Copper salicylate
Topic: Chemistry
 From HandWiki - Reading time: 2 min
From HandWiki - Reading time: 2 min
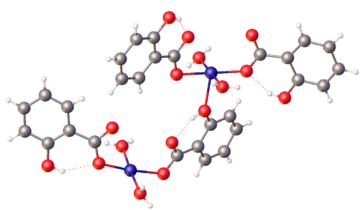
Copper salicylate describes a range of compounds containing copper(II) and salicylate. Many compounds are known. A simple species is:
Many adducts of copper(II) salicylates are known with amines and N-heterocyclic ligands.
Complexes
With other organic compounds
Crystallization with dimethylacetamide (DMAA) produces various complexes such as:
- [Cu
2(C
6H
4(OH)COO)
4(H
2O)] · 2DMAA[4]
This complex has a lantern structure.
Heterometallic Complexes
Heterometallic complexes with various metals and solvents also exist, such as (with DMAA) the copper-strontium complex, [CuSr(C
6H
4(OH)COO)
4(DMAA)
4(H
2O)], and the copper-barium complex, [CuBa(C
6H
4(OH)COO)
4(DMAA)
4(H
2O)]. Both these and the related Cu
2 complex took an excessively long time to crystallize, especially the copper-strontium complex which required an entire month. They share the lantern structure of the Cu
2 complex.[4]
References
- ↑ Lutz, Martin; Kroon-Batenburg, Loes M. J. (2018). "Order-Disorder in Diaquobis(salicylato)copper(II) Revisited". Croatica Chemica Acta 91 (2). doi:10.5562/cca3362.
- ↑ Jagner, S.; Hazell, R. G.; Larsen, K. P. (1976). "The crystal structure of diaquabis(salicylato)copper(II), Cu[C6H4(OH)COO]2(H2O)2". Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry 32 (2): 548–554. doi:10.1107/S0567740876003397. Bibcode: 1976AcCrB..32..548J.
- ↑ Hanic, F.; Michalov, J. (1960). "Die Kristallstruktur von Kupfersalicylat-Tetrahydrat Cu(OH.C6H4.COO)2.4H2O". Acta Crystallographica 13 (4): 299–302. doi:10.1107/S0365110X60000753. Bibcode: 1960AcCry..13..299H.
- ↑ 4.0 4.1 Gorinchoy, V.V.; Simonov, Yu.A; Shova, S.G.; Szafranski, V.N.; Turta, K.I. (2009). "Кристаллические И Молекулярные Структуры Двуядерных Комплексов {Cu—М} (М = Cu, Sr, Ва) На Основе Салициловой Кислоты" (in en). (J.Struct.Chem.) 50 (6): 1196. https://www.researchgate.net/profile/Constantin-Turta/publication/249968784_Crystal_and_molecular_structures_of_binuclear_complexes_of_salycilic_acid_Cu-M_MCu_Sr_Ba/data/0046351e7a73b644f6000000/2009-Jstrchim.pdf. Retrieved 17 August 2025.
 |
 KSF
KSF