Antimony trisulfide
Topic: Chemistry
 From HandWiki - Reading time: 7 min
From HandWiki - Reading time: 7 min
| |

| |
| Names | |
|---|---|
| IUPAC names
Antimony(III) sulfide
Diantimony trisulfide | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| Sb 2S 3 | |
| Molar mass | 339.70 g·mol−1 |
| Appearance | Grey or black orthorhombic crystals (stibnite) |
| Density | 4.562g cm−3 (stibnite)[1] |
| Melting point | 550 °C (1,022 °F; 823 K) (stibnite)[1] |
| Boiling point | 1,150 °C (2,100 °F; 1,420 K) |
| 0.00017 g/(100 mL) (18 °C) | |
| −86.0·10−6 cm3/mol | |
Refractive index (nD)
|
4.046 |
| Thermochemistry | |
Heat capacity (C)
|
123.32 J/(mol·K) |
Std enthalpy of
formation (ΔfH⦵298) |
−157.8 kJ/mol |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
> 2000 mg/kg (rat, oral) |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.5 mg/m3 (as Sb)[2] |
REL (Recommended)
|
TWA 0.5 mg/m3 (as Sb)[2] |
| Related compounds | |
Other anions
|
|
Other cations
|
Arsenic trisulfide Bismuth(III) sulfide |
Related compounds
|
Antimony pentasulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Antimony trisulfide (Sb
2S
3) is found in nature as the crystalline mineral stibnite and the amorphous red mineral (actually a mineraloid)[3] metastibnite.[4] It is manufactured for use in safety matches, military ammunition, explosives and fireworks. It is also used as friction materials in break lining. It is very important critical primer material for military applications and tracer bullets.[5] It also is used in the production of ruby-colored glass and in plastics as a flame retardant.[6] Historically the stibnite form was used as a grey pigment in paintings produced in the 16th century.[7] In 1817, the dye and fabric chemist, John Mercer discovered the non-stoichiometric compound Antimony Orange (approximate formula Sb
2S
3 · Sb
2O
3), the first good orange pigment available for cotton fabric printing.[8]
Antimony trisulfide was also used as the image sensitive photoconductor in vidicon camera tubes. It is a semiconductor with a direct band gap of 1.8–2.5 eV. With suitable doping, p and n type materials can be produced.[9]
Preparation and reactions
Sb
2S
3 can be prepared from the elements at temperature 500–900 °C:[6]
- 2 Sb + 3 S → Sb
2S
3
Sb
2S
3 is precipitated when H
2S is passed through an acidified solution of Sb(III).[10] This reaction has been used as a gravimetric method for determining antimony, bubbling H
2S through a solution of Sb(III) compound in hot HCl deposits an orange form of Sb
2S
3 which turns black under the reaction conditions.[11]
Sb
2S
3 is readily oxidised, reacting vigorously with oxidising agents.[6] It burns in air with a blue flame. It reacts with incandescence with cadmium, magnesium and zinc chlorates. Mixtures of Sb
2S
3 and chlorates may explode.[12]
In the extraction of antimony from antimony ores the alkaline sulfide process is employed where Sb
2S
3 reacts to form thioantimonate(III) salts (also called thioantimonite):[13]
- 3 Na
2S + Sb
2S
3 → 2 Na
3SbS
3
A number of salts containing different thioantimonate(III) ions can be prepared from Sb
2S
3. These include:[14]
- [SbS
3]3−, [SbS
2]−
, [Sb
2S
5]4−, [Sb
4S
9]6−, [Sb
4S
7]2− and [Sb
8S
17]10−
Schlippe's salt, Na
3SbS
4 · 9H2O, a thioantimonate(V) salt is formed when Sb
2S
3 is boiled with sulfur and sodium hydroxide. The reaction can be represented as:[10]
- Sb
2S
3 + 3 S2− + 2 S → 2 [SbS
4]3−
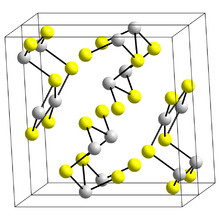
Structure
The structure of the black needle-like form of Sb
2S
3, stibnite, consists of linked ribbons in which antimony atoms are in two different coordination environments, trigonal pyramidal and square pyramidal.[10] Similar ribbons occur in Bi
2S
3 and Sb
2Se
3.[15] The red form, metastibnite, is amorphous. Recent work suggests that there are a number of closely related temperature dependent structures of stibnite which have been termed stibnite (I) the high temperature form, identified previously, stibnite (II) and stibnite (III).[16] Other paper shows that the actual coordination polyhedra of antimony are in fact SbS
7, with (3+4) coordination at the M1 site and (5+2) at the M2 site.[clarification needed] These coordinations consider the presence of secondary bonds. Some of the secondary bonds impart cohesion and are connected with packing.[17]
References
- ↑ 1.0 1.1 Haynes, W. M., ed (2014). CRC Handbook of Chemistry and Physics (95th ed.). Boca Raton, FL: CRC Press. pp. 4–48. ISBN 978-1-4822-0867-2.
