Iodine oxide
Topic: Chemistry
 From HandWiki - Reading time: 10 min
From HandWiki - Reading time: 10 min
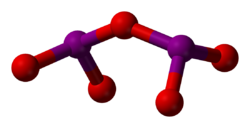
Iodine oxides are chemical compounds of oxygen and iodine. Iodine has only two stable oxides which are isolatable in bulk, iodine tetroxide and iodine pentoxide, but a number of other oxides are formed in trace quantities or have been hypothesized to exist. The chemistry of these compounds is complicated with only a few having been well characterized. Many have been detected in the atmosphere and are believed to be particularly important in the marine boundary layer.[1]
Molecular compounds
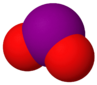
| Molecular formula | I2O | IO[3] | IO2 | I2O4 | I4O9 | I2O5 | I2O6 |
|---|---|---|---|---|---|---|---|
| Name | diiodine oxide | iodine monoxide | iodine dioxide | iodine tetroxide (diiodine tetroxide) | tetraiodine nonoxide | Iodine pentoxide (diiodine pentoxide) | Diiodine hexaoxide |
| Structure | I2O | IO | IO2 | O2IOIO | I(OIO2)3 | O(IO2)2 | [IO2]+[IO4]− |
| Molecular model | 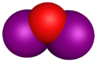 |
 |
 |
 |
|||
| CAS registry | 39319-71-6 | 14696-98-1 | 13494-92-3 | 1024652-24-1 | 66523-94-2 | 12029-98-0 | 65355-99-9 |
| Appearance | Unknown | purple gas | dilute gas; condenses to I2O4[4] | yellow solid | dark yellow solid | white crystalline solid | yellow solid |
| Oxidation state | +1 | +2 | +4 | +3 and +5 | +3 and +5 | +5 | +5 and +7 |
| Melting point | not isolable | not isolable | not isolable | decomp. 100 °C | decomp. 75 °C | decomp. 300–350 °C | decomp 150 °C |
| Specific gravity | 4.2 | 4.8 | 4.53 | ||||
| Solubility in water | decomp. to HIO3 + I2 | decomp. to HIO3 + I2 | 187 g/100 mL |
Diiodine monoxide has largely been the subject of theoretical study,[5] but there is some evidence that it may be prepared in a similar manner to dichlorine monoxide, via a reaction between HgO and I2.[6] The compound appears to be highly unstable but can react with alkenes to give halogenated products.[7]
Radical iodine oxide (IO), iodine dioxide (IO2), collectively referred to as IxOy and iodine tetroxide (I2O4) all possess significant and interconnected atmospheric chemistry. They are formed, in very small quantities, in the marine boundary layer by the photooxidation of diiodomethane, which is produced by macroalga such as seaweed or through the oxidation of molecular iodine, produced by the reaction of gaseous ozone and iodide present at the seasurface.[4][8] Despite the small quantities produced (typically below ppt) they are thought to be powerful ozone depletion agents.[9][10]
Diiodine pentoxide (I2O5) is the anhydride of iodic acid and the only stable anhydride of an iodine oxoacid.
Tetraiodine nonoxide (I4O9) has been prepared by the gas-phase reaction of I2 with O3 but has not been extensively studied.[11]
Iodate anions
Iodine oxides also form negatively charged anions, which (associated with complementary cations) are components of acids or salts. These include the iodates and periodates.
