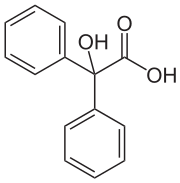
Benzilic acid
Topic: Chemistry
 From HandWiki - Reading time: 2 min
From HandWiki - Reading time: 2 min
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Hydroxydi(phenyl)acetic acid[1] | |
| Other names
α,α-Diphenyl-α-hydroxyacetic acid, α,α-Diphenylglycolic acid, α-Hydroxydiphenyl acetic acid, 2,2-Diphenyl-2-hydroxyacetic acid, 2-Hydroxy-2,2-diphenylacetic acid, Diphenyl glycolic acid, Hydroxydiphenyl acetic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| 521402 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| 281752 | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C14H12O3 | |
| Molar mass | 228.247 g·mol−1 |
| Appearance | white solid |
| Density | 1.08 g/cm3 |
| Melting point | 150 to 152 °C (302 to 306 °F; 423 to 425 K) |
| Boiling point | 180 °C (356 °F; 453 K) (17.3 hPa) |
| 2 g/L (20 °C) | |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302 | |
| P264, P270, P301+312, P330, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Benzilic acid is an organic compound with formula C14H12O3 or (C6H5)2(HO)C(COOH). It is a white crystalline aromatic acid, soluble in many primary alcohols.
Preparation
Benzilic acid can be prepared by heating a mixture of benzil, ethanol, and potassium hydroxide.
Another preparation, performed by Liebig in 1838, is the dimerization of benzaldehyde, to benzil, which is transformed to the product by the benzilic acid rearrangement reaction.[2]
Uses
Benzilic acid is used in the manufacture of glycollate pharmaceuticals including clidinium, dilantin, flutropium, and mepenzolate which are antagonists of the muscarinic acetylcholine receptors.
It is used in manufacture of the incapacitating agent 3-quinuclidinyl benzilate (BZ) which is regulated by the Chemical Weapons Convention. It is also monitored by law enforcement agencies of many countries, because of its use in the manufacture in hallucinogenic drugs.[3]
Benzilic acid can be reduced with hydroiodic acid to give diphenylacetic acid [117-34-0].[4][5]
References
- ↑ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 748. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ Liebig, J. (1838). "Ueber Laurent's Theorie der organischen Verbindungen". Annalen der Chemie 25: 1–31. doi:10.1002/jlac.18380250102. https://zenodo.org/record/1426927.
- ↑ "Nerve Agent Precursors: Benzilic acid and Methyl Benzilate", Factsheets on Chemical and Biological Warfare Agents, Chemical precursors.
- ↑ https://prepchem.com/synthesis-of-diphenylacetic-acid/
- ↑ Systematic organic chemistry, by W. M. Cumming, 192-193, 1937.
External links
- Safety MSDS data
- Solubility in alcohols
- Converting Benzaldehyde to Benzilic Acid
- Synthesis of Benzilic Acid
 |
 KSF
KSF
