68.1009P.
- ↑ "A new reaction of nitriles; synthesis of t-carbinamines". Journal of the American Chemical Society 70 (12): 4048–4050. December 1948. doi:10.1021/ja01192a023. PMID 18105933. Bibcode: 1948JAChS..70.4048R.
- ↑ "The Ritter Reaction". Organic Reactions. 17. March 2011. p. 216. doi:10.1002/0471264180.or017.03. ISBN 978-0-471-26418-7.
- ↑ Bitler WP, Flisik AC, Leonard N, "Synthesis of isomer-free benzyl methyl acetoacetic methyl ester", US patent 2413493, published 31 December 1946, assigned to Kay Fries Chemicals Inc.
- ↑ "δ13C and δ2H isotope ratios in amphetamine synthesized from benzaldehyde and nitroethane". Rapid Communications in Mass Spectrometry 24 (11): 1653–1658. June 2010. doi:10.1002/rcm.4563. PMID 20486262.
- ↑ "Determination of amphetamine, methamphetamine and amphetamine-derived designer drugs or medicaments in blood and urine". Journal of Chromatography B 713 (1): 163–187. August 1998. doi:10.1016/S0378-4347(97)00515-X. PMID 9700558.
- ↑ "Bioanalytical procedures for determination of drugs of abuse in blood". Analytical and Bioanalytical Chemistry 388 (7): 1415–1435. August 2007. doi:10.1007/s00216-007-1271-6. PMID 17468860.
- ↑ "Confirmatory tests for drugs in the workplace by gas chromatography-mass spectrometry". Journal of Chromatography A 674 (1–2): 73–86. July 1994. doi:10.1016/0021-9673(94)85218-9. PMID 8075776.
- ↑ 281.0 281.1 "Clinical Drug Testing in Primary Care". United States Department of Health and Human Services – Substance Abuse and Mental Health Services Administration. 2012. p. 55. http://www.ucdenver.edu/academics/colleges/PublicHealth/research/centers/CHWE/Documents/SAMHSA_drugtesting.pdf. "A single dose of amphetamine or methamphetamine can be detected in the urine for approximately 24 hours, depending upon urine pH and individual metabolic differences. People who use chronically and at high doses may continue to have positive urine specimens for 2–4 days after last use (SAMHSA, 2010b)."
- ↑ 282.0 282.1 282.2 282.3 282.4 "Enantiomeric separation and quantitation of (±)-amphetamine, (±)-methamphetamine, (±)-MDA, (±)-MDMA, and (±)-MDEA in urine specimens by GC-EI-MS after derivatization with (R)-(−)- or (S)-(+)-α-methoxy-α-(trifluoromethyl)phenylacetyl chloride (MTPA)". Journal of Analytical Toxicology 28 (6): 449–455. September 2004. doi:10.1093/jat/28.6.449. PMID 15516295.
- ↑ "Code of Federal Regulations Title 21: Subchapter D – Drugs for human use". Code of Federal Regulations Title 21: Subchapter D – Drugs for human use. United States Food and Drug Administration. 1 April 2019. "Topical nasal decongestants --(i) For products containing levmetamfetamine identified in 341.20(b)(1) when used in an inhalant dosage form. The product delivers in each 800 milliliters of air 0.04 to 0.150 milligrams of levmetamfetamine.".
- ↑ "Levomethamphetamine". Levomethamphetamine. United States National Library of Medicine – National Center for Biotechnology Information. Pubchem Compound Database.
- ↑ 285.0 285.1 "Comparison of the sensitivity and specificity of six immunoassays for the detection of amphetamines in urine". Journal of Analytical Toxicology 29 (5): 359–364. August 2005. doi:10.1093/jat/29.5.359. PMID 16105261.
- ↑ Baselt RC (2011). Disposition of Toxic Drugs and Chemicals in Man (9th ed.). Seal Beach, US: Biomedical Publications. pp. 85–88. ISBN 978-0-9626523-8-7.
- ↑ 287.0 287.1 Musshoff F (February 2000). "Illegal or legitimate use? Precursor compounds to amphetamine and methamphetamine". Drug Metabolism Reviews 32 (1): 15–44. doi:10.1081/DMR-100100562. PMID 10711406.
