Loganin
Topic: Chemistry
 From HandWiki - Reading time: 2 min
From HandWiki - Reading time: 2 min
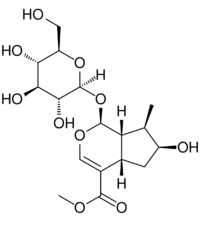
| |

| |
| Names | |
|---|---|
| IUPAC name
Methyl (1S,4aS,6S,7R,7aS)-1-(β-D-glucopyranosyloxy)-6-hydroxy-7-methyl-1,4a,5,6,7,7a-hexahydrocyclopenta[c]pyran-4-carboxylate
| |
| Systematic IUPAC name
Methyl (1S,4aS,6S,7R,7aS)-6-hydroxy-7-methyl-1-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-1,4a,5,6,7,7a-hexahydrocyclopenta[c]pyran-4-carboxylate | |
| Other names
Loganoside
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C17H26O10 | |
| Molar mass | 390.385 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Loganin is one of the best-known of the iridoid glycosides. It is named for the Loganiaceae, having first been isolated from the seeds of a member of that plant family, namely those of Strychnos nux-vomica. It also occurs in Alstonia boonei (Apocynaceae),[1] a medicinal tree of West Africa and in the medicinal/entheogenic shrub Desfontainia spinosa (Columelliaceae) native to Central America and South America.
Biosynthesis
Loganin is formed from loganic acid by the enzyme loganic acid O-methyltransferase (LAMT). Loganin then becomes a substrate for the enzyme secologanin synthase (SLS) to form secologanin, a secoiridoid monoterpene found as part of ipecac and terpene indole alkaloids.
References
- ↑ Adotey, J. P.; Adukpo, G. E.; Opoku-Boahen, Y.; Armah, F. A. (2012). "A Review of the Ethnobotany and Pharmacological Importance of Alstonia boonei De Wild (Apocynaceae)". ISRN Pharmacology 2012: 587160. doi:10.5402/2012/587160. PMID 22900200.
 |
Licensed under CC BY-SA 3.0 | Source: https://handwiki.org/wiki/Chemistry:Loganin7 views | Status: cached on October 31 2024 10:45:09↧ Download this article as ZWI file
 KSF
KSF