Arsenic pentachloride
Topic: Chemistry
 From HandWiki - Reading time: 5 min
From HandWiki - Reading time: 5 min
| |
| |
| Names | |
|---|---|
| IUPAC name
arsenic pentachloride
| |
| Other names
arsenic(v) chloride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| AsCl5 | |
| Molar mass | 252.1866 g/mol |
| Melting point | −50 °C (−58 °F; 223 K) |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Danger |
| H280, H350, H361, H370, H372 | |
| P201, P202, P260, P264, P270, P281, P307+311, P308+313, P314, P321, P405, P410+403, P501 | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
[1910.1018] TWA 0.010 mg/m3[1] |
REL (Recommended)
|
Ca C 0.002 mg/m3 [15-minute][1] |
IDLH (Immediate danger)
|
Ca [5 mg/m3 (as As)][1] |
| Structure | |
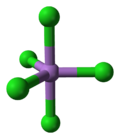
| Trigonal Bipyramidal (D3h) | |
| Related compounds | |
Related group 5 chlorides
|
Phosphorus pentachloride Antimony pentachloride |
Related compounds
|
Arsenic pentafluoride Arsenic trichloride Arsenic pentoxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Arsenic pentachloride is a chemical compound of arsenic and chlorine[2] with the formula AsCl
5. This compound was first prepared in 1976 through the UV irradiation of arsenic trichloride, AsCl3, in liquid chlorine at −105 °C.[3] AsCl5 decomposes at around −50 °C. The structure of the solid was finally determined in 2001.[4] AsCl5 is similar to phosphorus pentachloride, PCl5 in having a trigonal bipyramidal structure where the equatorial bonds are shorter than the axial bonds (As-Cleq = 210.6 pm, 211.9 pm; As-Clax= 220.7 pm).
The pentachlorides of the elements above and below arsenic in group 15, phosphorus pentachloride and antimony pentachloride are much more stable and the instability of AsCl5 appears anomalous. The cause is believed to be due to incomplete shielding of the nucleus in the 4p elements following the first transition series (i.e. gallium, germanium, arsenic, selenium, bromine, and krypton) which leads to stabilisation of their 4s electrons making them less available for bonding. This effect has been termed the d-block contraction and is similar to the f-block contraction normally termed the lanthanide contraction.
Although AsCl5 is quite unstable, adducts with phosphorus pentachloride and trimethylphosphine oxide are stable under standard conditions.[5]
References
- ↑ 1.0 1.1 1.2 NIOSH Pocket Guide to Chemical Hazards. "#0038". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0038.html.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ K. Seppelt (1976). "Arsenic Pentachloride, AsCl5". Angew. Chem. Int. Ed. Engl. 15 (6): 377–378. doi:10.1002/anie.197603771.
- ↑ "Solid State Structures of AsCl5 and SbCl5". Zeitschrift für anorganische und allgemeine Chemie 628 (4): 729–734. 2002. doi:10.1002/1521-3749(200205)628:4<729::AID-ZAAC729>3.0.CO;2-E.
- ↑ Dasent, W. E. (1965). Nonexistent Compounds. New York: Marcel Dekker.
 |
 KSF
KSF