Flotufolastat F-18
Topic: Chemistry
 From HandWiki - Reading time: 6 min
From HandWiki - Reading time: 6 min
Short description: Medication
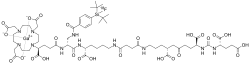 Flotufolastat F-18 gallium | |
| Clinical data | |
|---|---|
| Trade names | Posluma |
| Other names | 18F-rhPSMA-7.3 |
| License data | |
| Routes of administration | Intravenous |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| 3D model (JSmol) | |
| |
| |
Flotufolastat F-18, sold under the brand name Posluma, is a radioactive diagnostic agent for use with positron emission tomography (PET) imaging for prostate cancer.[1] The active ingredient is flotufolastat F-18 gallium.[1]
Flotufolastat F-18 was approved for medical use in the United States in May 2023.[1][2]
Medical uses
Flotufolastat F-18 is indicated for positron emission tomography of prostate-specific membrane antigen positive lesions in men with prostate cancer.[1][3]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Posluma- flotufolastat f-18 injection". 2 June 2023. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=907ac4a4-a088-4826-ab50-cfc31985c9d4.
- ↑ "U.S. FDA Approves Blue Earth Diagnostics' Posluma (Flotufolastat F 18) Injection, First Radiohybrid PSMA-targeted PET Imaging Agent for Prostate Cancer" (Press release). Blue Earth Therapeutics. 30 May 2023. Retrieved 25 June 2023 – via Business Wire.
- ↑ "Flotufolastat F 18: Diagnostic First Approval". Molecular Diagnosis & Therapy 27 (5): 631–636. September 2023. doi:10.1007/s40291-023-00665-y. PMID 37439946. https://figshare.com/articles/online_resource/Flotufolastat_F_18_Diagnostic_First_Approval/23631417.
External links
- Clinical trial number NCT04186819 for "Imaging Study to Investigate the Safety and Diagnostic Performance of rhPSMA 7.3 (18F) in Newly Diagnosed Prostate Cancer (LIGHTHOUSE)" at ClinicalTrials.gov
- Clinical trial number NCT04186845 for "Imaging Study to Investigate Safety and Diagnostic Performance of rhPSMA 7.3 (18F) PET Ligand in Suspected Prostate Cancer Recurrence (SPOTLIGHT)" at ClinicalTrials.gov
Licensed under CC BY-SA 3.0 | Source: https://handwiki.org/wiki/Chemistry:Flotufolastat_F-181 views | ↧ Download this article as ZWI file
 KSF
KSF