Their conjugate acids are:
| Iodine oxidation state | −1 | +1 | +3 | +5 | +7 |
|---|---|---|---|---|---|
| Name | Hydrogen iodide* | Hypoiodous acid | Iodous acid | Iodic acid | Periodic acid |
| Formula | HI | HIO | HIO2 | HIO3 | HIO4 or H5IO6 |
This page may be too long to read and navigate comfortably. |
Slovene pronunciation: [{{{1}}}]Thai pronunciation: [{{{1}}}]Neukirch, Jürgen; Schmidt, Alexander; Wingberg, Kay (2000), Cohomology of Number Fields, Grundlehren der Mathematischen Wissenschaften, 323, Berlin: Springer-Verlag, ISBN 978-3-540-66671-4Template:ScribuntoPersian pronunciation: [{{{1}}}]{| class="wikitable" width="100%"
! rowspan="3" colspan="2" width="14%" style="border-bottom:2px solid grey;" | Date/Time (UTC) ! Configuration ! Serial number ! Launch site ! Outcome |-
|style="text-align:center;background-color:#e3e9e9;" | Payload |style="text-align:center;background-color:#e3e9e9;" | Separation orbit |style="text-align:center;background-color:#e3e9e9;" | Operator |style="text-align:center;background-color:#e3e9e9;" | Function |- | colspan="4" style="text-align:center;background-color:#e4dfdf;border-bottom:2px solid grey;" | Remarks |-
Parameter 1=time required!
 | This template does not display in the mobile view of Wikipedia; it is desktop only. See Template:Navbox visibility for a brief explanation. |
This is a navigational template created using {{navbox}}. It can be transcluded on pages by placing {{Iodine oxide}} below the standard article appendices.
Initial visibility
This template's initial visibility currently defaults to autocollapse, meaning that if there is another collapsible item on the page (a navbox, sidebar, or table with the collapsible attribute), it is hidden apart from its title bar; if not, it is fully visible.
To change this template's initial visibility, the |state= parameter may be used:
{{Iodine oxide|state=collapsed}}will show the template collapsed, i.e. hidden apart from its title bar.{{Iodine oxide|state=expanded}}will show the template expanded, i.e. fully visible.
Templates using the classes class=navbox ({{navbox}}) or class=nomobile ({{sidebar}}) are not displayed in article space on the mobile web site of English Wikipedia. Mobile page views accounted for 60% to 70% of all page views from 2020 through 2025. Briefly, these templates are not included in articles because 1) they are not well designed for mobile, and 2) they significantly increase page sizes—bad for mobile downloads—in a way that is not useful for the mobile use case. You can review/watch phab:T124168 for further discussion.
TemplateData
A navigational box that can be placed at the bottom of articles.
| Parameter | Description | Type | Status | |
|---|---|---|---|---|
| State | state | The initial visibility of the navbox
| String | suggested |
Template transclusions
| Transclusion maintenance |
|---|
| Check completeness of transclusions |
Hawaiian pronunciation: [{{{1}}}]German pronunciation: [{{{1}}}]Dutch pronunciation: [{{{1}}}]Belarusian pronunciation: [{{{1}}}]Catalan pronunciation: [{{{1}}}]{{{1}}}Mongolian pronunciation: [{{{1}}}]Javanese pronunciation: [{{{1}}}]![]() Not doneLatin pronunciation: [{{{1}}}]
Not doneLatin pronunciation: [{{{1}}}]
| rowspan="1" colspan="2" style="border-top:2px solid #aabbcc;" |
| style="border-top:2px solid #aabbcc;" | | style="border-top:2px solid #aabbcc;" | | style="border-top:2px solid #aabbcc;" | | style="border-top:2px solid #aabbcc;" |
|-
|-
Vietnamese pronunciation: [{{{1}}}]
