Helicene
Topic: Chemistry
 From HandWiki - Reading time: 7 min
From HandWiki - Reading time: 7 min
In organic chemistry, helicenes are ortho-condensed polycyclic aromatic compounds in which benzene rings or other aromatics are angularly annulated to give helically-shaped chiral molecules.[1] The chemistry of helicenes has attracted continuing attention because of their unique structural, spectral, and optical features.[2][3][4][5][6][7][8]
Structure and properties
The systematic naming for this class of compounds is based on the number of rings: [n]helicene is the structure consisting of n rings. According to IUPAC, only structures where n is at least 5 are considered helicenes.[1] Some specific compounds also have alternate or trivial names. As the number of rings increases, starting at four, the structure becomes non-planar, but instead the planes of consecutive rings tilt to prevent steric collisions. For helicenes with six benzene units, a 360° turn is completed. In the helicene series the dihedral angles between the extremities increases going from [4]helicene (26°) to [6]helicene (58°) and then decreases again for example in [7]helicene (30°).
Helicenes are notable for having chirality despite lacking both asymmetric carbons and chiral centers. Instead, there is axial chirality, which results from the handedness of the helicity itself. The clockwise and counterclockwise helices are non-superposable. By convention a left-handed helix is minus and labeled (M), a right-handed helix is plus and labeled (P). Evidence from CD spectroscopy suggests left-handed helices are levorotatory and right-handed helices are dextrorotatory.
The stability of the two complementary helical enantiomers with respect to interconversion and the mechanism by which they interconvert depend on n.[9]
Synthesis
The first helicene structure was reported by Jakob Meisenheimer in 1903 as the reduction product of 2-nitronaphthalene.[10] [5]helicene was synthesized in 1918 by Weitzenböck & Klingler.[11] The first [6]helicene (also called hexahelicene) was synthesized by M. S. Newman and D. Lednicer in 1955 via a scheme that closed the two central rings by Friedel–Crafts cyclization of carboxylic acid compounds.[12][13] Since then, several methods for synthesizing helicenes with different lengths and substituents are used. The oxidative photocyclization of a stilbene-type precursor is used most often as the key step. The longest helicene prepared by this method is [16]helicene in 2015.[14]
In one study,[15] [5]helicene was synthesized in an olefin metathesis reaction of a divinyl compound (prepared from 1,1′-bi-2-naphthol (BINOL) in several steps), with Grubbs' second generation catalyst:
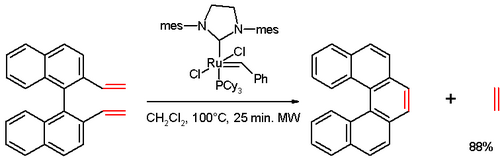
Other approach is also non-photochemical and is based on assembly of biphenylyl-naphthalenes and their platinum-catalyzed double cycloisomerization leading to various [6]helicenes:[16]
-
[4]Helicene
-
[5]Helicene
-
[6]Helicene
-
[6]Helicene, minus chirality
-
[7]Helicene
-
[7]Helicene, minus chirality
-
[8]Helicene
-
[9]Helicene
-
[10]Helicene
-
[11]Helicene
-
[12]Helicene
-
[13]Helicene
-
[14]Helicene
-
[15]Helicene
-
[16]Helicene
-
[18]Helicene
Applications
Helicenes have been studied with respect to nonlinear optics,[17] CPL,[18][19] organocatalysis,[20] conformational analysis,[21] chirality sensing,[22] chemical sensors[23] and hetero-atom substitution.[24][25][26][27]
See also
- Other configurations of consecutively-fused benzene rings:
- Acenes, linear
- Circulenes, closed ring
- Phenacenes, zig-zag
References
- ↑ 1.0 1.1 IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "helicenes". doi:10.1351/goldbook.H02762
- ↑ Martin, Richard Henri (1974). "The Helicenes". Angewandte Chemie International Edition in English 13 (10): 649–660. doi:10.1002/anie.197406491.
