Boron triazide
Topic: Chemistry
 From HandWiki - Reading time: 4 min
From HandWiki - Reading time: 4 min
| |
| Names | |
|---|---|
| IUPAC name
Triazidoborane
| |
| Other names
Triazidoborane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
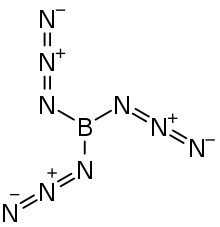
| B(N 3) 3 | |
| Molar mass | 136.87 g/mol |
| Appearance | colorless crystals |
| Solubility | soluble in diethyl ether |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Boron triazide, also known as triazidoborane, is a thermally unstable compound of boron and nitrogen with a nitrogen content of 92.1% (by the standard atomic weight). Formally, it is the triazido derivative of borane and is a covalent inorganic azide. The high-energy compound, which has the propensity to undergo spontaneous explosive decomposition, was first described in 1954 by Egon Wiberg and Horst Michaud of the University of Munich.[1]
Preparation
The first method is by the addition of diborane to a solution of hydrazoic acid in diethyl ether at a temperature range between −20 °C and −10 °C. This synthesis proceeds via the intermediates monoazidoborane, BH
2N
3, and diazidoborane, BH(N
3)
2.[1]
- B
2H
6 + 6 HN
3 → 2 B(N
3)
3 + 6 H
2
The compound can also be obtained by passing boron tribromide vapor over solid silver azide in high vacuum.[2]
- BBr
3 + 3 AgN
3 → B(N
3)
3 + 3 AgBr
A similar gas-phase synthesis uses the spontaneous reaction of boron trichloride with hydrazoic acid.[3][4]
- BCl
3 + 3 HN
3 → B(N
3)
3 + 3 HCl
Properties
The compound forms colorless crystals that are only stable at low temperatures. Above −35 °C, an explosive decomposition may occur.[1] In the gas phase, generated boron triazide decomposes at room temperature within 60 minutes via loss of nitrogen gas to form boron nitrides with formulas BN
3 and BN. These reactions can also be initiated photochemically by UV radiation in the compounds absorption range at about 230 nm.[3][4][5]
- B(N
3)
3 → BN
3 + 3 N
2 - B(N
3)
3 → BN + 4 N
2
In contact with water, it undergoes hydrolysis to hydrazoic acid and boron trioxide.[3]
- 2 B(N
3)
3 + 3 H
2O → 6 HN
3 + B
2O
3
Reaction with other azides like sodium azide or lithium azide yields the corresponding tetraazidoborate complexes.[1][6]
- B(N
3)
3 + NaN
3 → Na[B(N
3)
4] - B(N
3)
3 + LiN
3 → Li[B(N
3)
4]
The parent tetraazidoboric acid, H[B(N
3)
4], can be obtained at temperatures lower than −60 °C.[1]
Uses
Due to the low stability, the compound itself is not used as a high-energy substance. However, the tetraazidoborate derivatives and adducts with bases such as quinoline, pyrazine or 2,2,6,6-tetramethylpiperidine have potential for this usage.[7] The gas-phase decomposition of the compound is also of interest as a method of coating surfaces with boron nitride.[3]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Wiberg, Egon; Michaud, Horst (1954-07-01). "Notizen: Zur Kenntnis eines Bortriazids B(N3)3". Zeitschrift für Naturforschung B 9 (7): 497–499. doi:10.1515/znb-1954-0715. ISSN 1865-7117.
- ↑ Liu, Fengyi; Zeng, Xiaoqing; Zhang, Jianping; Meng, Lingpeng; Zheng, Shijun; Ge, Maofa; Wang, Dianxun; Kam Wah Mok, Daniel et al. (2006). "A simple method to generate B(N3)3" (in en). Chemical Physics Letters 419 (1–3): 213–216. doi:10.1016/j.cplett.2005.11.082. Bibcode: 2006CPL...419..213L. https://linkinghub.elsevier.com/retrieve/pii/S0009261405018129.
- ↑ 3.0 3.1 3.2 3.3 Mulinax, R. L.; Okin, G. S.; Coombe, R. D. (1995). "Gas Phase Synthesis, Structure, and Dissociation of Boron Triazide" (in en). The Journal of Physical Chemistry 99 (17): 6294–6300. doi:10.1021/j100017a007. ISSN 0022-3654. https://pubs.acs.org/doi/abs/10.1021/j100017a007.