Piedmontese pronunciation: [{{{1}}}]Slovak pronunciation: [{{{1}}}]text-align: auto;Expression error: Unrecognized punctuation character "{"./Expression error: Unrecognized punctuation character "{". (age Expression error: Unrecognized punctuation character "{".–Expression error: Unrecognized punctuation character "{".)Occitan pronunciation: [{{{1}}}]data-sort-value="" style="background: #ececec; color: #2C2C2C; vertical-align: middle; font-size: smaller; text-align: center; " class="table-na" | VariesHejazi pronunciation: [{{{1}}}]Spanish pronunciation: [{{{1}}}]Portuguese pronunciation: [{{{1}}}]Irish pronunciation: [{{{1}}}]Mayan pronunciation: [{{{1}}}]Nahuatl pronunciation: [{{{1}}}]Kyrgyz pronunciation: [{{{1}}}]Scottish Gaelic pronunciation: [{{{1}}}]Ukrainian pronunciation: [{{{1}}}]Burmese pronunciation: [{{{1}}}]Hindi pronunciation: [{{{1}}}]![]() Done
Done![]() To doRussian pronunciation: [{{{1}}}]Greek pronunciation: [{{{1}}}]
To doRussian pronunciation: [{{{1}}}]Greek pronunciation: [{{{1}}}]![]() Danish pronunciation: [{{{1}}}]Northern Sami pronunciation: [{{{1}}}]Mandarin pronunciation: [{{{ipa}}}]Cantonese pronunciation: [{{{1}}}]Entry from Iodine oxide from TCI Europe, retrieved on {{{Date}}}
Danish pronunciation: [{{{1}}}]Northern Sami pronunciation: [{{{1}}}]Mandarin pronunciation: [{{{ipa}}}]Cantonese pronunciation: [{{{1}}}]Entry from Iodine oxide from TCI Europe, retrieved on {{{Date}}}
 | This template does not display in the mobile view of Wikipedia; it is desktop only. See Template:Navbox visibility for a brief explanation. |
This is a navigational template created using {{navbox}}. It can be transcluded on pages by placing {{Iodine oxide}} below the standard article appendices.
Initial visibility
This template's initial visibility currently defaults to autocollapse, meaning that if there is another collapsible item on the page (a navbox, sidebar, or table with the collapsible attribute), it is hidden apart from its title bar; if not, it is fully visible.
To change this template's initial visibility, the |state= parameter may be used:
{{Iodine oxide|state=collapsed}}will show the template collapsed, i.e. hidden apart from its title bar.{{Iodine oxide|state=expanded}}will show the template expanded, i.e. fully visible.
Templates using the classes class=navbox ({{navbox}}) or class=nomobile ({{sidebar}}) are not displayed in article space on the mobile web site of English Wikipedia. Mobile page views accounted for 60% to 70% of all page views from 2020 through 2025. Briefly, these templates are not included in articles because 1) they are not well designed for mobile, and 2) they significantly increase page sizes—bad for mobile downloads—in a way that is not useful for the mobile use case. You can review/watch phab:T124168 for further discussion.
TemplateData
A navigational box that can be placed at the bottom of articles.
| Parameter | Description | Type | Status | |
|---|---|---|---|---|
| State | state | The initial visibility of the navbox
| String | suggested |
Chambers, Robert; Thomson, Thomas Napier (1857). "[[s:A biographical dictionary of eminent Scotsmen/|]]". A Biographical Dictionary of Eminent Scotsmen. Glasgow: Blackie and Son.{{{1}}}Polish pronunciation: [{{{1}}}]Malagasy pronunciation: [{{{1}}}]Japanese pronunciation: [{{{1}}}]Armenian pronunciation: [{{{1}}}]Czech pronunciation: [{{{1}}}]∶Manx pronunciation: [{{{1}}}]http://www.iucnredlist.org/apps/redlist/details/full/{{{1}}}/0 Alemannic German pronunciation: [{{{1}}}]Tagalog pronunciation: [{{{1}}}]Egyptian Arabic pronunciation: [{{{1}}}]French pronunciation: [{{{1}}}]Error: Invalid time.![]() In progressPalomares, M. L. D. and Pauly, D., eds. (2011). "{{{1}}} {{{2}}}" in SeaLifeBase. April 2011 version.Turkish pronunciation: [{{{1}}}]Finnish pronunciation: [{{{1}}}]Froese, Rainer and Pauly, Daniel, eds. (2006). "{{{1}}} {{{2}}}" in FishBase. April 2006 version.