- ↑ Shen, Yun; Chen, Chuan-Feng (2012). "Helicenes: Synthesis and Applications". Chemical Reviews 112 (3): 1463–1535. doi:10.1021/cr200087r. PMID 22017405. Bibcode: 2012ChRv..112.1463S.
- ↑ Wang, David Zhigang; Katz, Thomas J.; Golen, James; Rheingold, Arnold L. (2004). "Diels−Alder Additions of Benzynes within Helicene Skeletons". The Journal of Organic Chemistry 69 (22): 7769–7771. doi:10.1021/jo048707h. PMID 15498014.
- ↑ Gingras, Marc (2013). "One hundred years of helicene chemistry. Part 1: Non-stereoselective syntheses of carbohelicenes". Chem. Soc. Rev. 42 (3): 968–1006. doi:10.1039/C2CS35154D. PMID 23151799. Bibcode: 2013CSRev..42..968G.
- ↑ Gingras, Marc; Félix, Guy; Peresutti, Romain (2013). "One hundred years of helicene chemistry. Part 2: Stereoselective syntheses and chiral separations of carbohelicenes". Chem. Soc. Rev. 42 (3): 1007–1050. doi:10.1039/C2CS35111K. PMID 23151610. Bibcode: 2013CSRev..42.1007G.
- ↑ Gingras, Marc (2013). "One hundred years of helicene chemistry. Part 3: Applications and properties of carbohelicenes". Chem. Soc. Rev. 42 (3): 1051–1095. doi:10.1039/C2CS35134J. PMID 23151680. Bibcode: 2013CSRev..42.1051G.
- ↑ Kamikawa, Ken (2014). "Recent Development of Helicene Synthesis". Journal of Synthetic Organic Chemistry, Japan 72: 58–67. doi:10.5059/yukigoseikyokaishi.72.58.
- ↑ Freixas, Victor M.; Rouxel, Jérémy R.; Nam, Yeonsig; Tretiak, Sergei; Govind, Niranjan; Mukamel, Shaul (2023). "X-ray and Optical Circular Dichroism as Local and Global Ultrafast Chiral Probes of [12]Helicene Racemization". J. Am. Chem. Soc. 145 (38): 21012–21019. doi:10.1021/jacs.3c07032. PMID 37704187. Bibcode: 2023JAChS.14521012F.
- ↑ Meisenheimer, Jakob; Witte, Klaus (1903). "Reduction von 2-Nitronaphtalin". Berichte der Deutschen Chemischen Gesellschaft 36 (4): 4153–4164. doi:10.1002/cber.19030360481.
- ↑ Weitzenböck, Richard; Klingler, Albert (1918). "Synthese der isomeren Kohlenwasserstoffe 1, 2?5, 6-Dibenzanthracen und 3, 4?5, 6-Dibenzphenanthren". Monatshefte für Chemie 39 (5): 315–323. doi:10.1007/BF01524529.
- ↑ Newman, Melvin S.; Lutz, Wilson B.; Lednicer, Daniel (1955). "A New Reagent for Resolution by Complex Formation: The Resolution of Phenanthro-[3,4-c]phenanthrene". Journal of the American Chemical Society 77 (12): 3420–3421. doi:10.1021/ja01617a097.
- ↑ Newman, Melvin S.; Lednicer, Daniel (1956). "The Synthesis and Resolution of Hexahelicene". Journal of the American Chemical Society 78 (18): 4765–4770. doi:10.1021/ja01599a060. Bibcode: 1956JAChS..78.4765N.
- ↑ Mori, Kazuyuki; Murase, Takashi; Fujita, Makoto (2015). "One-Step Synthesis of [16]Helicene". Angew. Chem. Int. Ed. 54 (23): 6847–6851. doi:10.1002/anie.201502436. PMID 25907312. Bibcode: 2015ACIE...54.6847M.
- ↑ Collins, Shawn K.; Grandbois, Alain; Vachon, Martin P.; Côté, Julie (2006). "Preparation of Helicenes through Olefin Metathesis". Angewandte Chemie International Edition 45 (18): 2923–2926. doi:10.1002/anie.200504150. PMID 16568482. Bibcode: 2006ACIE...45.2923C.
