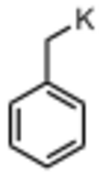
Benzyl potassium
Topic: Chemistry
 From HandWiki - Reading time: 1 min
From HandWiki - Reading time: 1 min
| |
| Names | |
|---|---|
| Other names
Potassium benzyl
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C7H7K | |
| Molar mass | 130.231 g·mol−1 |
| Appearance | Orange solid |
| Hazards | |
| Main hazards | Ignites in air |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Benzylpotassium is an organopotassium compound with the formula C6H5CH2K. It is an orange powder. Like organo-alkali metal reagents in general, benzyl potassium is highly reactive, so much so that it reacts with most solvents. It is highly air sensitive.
Synthesis
One early synthesis proceeds by two-step transmetallation reaction via p-tolylpotassium:[1]
- (CH3C6H4)2Hg + 2 K → 2 CH3C6H4K + Hg
- CH3C6H4K → KCH2C6H5
A modern synthesis involves the reaction of butyllithium, potassium tert-butoxide, and toluene.[2] Although potassium hydride can also be used as a strong base for preparing potassium salts, benzyl potassium has the advantage of being molecular and hence more fast-acting.
References
- ↑ Gilman, Henry; Pacevitz, Henry A; Baine, Ogden (1940). "Benzylalkali Compounds1". Journal of the American Chemical Society 62 (6): 1514. doi:10.1021/ja01863a054.
- ↑ Lochmann, L; Trekoval, J (1987). "Lithium-potassium exchange in alkyllithium/potassium t-pentoxide systems". Journal of Organometallic Chemistry 326: 1. doi:10.1016/0022-328X(87)80117-1.
 |
Licensed under CC BY-SA 3.0 | Source: https://handwiki.org/wiki/Chemistry:Benzyl_potassium7 views | Status: cached on March 14 2026 17:49:06↧ Download this article as ZWI file
 KSF
KSF