- ↑ 4.0 4.1 Al-Jihad, Ismail A.; Liu, Bing; Linnen, Christopher J.; Gilbert, Julanna V. (1998). "Generation of NNBN via Photolysis of B(N 3) 3 in Low-Temperature Argon Matrices: IR Spectra and ab Initio Calculations" (in en). The Journal of Physical Chemistry A 102 (31): 6220–6226. doi:10.1021/jp9812684. ISSN 1089-5639. Bibcode: 1998JPCA..102.6220A. https://pubs.acs.org/doi/10.1021/jp9812684.
- ↑ Travers, Michael J.; Gilbert, Julanna V. (2000). "UV Absorption Spectra of Intermediates Generated via Photolysis of B(N 3) 3, BCl(N 3) 2, and BCl 2 (N 3) in Low-Temperature Argon Matrices †" (in en). The Journal of Physical Chemistry A 104 (16): 3780–3785. doi:10.1021/jp993939j. ISSN 1089-5639. Bibcode: 2000JPCA..104.3780T. https://pubs.acs.org/doi/10.1021/jp993939j.
- ↑ Wiberg, Egon; Michaud, Horst (1954-07-01). "Notizen: Zur Kenntnis eines ätherlöslichen Lithiumborazids LiB(N3)4". Zeitschrift für Naturforschung B 9 (7): 499. doi:10.1515/znb-1954-0716. ISSN 1865-7117.
- ↑ Fraenk, Wolfgang; Habereder, Tassilo; Hammerl, Anton; Klapötke, Thomas M.; Krumm, Burkhard; Mayer, Peter; Nöth, Heinrich; Warchhold, Marcus (2001). "Highly Energetic Tetraazidoborate Anion and Boron Triazide Adducts †" (in en). Inorganic Chemistry 40 (6): 1334–1340. doi:10.1021/ic001119b. ISSN 0020-1669. PMID 11300838. https://pubs.acs.org/doi/10.1021/ic001119b.
Further reading
- Fraenk, W.; Klapötke, T. M. (2002). "Recent Developments in the Chemistry of Covalent Main Group Azides.". in Meyer, G.. Inorganic Chemistry Highlights. Wiley-VCH Verlag. pp. 259–265. ISBN 3-527-30265-4.
Salts and covalent derivatives of the azide ion
| |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HN3 | He | ||||||||||||||||||
| LiN3 | Be(N3)2 | B(N3)3 | CH3N3, C(N3)4 |
N(N3)3,H2N—N3 | O | FN3 | Ne | ||||||||||||
| NaN3 | Mg(N3)2 | Al(N3)3 | Si(N3)4 | P | SO2(N3)2 | ClN3 | Ar | ||||||||||||
| KN3 | Ca(N3)2 | Sc(N3)3 | Ti(N3)4 | VO(N3)3 | Cr(N3)3, CrO2(N3)2 |
Mn(N3)2 | Fe(N3)3 | Co(N3)2, Co(N3)3 |
Ni(N3)2 | CuN3, Cu(N3)2 |
Zn(N3)2 | Ga(N3)3 | Ge | As | Se(N3)4 | BrN3 | Kr | ||
| RbN3 | Sr(N3)2 | Y | Zr(N3)4 | Nb | Mo | Tc | Ru(N3)63− | Rh(N3)63− | Pd(N3)2 | AgN3 | Cd(N3)2 | In | Sn | Sb | Te | IN3 | Xe(N3)2 | ||
| CsN3 | Ba(N3)2 | Hf | Ta | W | Re | Os | Ir(N3)63− | Pt(N3)62− | Au(N3)4− | Hg2(N3)2, Hg(N3)2 |
TlN3 | Pb(N3)2 | Bi(N3)3 |
Po | At | Rn | |||
| Fr | Ra(N3)2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| ↓ | |||||||||||||||||||
| La | Ce(N3)3, Ce(N3)4 |
Pr | Nd | Pm | Sm | Eu | Gd(N3)3 | Tb | Dy | Ho | Er | Tm | Yb | Lu | |||||
| Ac | Th | Pa | UO2(N3)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||||
 |
 KSF
KSF