- ↑ Storch, Jan; Sýkora, Jan; Čermák, Jan; Karban, Jindřich; Císařová, Ivana; Růžička, Aleš (2009). "Synthesis of Hexahelicene and 1-Methoxyhexahelicene via Cycloisomerization of Biphenylyl-Naphthalene Derivatives". The Journal of Organic Chemistry 74 (8): 3090–3093. doi:10.1021/jo900077j. PMID 19284794.
- ↑ Coe, Benjamin J.; Rusanova, Daniela; Joshi, Vishwas D.; Sánchez, Sergio; Vávra, Jan; Khobragade, Dushant; Severa, Lukáš; Císařová, Ivana et al. (2016). "Helquat Dyes: Helicene-like Push–Pull Systems with Large Second-Order Nonlinear Optical Responses". The Journal of Organic Chemistry 81 (5): 1912–1920. doi:10.1021/acs.joc.5b02692. PMID 26844587.
- ↑ Yamamoto, Yuki; Sakai, Hayato; Yuasa, Junpei; Araki, Yasuyuki; Wada, Takehiko; Sakanoue, Tomo; Takenobu, Taishi; Kawai, Tsuyoshi et al. (2016). "Synthetic Control of the Excited-State Dynamics and Circularly Polarized Luminescence of Fluorescent "Push–Pull" Tetrathia[9]helicenes". Chemistry – A European Journal 22 (12): 4263–4273. doi:10.1002/chem.201504048. PMID 26863928. Bibcode: 2016ChEuJ..22.4263Y.
- ↑ Yamamoto, Yuki; Sakai, Hayato; Yuasa, Junpei; Araki, Yasuyuki; Wada, Takehiko; Sakanoue, Tomo; Takenobu, Taishi; Kawai, Tsuyoshi et al. (2016). "Controlled Excited-State Dynamics and Enhanced Fluorescence Property of Tetrasulfone[9]helicene by a Simple Synthetic Process". The Journal of Physical Chemistry C 120 (13): 7421–7427. doi:10.1021/acs.jpcc.6b01123.
- ↑ Dova, Davide; Viglianti, Lucia; Mussini, Patrizia R.; Prager, Stefan; Dreuw, Andreas; Voituriez, Arnaud; Licandro, Emanuela; Cauteruccio, Silvia (2016). "Tetrathia[7]helicene Phosphorus Derivatives: Experimental and Theoretical Investigations of Electronic Properties, and Preliminary Applications as Organocatalysts". Asian Journal of Organic Chemistry 5 (4): 537–549. doi:10.1002/ajoc.201600025.
- ↑ Fujikawa, Takao; Segawa, Yasutomo; Itami, Kenichiro (2016). "Synthesis and Structural Features of Quadruple Helicenes: Highly Distorted π Systems Enabled by Accumulation of Helical Repulsions". Journal of the American Chemical Society 138 (10): 3587–3595. doi:10.1021/jacs.6b01303. PMID 26918641. Bibcode: 2016JAChS.138.3587F.
- ↑ Huang, Qinfei; Jiang, Liangwei; Liang, Wenting; Gui, Jianchang; Xu, Dingguo; Wu, Wanhua; Nakai, Yoshito; Nishijima, Masaki et al. (2016). "Inherently Chiral Azonia[6]helicene-Modified β-Cyclodextrin: Synthesis, Characterization, and Chirality Sensing of Underivatized Amino Acids in Water". The Journal of Organic Chemistry 81 (8): 3430–3434. doi:10.1021/acs.joc.6b00130. PMID 27002931.
- ↑ Tounsi, Moncef; Ben Braiek, Mourad; Baraket, Abdoullatif; Lee, Michael; Zine, Nadia; Zabala, Miguel; Bausells, Joan; Aloui, Faouzi et al. (2016). "Electrochemical Capacitive K+ EMIS Chemical Sensor Based on the Dibromoaza[7]helicene as an Ionophore for Potassium Ions Detection". Electroanalysis 28 (12): 2892–2899. doi:10.1002/elan.201600104.