In progressPalomares, M. L. D. and Pauly, D., eds. (2011). "{{{1}}} {{{2}}}" in SeaLifeBase. April 2011 version.Turkish pronunciation: [{{{1}}}]Finnish pronunciation: [{{{1}}}]Froese, Rainer and Pauly, Daniel, eds. (2006). "{{{1}}} {{{2}}}" in FishBase. April 2006 version.
|- | ... | — | — | — | — | — | —
|-Old Norse pronunciation: [{{{1}}}]Salish pronunciation: [{{{1}}}]Basque pronunciation: [{{{1}}}]{{{1}}}data-sort-value="" style="background: #ececec; color: #2C2C2C; vertical-align: middle; font-size: smaller; text-align: center; " class="table-na" | UnreleasedNorwegian pronunciation: [{{{1}}}] (aged {{{4}}})Hungarian pronunciation: [{{{1}}}]Quechua pronunciation: [{{{1}}}]Arabic pronunciation: [{{{1}}}]Punjabi pronunciation: [{{{1}}}]Afrikaans pronunciation: [{{{1}}}]Romanian pronunciation: [{{{1}}}]Hebrew pronunciation: [{{{1}}}][INVALID OR MISSING PARAMETER IN TEMPLATE Iodine oxide]Uto-Aztecan pronunciation: [{{{1}}}]Tamil pronunciation: [{{{1}}}]Hindustani pronunciation: [{{{1}}}]Swedish pronunciation: [{{{1}}}]Kazakh pronunciation: [{{{1}}}]
Lao pronunciation: [{{{1}}}]Tibetan pronunciation: [{{{1}}}]Khmer pronunciation: [{{{1}}}]data-sort-value="" style="vertical-align:middle; text-align:center" class="table-na" | —{{{1}}}Welsh pronunciation: [{{{1}}}]![]() Partially doneAthabaskan pronunciation: [{{{1}}}]Māori pronunciation: [{{{1}}}]IPA: [{{{1}}}]Bulgarian pronunciation: [{{{1}}}]Korean pronunciation: [{{{1}}}]Icelandic pronunciation: [{{{1}}}]Sanskrit pronunciation: [{{{1}}}]Bengali pronunciation: [{{{1}}}]Indonesian pronunciation: [{{{1}}}]Serbo-Croatian pronunciation: [{{{1}}}]CROSBI {{{1}}}The −1 oxidation state, hydrogen iodide, is not an oxide, but it is included in this table for completeness.
Partially doneAthabaskan pronunciation: [{{{1}}}]Māori pronunciation: [{{{1}}}]IPA: [{{{1}}}]Bulgarian pronunciation: [{{{1}}}]Korean pronunciation: [{{{1}}}]Icelandic pronunciation: [{{{1}}}]Sanskrit pronunciation: [{{{1}}}]Bengali pronunciation: [{{{1}}}]Indonesian pronunciation: [{{{1}}}]Serbo-Croatian pronunciation: [{{{1}}}]CROSBI {{{1}}}The −1 oxidation state, hydrogen iodide, is not an oxide, but it is included in this table for completeness.
The periodates include two variants: metaperiodate IO−4 and orthoperiodate IO5−6.
See also
References
- ↑ Kaltsoyannis, Nikolas; Plane, John M. C. (2008). "Quantum chemical calculations on a selection of iodine-containing species (IO, OIO, INO3, (IO)2, I2O3, I2O4 and I2O5) of importance in the atmosphere". Physical Chemistry Chemical Physics 10 (13): 1723–33. doi:10.1039/B715687C. PMID 18350176. Bibcode: 2008PCCP...10.1723K.
- ↑ Lide, D. R., ed (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ↑ Nikitin, I V (31 August 2008). "Halogen monoxides". Russian Chemical Reviews 77 (8): 739–749. doi:10.1070/RC2008v077n08ABEH003788. Bibcode: 2008RuCRv..77..739N.