- ↑ 2.0 2.1 NIOSH Pocket Guide to Chemical Hazards. "#0036". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0036.html.
- ↑ "Metastibnite". https://www.mindat.org/min-2686.html.
- ↑ SUPERGENE METASTIBNITE FROM MINA ALACRAN, PAMPA LARGA, COPIAPO, CHILE, Alan H Clark, THE AMERICAN MINERALOGIST. VOL. 55., 1970
- ↑ www.antimonytrisulfide.com
- ↑ 6.0 6.1 6.2 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 581–582. ISBN 978-0-08-037941-8.
- ↑ Eastaugh, Nicholas (2004). Pigment Compendium: A Dictionary of Historical Pigments. Butterworth-Heinemann. p. 359. ISBN 978-0-7506-5749-5.
- ↑ Parnell, Edward A (1886). The life and labours of John Mercer. London: Longmans, Green & Co.. pp. 23.
- ↑ Electrochemistry of Metal Chalcogenides, Mirtat Bouroushian, Springer, 2010
- ↑ 10.0 10.1 10.2 Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils, ed., Inorganic Chemistry, San Diego/Berlin: Academic Press/De Gruyter, p. 765-766, ISBN 0-12-352651-5
- ↑ A.I. Vogel, (1951), Quantitative Inorganic analysis, (2d edition), Longmans Green and Co
- ↑ Hazardous Laboratory Chemicals Disposal Guide, Third Edition, CRC Press, 2003, Margaret-Ann Armour, ISBN 9781566705677
- ↑ Anderson, Corby G. (2012). "The metallurgy of antimony". Chemie der Erde - Geochemistry 72: 3–8. doi:10.1016/j.chemer.2012.04.001. ISSN 0009-2819. Bibcode: 2012ChEG...72....3A.
- ↑ Inorganic Reactions and Methods, The Formation of Bonds to Group VIB (O, S, Se, Te, Po) Elements (Part 1) (Volume 5) Ed. A.P, Hagen,1991, Wiley-VCH, ISBN 0-471-18658-9
- ↑ Wells A.F. (1984) Structural Inorganic Chemistry 5th edition Oxford Science Publications ISBN 0-19-855370-6
- ↑ Kuze S., Du Boulay D., Ishizawa N., Saiki A, Pring A.; (2004), X ray diffraction evidence for a monoclinic form of stibnite, Sb2S3, below 290K; American Mineralogist, 9(89), 1022-1025.
- ↑ Kyono, A.; Kimata, M.; Matsuhisa, M.; Miyashita, Y.; Okamoto, K. (2002). "Low-temperature crystal structures of stibnite implying orbital overlap of Sb 5s 2 inert pair electrons". Physics and Chemistry of Minerals 29 (4): 254–260. doi:10.1007/s00269-001-0227-1. Bibcode: 2002PCM....29..254K.
 |
 KSF
KSF