- ↑ Wang, Ying; Zhang, Hui; Pink, Maren; Olankitwanit, Arnon; Rajca, Suchada; Rajca, Andrzej (2016). "Radical Cation and Neutral Radical of Aza-thia[7]helicene with SOMO–HOMO Energy Level Inversion". Journal of the American Chemical Society 138 (23): 7298–7304. doi:10.1021/jacs.6b01498. PMID 27219299. Bibcode: 2016JAChS.138.7298W.
- ↑ Shyam Sundar, M.; Sahoo, Sibaprasad; Bedekar, Ashutosh V. (2016). "Synthesis and study of the structural properties of oxa[5]helicene derivatives". Tetrahedron: Asymmetry 27 (16): 777–781. doi:10.1016/j.tetasy.2016.06.020.
- ↑ doi: 10.1021/acs.joc.6b01395
- ↑ Virk, Tarunpreet Singh; Ilawe, Niranjan V.; Zhang, Guoxian; Yu, Craig P.; Wong, Bryan M.; Chan, Julian M. W. (2016). "Sultam-Based Hetero[5]helicene: Synthesis, Structure, and Crystallization-Induced Emission Enhancement". ACS Omega 1 (6): 1336–1342. doi:10.1021/acsomega.6b00335. PMID 31457199.
- General references
- Chuan-Feng; Yun Shen (2017). Helicene Chemistry: From Synthesis to Applications. Springer. doi:10.1007/978-3-662-53168-6. ISBN 978-3-662-53168-6.
 |
 KSF
KSF
![[4]Helicene](https://handwiki.org/wiki/images/thumb/e/ee/Tetrahelicene.jpg/120px-Tetrahelicene.jpg)
![[5]Helicene](https://handwiki.org/wiki/images/thumb/7/7c/Pentahelicene.jpg/120px-Pentahelicene.jpg)
![[6]Helicene](https://handwiki.org/wiki/images/thumb/e/e2/Hexahelicene2.jpg/119px-Hexahelicene2.jpg)
![[6]Helicene, minus chirality](https://handwiki.org/wiki/images/thumb/9/95/Hexahelicene.jpg/120px-Hexahelicene.jpg)
![[7]Helicene](https://handwiki.org/wiki/images/thumb/9/9f/Heptahelicene2.jpg/120px-Heptahelicene2.jpg)
![[7]Helicene, minus chirality](https://handwiki.org/wiki/images/thumb/2/29/Heptahelicene.jpg/120px-Heptahelicene.jpg)
![[8]Helicene](https://handwiki.org/wiki/images/thumb/6/64/Octahelicene.jpg/120px-Octahelicene.jpg)
![[9]Helicene](https://handwiki.org/wiki/images/thumb/5/57/Nonahelicene.jpg/120px-Nonahelicene.jpg)
![[10]Helicene](https://handwiki.org/wiki/images/thumb/d/d6/Decahelicene.jpg/90px-Decahelicene.jpg)
![[11]Helicene](https://handwiki.org/wiki/images/thumb/2/2e/Undecahelicene.jpg/120px-Undecahelicene.jpg)
![[12]Helicene](https://handwiki.org/wiki/images/thumb/e/e5/Dodecahelicene.jpg/111px-Dodecahelicene.jpg)
![[13]Helicene](https://handwiki.org/wiki/images/thumb/0/08/Tridecahelicene.jpg/99px-Tridecahelicene.jpg)
![[14]Helicene](https://handwiki.org/wiki/images/thumb/8/86/Tetradecahelicene.jpg/120px-Tetradecahelicene.jpg)
![[15]Helicene](https://handwiki.org/wiki/images/thumb/c/cc/Pentadecahelicene.jpg/120px-Pentadecahelicene.jpg)
![[16]Helicene](https://handwiki.org/wiki/images/thumb/e/e6/Hexadecahelicene.jpg/112px-Hexadecahelicene.jpg)
![[18]Helicene](https://handwiki.org/wiki/images/thumb/0/04/Octadecahelicene.jpg/97px-Octadecahelicene.jpg)
