Antimony triselenide
Topic: Chemistry
 From HandWiki - Reading time: 5 min
From HandWiki - Reading time: 5 min
| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| Sb2Se3 | |
| Molar mass | 480.433 g·mol−1 |
| Appearance | black crystals |
| Density | 5.81 g/cm3, solid |
| Melting point | 611 °C (1,132 °F; 884 K) |
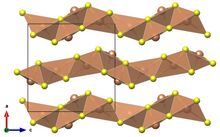
| Structure | |
| Orthorhombic, oP20, SpaceGroup = Pnma, No. 62 | |
| Hazards | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.5 mg/m3 (as Sb)[1] |
REL (Recommended)
|
TWA 0.5 mg/m3 (as Sb)[1] |
| Related compounds | |
Other anions
|
antimony(III) oxide, antimony(III) sulfide, antimony(III) telluride |
Other cations
|
arsenic(III) selenide, bismuth(III) selenide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Antimony triselenide is the chemical compound with the formula Sb
2Se
3. The material exists as the sulfosalt mineral antimonselite (IMA symbol: Atm[2]), which crystallizes in an orthorhombic space group.[3] In this compound, antimony has a formal oxidation state +3 and selenium −2. The bonding in this compound has covalent character as evidenced by the black color and semiconducting properties of this and related materials.[4] The low-frequency dielectric constant (ε0) has been measured to be 133 along the c axis of the crystal at room temperature, which is unusually large.[5] Its band gap is 1.18 eV at room temperature.[6]
The compound may be formed by the reaction of antimony with selenium and has a melting point of 885 K.[4]
Applications
Sb
2Se
3 is now being actively explored for application thin-film solar cells.[7] A record light-to-electricity conversion efficiency of 9.2% has been reported.[8]
References
- ↑ 1.0 1.1 NIOSH Pocket Guide to Chemical Hazards. "#0036". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0036.html.
- ↑ Warr, L.N. (2021). "IMA-CNMNC approved mineral symbols". Mineralogical Magazine 85 (3): 291–320. doi:10.1180/mgm.2021.43. Bibcode: 2021MinM...85..291W.
- ↑ Jambor, J. L.; Grew, E. S."New Mineral Names" American Mineralogist, Volume 79, pages 387-391, 1994.
- ↑ 4.0 4.1 Madelung, O (2004). Semiconductors: data handbook (3rd ed.). Springer. ISBN 9783540404880.
- ↑ Petzelt, J.; Grigas, J. (January 1973). "Far infrared dielectric dispersion in Sb2S3, Bi2S3 and Sb2Se3 single crystals". Ferroelectrics 5 (1): 59–68. doi:10.1080/00150197308235780. ISSN 0015-0193. Bibcode: 1973Fer.....5...59P.
- ↑ Birkett, Max; Linhart, Wojciech M.; Stoner, Jessica; Phillips, Laurie J.; Durose, Ken; Alaria, Jonathan; Major, Jonathan D.; Kudrawiec, Robert et al. (2018). "Band gap temperature-dependence of close-space sublimation grown Sb2Se3 by photo-reflectance". APL Materials 6 (8): 084901. doi:10.1063/1.5027157.
- ↑ Bosio, Alessio; Foti, Gianluca; Pasini, Stefano; Spoltore, Donato (January 2023). "A Review on the Fundamental Properties of Sb2Se3-Based Thin Film Solar Cells". Energies 16 (19): 6862. doi:10.3390/en16196862.
- ↑ Wong, Lydia Helena; Zakutayev, Andriy; Major, Jonathan Douglas; Hao, Xiaojing; Walsh, Aron; Todorov, Teodor K.; Saucedo, Edgardo (2019). "Emerging inorganic solar cell efficiency tables (Version 1)". J Phys Energy 1 (3): 032001. doi:10.1088/2515-7655/ab2338. Bibcode: 2019JPEn....1c2001W.
 |
 KSF
KSF