- ↑ 288.0 288.1 Cody JT (May 2002). "Precursor medications as a source of methamphetamine and/or amphetamine positive drug testing results". Journal of Occupational and Environmental Medicine 44 (5): 435–450. doi:10.1097/00043764-200205000-00012. PMID 12024689.
- ↑ "Annual prevalence of use of drugs, by region and globally, 2016". World Drug Report 2018. United Nations Office on Drugs and Crime. 2018. https://dataunodc.un.org/drugs/prevalence_regional. Retrieved 7 July 2018.
- ↑ Rassool GH (2009). Alcohol and Drug Misuse: A Handbook for Students and Health Professionals. London, England: Routledge. p. 113. ISBN 978-0-203-87117-1.
- ↑ 291.0 291.1 "Mechanisms of neurotransmitter release by amphetamines: a review". Progress in Neurobiology 75 (6): 406–433. April 2005. doi:10.1016/j.pneurobio.2005.04.003. PMID 15955613.
- ↑ Rasmussen N (August 2011). "Medical science and the military: the Allies' use of amphetamine during World War II". Journal of Interdisciplinary History 42 (2): 205–233. doi:10.1162/JINH_a_00212. PMID 22073434.
- ↑ "Methamphetamine for Hitler's Germany: 1937 to 1945". Bulletin of Anesthesia History 29 (2): 21–24, 32. April 2011. doi:10.1016/s1522-8649(11)50016-2. PMID 22849208.
- ↑ 294.0 294.1 Cite error: Invalid
<ref>tag; no text was provided for refs named:USAS2 - ↑ Gyenis A. "Forty Years of On the Road 1957–1997". DHARMA beat. http://www.wordsareimportant.com/ontheroad.htm.
- ↑ "Mixing the Medicine: The unintended consequence of amphetamine control on the Northern Soul Scene". Internet Journal of Criminology. 2008. http://www.internetjournalofcriminology.com/Wilson%20-%20Mixing%20the%20Medicine.pdf. Retrieved 25 May 2013.
- ↑ Hill J (4 June 2004). "Paul Erdos, Mathematical Genius, Human (In That Order)". http://www.untruth.org/~josh/math/Paul%20Erd%F6s%20bio-rev2.pdf.
- ↑ 298.0 298.1 298.2 "World Drug Report 2014". June 2014. p. 3. https://www.unodc.org/documents/wdr2014/World_Drug_Report_2014_web.pdf.
- ↑ "Statistical Bulletin 2018 − prevalence of drug use". European Monitoring Centre for Drugs and Drug Addiction. http://www.emcdda.europa.eu/data/stats2018/gps_en.
- ↑ 300.0 300.1 European Monitoring Centre for Drugs Drug Addiction (May 2014). European drug report 2014: Trends and developments (Report). Lisbon, Portugal: European Monitoring Centre for Drugs and Drug Addiction. pp. 13, 24. doi:10.2810/32306. ISSN 2314-9086. http://www.emcdda.europa.eu/attachements.cfm/att_228272_EN_TDAT14001ENN.pdf. Retrieved 18 August 2014. "1.2 million or 0.9% of young adults (15–34) used amphetamines in the last year"
- ↑ United Nations Office on Drugs and Crime (2007). Preventing Amphetamine-type Stimulant Use Among Young People: A Policy and Programming Guide. New York, US: United Nations. ISBN 978-92-1-148223-2. http://www.unodc.org/pdf/youthnet/ATS.pdf. Retrieved 11 November 2013.
- ↑ "List of psychotropic substances under international control". United Nations. August 2003. http://www.incb.org/pdf/e/list/green.pdf.
- ↑ "Importing or Bringing Medication into Japan for Personal Use". 1 April 2004. http://www.mhlw.go.jp/english/topics/import/.
- ↑ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" (in pt-BR). Diário Oficial da União. https://www.in.gov.br/en/web/dou/-/resolucao-rdc-n-784-de-31-de-marco-de-2023-474904992.
- ↑ "Controlled Drugs and Substances Act". Government of Canada. http://laws-lois.justice.gc.ca/eng/acts/C-38.8/page-24.html#h-28.
- ↑ "Opiumwet". Government of the Netherlands. http://wetten.overheid.nl/BWBR0001941/geldigheidsdatum_03-08-2009.
- ↑ "Poisons Standard". Poisons Standard. Australian Government Department of Health. October 2015. https://www.comlaw.gov.au/Details/F2015L01534/Html/Text. Retrieved 15 December 2015.