- ↑ 4.0 4.1 Hoffmann, Thorsten; O'Dowd, Colin D.; Seinfeld, John H. (15 May 2001). "Iodine oxide homogeneous nucleation: An explanation for coastal new particle production". Geophysical Research Letters 28 (10): 1949–1952. doi:10.1029/2000GL012399. Bibcode: 2001GeoRL..28.1949H. https://authors.library.caltech.edu/50961/1/grl14261.pdf.
- ↑ Novak, Igor (1998). "Theoretical study of I2O". Heteroatom Chemistry 9 (4): 383–385. doi:10.1002/(SICI)1098-1071(1998)9:4<383::AID-HC6>3.0.CO;2-9.
- ↑ Forbes, Craig P.; Goosen, André; Laue, Hugh A. H. (1974). "Hypoiodite reaction: kinetic study of the reaction of 1,1-diphenyl-ethylene with mercury(II) oxide iodine". Journal of the Chemical Society, Perkin Transactions 1: 2350–2353. doi:10.1039/P19740002350.
- ↑ Cambie, Richard C.; Hayward, Rodney C.; Lindsay, Barry G.; Phan, Alice I. T.; Rutledge, Peter S.; Woodgate, Paul D. (1976). "Reactions of iodine oxide with alkenes". Journal of the Chemical Society, Perkin Transactions 1 (18): 1961. doi:10.1039/P19760001961.
- ↑ Carpenter, Lucy J.; MacDonald, Samantha M.; Shaw, Marvin D.; Kumar, Ravi; Saunders, Russell W.; Parthipan, Rajendran; Wilson, Julie; Plane, John M.C. (13 January 2013). "Atmospheric iodine levels influenced by seasurface emissions of inorganic iodine". Nature Geoscience 6 (2): 108–111. doi:10.1038/ngeo1687. Bibcode: 2013NatGe...6..108C. http://eprints.whiterose.ac.uk/76461/7/Nge01687%20-%20pre-publication%20version_with_coversheet.pdf.
- ↑ Saiz-Lopez, A.; Fernandez, R. P.; Ordóñez, C.; Kinnison, D. E.; Gómez Martín, J. C.; Lamarque, J.-F.; Tilmes, S. (10 December 2014). "Iodine chemistry in the troposphere and its effect on ozone". Atmospheric Chemistry and Physics 14 (23): 13119–13143. doi:10.5194/acp-14-13119-2014. Bibcode: 2014ACP....1413119S.
- ↑ Cox, R. A.; Bloss, W. J.; Jones, R. L.; Rowley, D. M. (1 July 1999). "OIO and the atmospheric cycle of iodine". Geophysical Research Letters 26 (13): 1857–1860. doi:10.1029/1999GL900439. Bibcode: 1999GeoRL..26.1857C. http://pure-oai.bham.ac.uk/ws/files/11849889/Cox_OIO_and_the_Atmospheric_Cycle_of_Iodine_GRL_1999.pdf.
- ↑ Sunder, S.; Wren, J. C.; Vikis, A. C. (December 1985). "Raman spectra of I4O9 formed by the reaction of iodine with ozone". Journal of Raman Spectroscopy 16 (6): 424–426. doi:10.1002/jrs.1250160611. Bibcode: 1985JRSp...16..424S.
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 |
IF, IF3, IF5, IF7 |
Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 |
S | ICl, ICl3 |
Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | CrI3 | MnI2 | FeI2 | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 |
AsI3 | Se | IBr | Kr |
| RbI | SrI2 | YI3 | ZrI4 | NbI5 | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 |
SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | HfI4 | TaI5 | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 |
TlI | PbI2 | BiI3 | Po | AtI | Rn | |
| Fr | RaI2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | UI3, UI4 |
Np | Pu | Am | Cm | Bk | Cf | EsI3 | Fm | Md | No | Lr | |||
 |
 KSF
KSF