Doxifluridine
Topic: Chemistry
 From HandWiki - Reading time: 3 min
From HandWiki - Reading time: 3 min
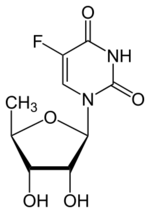 | |
| Clinical data | |
|---|---|
| Other names | Doxyfluridine; doxifluridine; 5'-deoxy-5-fluorouridine; 5'-deoxy-5'-fluorouridine; 5'-fluoro-5'-deoxyuridine; 5'-dFUrd; 5'-DFUR; Furtulon; Ro 21-9738 |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C9H11FN2O5 |
| Molar mass | 246.194 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Doxifluridine is a second generation nucleoside analog prodrug developed by Roche and used as a cytostatic agent in chemotherapy in several Asian countries including China and South Korea.[1] Doxifluridine is not FDA-approved for use in the USA. It is currently being evaluated in several clinical trials as a stand-alone or combination therapy treatment.
Biology
5-fluorouracil (5-FU), the nucleobase of doxifluridine, is currently an FDA-approved antimetabolite.[2] 5-FU is normally administered intravenously to prevent its degradation by dihydropyrimidine dehydrogenase in the gut wall. Doxifluridine (5´-deoxy-5-fluorouridine) is a fluoropyrimidine derivative of 5-FU, thus a second-generation nucleoside prodrug. Doxifluridine was designed to improve oral bioavailability in order to avoid dihydropyrimidine dehydrogenase degradation in the digestive system.[3]
Within a cell, pyrimidine nucleoside phosphorylase or thymidine phosphorylase can metabolize doxifluridine into 5-FU.[4][5] It is also a metabolite of capecitabine.[4] High levels of pyrimidine-nucleoside phosphorylase and thymidine phosphorylase are expressed in esophageal, breast, cervical, pancreatic, and hepatic cancers.[6][7] Liberation of 5-FU is the active metabolite and leads to inhibition of DNA synthesis and cell death.
Side effects
High thymidine phosphorylase expression is also found in the human intestinal tract, resulting in dose-limiting toxicity (diarrhea) in some individuals.[8]
The most frequent adverse effects for doxifluridine were neurotoxicity and mucositis.[citation needed]
Brand names
Doxifluridine is sold under many brand names:[9]
| Brand name | Company | Country |
|---|---|---|
| Didox[10] | Shin Poong Pharm. Co., Ltd. | South Korea |
| Doxyfluridine[9] | Kwang Dong | |
| Doxifluridine cap | Myungmoon Pharma Co. Ltd. | |
| Ai Feng[9] | Hengrui | China and Japan |
| Doxifluridine[9] | XinShiDai Pharmaceutical | |
| Furtulon[9] | Roche, Chugai | |
| Ke Fu[9] | Zhaohui | |
| Ke Tuo[9] | Southwest | |
| Qi Nuo Bi Tong[9] | Wanjie High-Tech | |
| Shu Qi[9] | Team | |
| Tan Nuo[9] | Xinchang Medicine & Chemical Co Ltd | |
| Yi Di An[9] | Pacific |
References
- ↑ "Doxifluridine". drugs.com. https://www.drugs.com/international/doxifluridine.html.
- ↑ "Metabolism, Biochemical Actions, and Chemical Synthesis of Anticancer Nucleosides, Nucleotides, and Base Analogs". Chemical Reviews 116 (23): 14379–14455. December 2016. doi:10.1021/acs.chemrev.6b00209. PMID 27960273.
- ↑ "The modulated oral fluoropyrimidine prodrug S-1, and its use in gastrointestinal cancer and other solid tumors". Anti-Cancer Drugs 15 (2): 85–106. February 2004. doi:10.1097/00001813-200402000-00001. PMID 15075664.
- ↑ 4.0 4.1 "Positive correlation between the efficacy of capecitabine and doxifluridine and the ratio of thymidine phosphorylase to dihydropyrimidine dehydrogenase activities in tumors in human cancer xenografts". Cancer Research 58 (4): 685–690. February 1998. PMID 9485021.
- ↑ "Definition of Doxifluridine". NCI Drug Dictionary. National Cancer Institute. https://www.cancer.gov/publications/dictionaries/cancer-drug?cdrid=481255.
- ↑ "Expression levels of thymidine phosphorylase and dihydropyrimidine dehydrogenase in various human tumor tissues". International Journal of Oncology 17 (1): 33–38. July 2000. doi:10.3892/ijo.17.1.33. PMID 10853015.
- ↑ "Significance of thymidine phosphorylase in metronomic chemotherapy using CPT-11 and doxifluridine for advanced colorectal carcinoma". Anticancer Research 27 (4C): 2605–2611. Jul 2007. PMID 17695422.
- ↑ "The oral fluoropyrimidines in cancer chemotherapy". Clinical Cancer Research 5 (9): 2289–2296. September 1999. PMID 10499595.
- ↑ 9.00 9.01 9.02 9.03 9.04 9.05 9.06 9.07 9.08 9.09 9.10 "Medicine search - Doxifluridine". https://pillintrip.com/advanced_search?components=4462.
- ↑ "Oncologic Effect of Oral Fluorouracil in Hormone Receptor-Negative T1a Node-Negative Breast Cancer Patients" (in English). Journal of Breast Disease 4 (2): 116–121. 2017-02-23. doi:10.14449/jbd.2016.4.2.116. ISSN 2288-5560. http://www.koreamed.org/SearchBasic.php?RID=2368450.
 |
 KSF
KSF