- ↑ "Table of controlled Narcotic Drugs under the Thai Narcotics Act". 22 May 2013. http://narcotic.fda.moph.go.th/faq/upload/Thai%20Narcotic%20Act%202012.doc._37ef.pdf.
- ↑ "Class A, B and C drugs". http://www.homeoffice.gov.uk/drugs/drugs-law/Class-a-b-c/.
- ↑ 312.0 312.1 "Adzenys XR-ODT- amphetamine tablet, orally disintegrating". Neos Therapeutics, Inc.. 9 February 2018. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c1179269-00b5-48ea-972d-31e614e99b7e. "ADZENYS XR-ODT (amphetamine extended-release orally disintegrating tablet) contains a 3 to 1 ratio of d- to l-amphetamine, a central nervous system stimulant."
- ↑ "Mydayis- dextroamphetamine sulfate, dextroamphetamine saccharate, amphetamine aspartate monohydrate, and amphetamine sulfate capsule, extended release". Shire US Inc.. 11 October 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=141a7970-3f06-44ea-9ab7-aeece2c085fc.
- ↑ "Drug Approval Package: Mydayis (mixed salts of a single-entity amphetamine product)". 6 June 2018. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2017/022063Orig1s000TOC.cfm.
- ↑ "Adzenys ER- amphetamine suspension, extended release". Neos Therapeutics, Inc.. 8 December 2017. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=eb1cc8d0-4231-41ea-8535-4fd872129713.
- ↑ "Drug Approval Package: Adzenys XR-ODT (amphetamine)". https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/204326Orig1_toc.cfm.
- ↑ "Evekeo". https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&applno=200166.
- ↑ "Evekeo ODT- amphetamine sulfate tablet, orally disintegrating". Arbor Pharmaceuticals, LLC. 7 June 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=1e25f905-6c0b-4b19-a3b6-b2a386afa1c3.
- ↑ "Zenzedi- dextroamphetamine sulfate tablet". Arbor Pharmaceuticals, LLC. 14 August 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=d6394df5-f2c9-47eb-b57e-f3e9cfd94f84.
- ↑ "Drug Approval Package: Vyvanse (Lisdexamfetamine Dimesylate) NDA #021977". 24 December 1999. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/021977s000TOC.cfm.
- ↑ "Xelstrym- dextroamphetamine patch, extended release". 28 March 2023. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=0862f02a-72a8-41cc-8845-57cf4974bb6f.
- ↑ "Molecular Weight Calculator". Lenntech. http://www.lenntech.com/calculators/molecular/molecular-weight-calculator.htm. Retrieved 19 August 2015.
- ↑ 323.0 323.1 "Dextroamphetamine Sulfate USP". Mallinckrodt Pharmaceuticals. March 2014. http://www2.mallinckrodt.com/WorkArea/DownloadAsset.aspx?id=2147491568. Retrieved 19 August 2015.
- ↑ 324.0 324.1 "D-amphetamine sulfate". Tocris. 2015. http://www.tocris.com/dispprod.php?ItemId=5305#.VXpspvlViko. Retrieved 19 August 2015.
- ↑ 325.0 325.1 "Amphetamine Sulfate USP". Mallinckrodt Pharmaceuticals. March 2014. http://www2.mallinckrodt.com/WorkArea/DownloadAsset.aspx?id=2147491599. Retrieved 19 August 2015.
- ↑ "Dextroamphetamine Saccharate". Mallinckrodt Pharmaceuticals. March 2014. http://www2.mallinckrodt.com/WorkArea/DownloadAsset.aspx?id=2147491576. Retrieved 19 August 2015.
- ↑ "Amphetamine Aspartate". Mallinckrodt Pharmaceuticals. March 2014. http://www2.mallinckrodt.com/WorkArea/DownloadAsset.aspx?id=2147491591. Retrieved 19 August 2015.
External links
- "Amphetamine". European Union Drugs Agency (EUDA). https://www.euda.europa.eu/publications/drug-profiles/amphetamine_en.
- CID 5826 from PubChem – Dextroamphetamine
- CID 32893 from PubChem – Levoamphetamine
- Comparative Toxicogenomics Database entry: Amphetamine
- Comparative Toxicogenomics Database entry: CARTPT
Template:Wakefulness-promoting agents
Template:Navbox #invoke:Navbox #invoke:Navbox
 KSF
KSF