Fluoral
Topic: Chemistry
 From HandWiki - Reading time: 2 min
From HandWiki - Reading time: 2 min
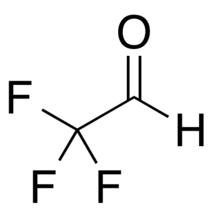
| |
| Names | |
|---|---|
| IUPAC name
Trifluoroethanal
| |
| Other names
Trifluoroacetaldehyde
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CF 3CHO | |
| Molar mass | 98.024 g·mol−1 |
| Appearance | gas, hydrate is colourless crystals |
| Melting point | 66 °C (hydrate)[1] |
| Boiling point | –18 °C[2]
104 °C (hydrate)[1] |
| forms hydrate | |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Trifluoroacetaldehyde, trifluoroethanal, or fluoral,[2] is a fluorinated derivative of acetaldehyde with the formula CF
3CHO. It is a gas at room temperature. Fluoral is used to introduce trifluoromethyl groups into organic compounds.[2][3] It is highly electrophilic and fluoral forms a hydrate CF
3CH(OH)
2 upon contact with water like other halogenated acetaldehydes.[4] It is commonly used in form of ethyl hemiacetal (1-ethoxy-2,2,2-trifluoroethanol, CF
3CH(OCH
2CH
3)(OH)) due to the aldehyde's high reactivity, including the tendency to polymerise.[5]
Synthesis and reactions
Upon storage, fluoral polymerises into a waxy, white solid that is soluble in diethyl ether and acetone but not water and chlorocarbons. Heating of this polymer gives monomeric fluoral.[6] Fluoral can be prepared from trifluoroacetic acid with lithium aluminium hydride in diethyl ether:[4] or with concentrated sulfuric acid.[clarification needed]
Cathodic reduction of bromotrifluoromethane in dimethylformamide with aluminium as anode gives high yields of fluoral. In this reaction, DMF acts both as the solvent and the formylation agent.[7] Vapour-phase oxidation of trifluoroethanol also gives fluoral.[5]
Photolysis of fluoral gives fluoroform, hexafluoroethane and carbon monoxide, along with some hydrogen.[8]
See also
References
- ↑ 1.0 1.1 The Radiolysis of Some Organic Halogen Compounds in Aqueous Solutions, R. J. Woods and J. W. T Spinks (1960), Canadian Journal of Chemistry vol.38
- ↑ 2.0 2.1 2.2 Trifluoroacetaldehyde, Peter Lin (2003), DOI:10.1002/047084289X.RN00213
- ↑ 2. Aldehydes, Fluorocarbon and Related Chemistry: Volume 2 (2007) Royal Society of Chemistry.
- ↑ 4.0 4.1 Kitazume, T., Yamazaki, T. (2019). Experimental Methods in Organic Fluorine Chemistry
- ↑ 5.0 5.1 Trifluoroacetaldehyde: A useful industrial bulk material for the synthesis of trifluoromethylated amino compounds, Hideyuki Mimura, Kosuke Kawada, Tetsuya Yamashita, Takeshi Sakamoto, Yasuo Kikugawa. Journal of Fluorine Chemistry Volume 131, Issue 4, April 2010, Pages 477-486
- ↑ Oxidative Nitration of 1,1,1-Trifluoropropane. Trifluoroacetaldehyde, Harold Shechter and Franklin Conrad, in J. Am. Chem. Soc. 1950, 72, 8, 3371–3373
- ↑ Advances in Electron Transfer Chemistry. (1999)
- ↑ The photolysis of trifluoroacetaldehyde R. E. Dodd and J. Watson Smith, J. Chem. Soc., 1957, 1465-1473 https://doi.org/10.1039/JR9570001465
 |
 KSF
KSF