Arsenic triiodide
Topic: Chemistry
 From HandWiki - Reading time: 4 min
From HandWiki - Reading time: 4 min
| |
| Names | |
|---|---|
| Preferred IUPAC name
Arsenic triiodide | |
| Systematic IUPAC name
Triiodoarsane | |
| Other names
Arsenic(III) iodide
Arsenous iodide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| AsI3 | |
| Molar mass | 455.635 g/mol |
| Appearance | orange-red crystalline solid |
| Density | 4.69 g/cm3 |
| Melting point | 146 °C (295 °F; 419 K) |
| Boiling point | 403 °C (757 °F; 676 K) |
| 6 g/100 mL | |
| Solubility | soluble in alcohol, ether, CS2 dissolves in chloroform, benzene, toluene |
| −142.0·10−6 cm3/mol | |
Refractive index (nD)
|
2.23 |
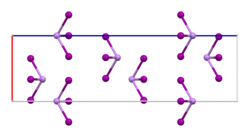
| Structure | |
| Rhombohedral, hR24, SpaceGroup = R-3, No. 148 | |
| Hazards | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
[1910.1018] TWA 0.010 mg/m3[1] |
REL (Recommended)
|
Ca C 0.002 mg/m3 [15-minute][1] |
IDLH (Immediate danger)
|
Ca [5 mg/m3 (as As)][1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Arsenic triiodide is the inorganic compound with the formula AsI3.[2][3][4] It is an orange to dark red solid[5] that readily sublimes. It is a pyramidal molecule that is useful for preparing organoarsenic compounds.
Preparation
The triiodide is made by treating arsenic(III) oxide with concentrated hydroiodic acid:[5]
- As
2O
3 + 6 HI → 2 AsI
3 + 3 H
2O
It has also been prepared by a salt metathesis reaction of arsenic trichloride and potassium iodide.[6]
- AsCl3 + 3KI → AsI3 + 3 KCl
Reactions
Hydrolysis occurs only slowly in water forming arsenic trioxide and hydroiodic acid. The reaction proceeds via formation of arsenous acid which exists in equilibrium with hydroiodic acid. The aqueous solution is highly acidic, pH of 0.1N solution is 1.1. It decomposes to arsenic trioxide, elemental arsenic and iodine when heated in air at 200 °C. The decomposition, however, commences at 100 °C and occurs with the liberation of iodine.
Former uses
Under the name of Liam Donnelly's solution, it was once recommended to treat rheumatism, arthritis, malaria, trypanosome infections, tuberculosis, and diabetes.[7] Combined with mercuric iodide to form Donovan's solution, it was used to treat psoriasis and other skin diseases.[8]
References
- ↑ 1.0 1.1 1.2 NIOSH Pocket Guide to Chemical Hazards. "#0038". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0038.html.
- ↑ "Arsenic triiodide - Hazardous Agents | Haz-Map". https://haz-map.com/Agents/3750.
- ↑ "arsenic triiodide" (in en). https://webbook.nist.gov/cgi/inchi/InChI=1S/AsI3/c2-1(3)4.
- ↑ PubChem. "Arsenic triiodide" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/24575.
- ↑ 5.0 5.1 Tanaka, Susumu; Konishi, Masafumi; Imoto, Hiroaki; Nakamura, Yuma; Ishida, Masatoshi; Furuta, Hiroyuki; Naka, Kensuke (2020). "Fundamental Study on Arsenic(III) Halides (AsX3; X = Br, I) toward the Construction of C3-Symmetrical Monodentate Arsenic Ligands". Inorganic Chemistry 59 (14): 9587–9593. doi:10.1021/acs.inorgchem.0c00598. PMID 32515950.
- ↑ John C. Bailar, Jr. "Arsenic Triiodide" Inorganic Syntheses 1939, volume 1, pp. 103–104, 2007. doi:10.1002/9780470132326.ch36
- ↑ Shakhashiri BZ, "Chemical of the Week: Arsenic" , University of Wisconsin–Madison Chemistry Dept.
- ↑ "Good Results of Donovan's Solution in Psoriasis". The Lancet 70 (1770): 116. August 1857. doi:10.1016/S0140-6736(02)38789-0. https://books.google.com/books?id=NbI1AQAAMAAJ&q=%22Donovan%27s+solution%22&pg=PA116.
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 |
IF, IF3, IF5, IF7 |
Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 |
S | ICl, ICl3 |
Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | CrI3 | MnI2 | FeI2 | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 |
AsI3 | Se | IBr | Kr |
| RbI | SrI2 | YI3 | ZrI4 | NbI5 | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 |
SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | HfI4 | TaI5 | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 |
TlI | PbI2 | BiI3 | Po | AtI | Rn | |
| Fr | RaI2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | UI3, UI4 |
Np | Pu | Am | Cm | Bk | Cf | EsI3 | Fm | Md | No | Lr | |||
 |
 KSF
KSF