Lanthanum hydroxide
Topic: Chemistry
 From HandWiki - Reading time: 5 min
From HandWiki - Reading time: 5 min
 __ La3+ __ OH−
| |
| Names | |
|---|---|
| IUPAC name
Lanthanum(III) hydroxide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| La(OH)3 | |
| Molar mass | 189.93 g/mol |
| Ksp= 2.00·10−21 | |
| Structure | |
| hexagonal | |
| P63/m, No. 176 | |
a = 6.547 Å, c = 3.854 Å
| |
| Hazards | |
| Main hazards | Irritant |
| GHS pictograms | 
|
| GHS Signal word | Danger |
| H314 | |
| P260, P264, P280, P301+330+331, P303+361+353, P304+340, P305+351+338, P310, P321, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions
|
Lanthanum(III) chloride |
Other cations
|
Cerium(III) hydroxide Actinium(III) hydroxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Lanthanum hydroxide is La(OH)3, a hydroxide of the rare-earth element lanthanum.
Synthesis
Lanthanum hydroxide can be obtained by adding an alkali such as ammonia to aqueous solutions of lanthanum salts such as lanthanum nitrate. This produces a gel-like precipitate that can then be dried in air.[2]
- La(NO
3)
3 + 3 NH
4OH → La(OH)
3 + 3 NH
4NO
3
Alternatively, it can be produced by hydration reaction (addition of water) to lanthanum oxide.[3]
- La
2O
3 + 3 H
2O → 2 La(OH)
3
Characteristics
Lanthanum hydroxide does not react much with alkaline substances, however is slightly soluble in acidic solution.[2] In temperatures above 330 °C it decomposes into lanthanum oxide hydroxide (LaOOH), which upon further heating decomposes into lanthanum oxide (La
2O
3):[4]
- La(OH)
3 LaOOH
- 2 LaOOH La
2O
3
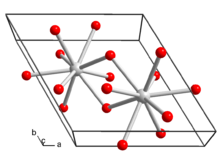
Lanthanum hydroxide crystallizes in the hexagonal crystal system. Each lanthanum ion in the crystal structure is surrounded by nine hydroxide ions in a tricapped trigonal prism.[5]
References
- ↑ "C&L Inventory". https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/123392.
- ↑ 2.0 2.1 E.V. Shkolnikov (2009). "Thermodynamic Characterization of the Amphoterism of Hydroxides and Oxides of Scandium Subgroup Elements in Aqueous Media". Russian Journal of Applied Chemistry 82 (2): 2098–2104. doi:10.1134/S1070427209120040.
- ↑ Ding, Jiawen; Wu, Yanli; Sun, Weili; Li, Yongxiu (2006). "Preparation of La(OH)3 and La2O3 with Rod Morphology by Simple Hydration of La2O3". Journal of Rare Earths 24 (4): 440–442. doi:10.1016/S1002-0721(06)60139-7.
- ↑ Michael E. Brown, Patrick Kent Gallagher (2008). Handbook of Thermal Analysis and Calorimetry. Elsevier. p. 482. ISBN 978-0-44453123-0.
- ↑ Beall, G.W.; Milligan, W.O.; Wolcott, Herbert A. (1977). "Structural trends in the lanthanide trihydroxides" (in en). Journal of Inorganic and Nuclear Chemistry 39 (1): 65–70. doi:10.1016/0022-1902(77)80434-X.
External links
- "Lanthanum - Element information, properties and uses - Periodic Table". rsc.org. http://www.rsc.org/periodic-table/element/57/lanthanum.
- External MSDS 1
- External MSDS 2
- Lanthanum Oxide MSDS
 |
 KSF
KSF
