Saponin
Topic: Chemistry
 From HandWiki - Reading time: 10 min
From HandWiki - Reading time: 10 min
Saponins (Latin "sapon", soap + "-in", one of), also selectively referred to as triterpene glycosides, are bitter-tasting usually toxic plant-derived organic chemicals that have a foamy quality when agitated in water. They are widely distributed but found particularly in soapwort (genus Saponaria), a flowering plant, the soapbark tree (Quillaja saponaria) and soybeans (Glycine max L.). They are used in soaps, medicines, fire extinguishers, speciously as dietary supplements, for synthesis of steroids, and in carbonated beverages (for example, being responsible for maintaining the head on root beer). Saponins are both water and fat soluble, which gives them their useful soap properties. Some examples of these chemicals are glycyrrhizin (licorice flavoring) and quillaia (alt. quillaja), a bark extract used in beverages.[1][2]
Classification based on chemical structure
Structurally, they are glycosides, which are sugars bonded to one or more organic molecules. In a glycoside molecule, the sugar is the glycone part, while one or more non-sugar organic molecules form the aglycone part.
Steroid glycosides
Their aglycone is a steroid.[3]
Triterpene glycosides
Their aglycone is a triterpene.[3]
Uses
The saponins are a subclass of terpenoids, the largest class of plant extracts. The amphipathic nature of saponins gives them activity as surfactants with potential ability to interact with cell membrane components, such as cholesterol and phospholipids, possibly making saponins useful for development of cosmetics and drugs.[4] Saponins have also been used as adjuvants in development of vaccines,[5] such as Quil A, an extract from the bark of Quillaja saponaria.[4][6] This makes them of interest for possible use in subunit vaccines and vaccines directed against intracellular pathogens.[5] In their use as adjuvants for manufacturing vaccines, toxicity associated with sterol complexation remains a concern.[7]
Quillaja is toxic when consumed in large amounts, involving possible liver damage, gastric pain, diarrhea, or other adverse effects.[6] The NOAEL of saponins is around 300 mg/kg in rodents, so a dose of 3 mg/kg should be safe with a safety factor (see Therapeutic index) of 100.[8]
Saponins are used for their effects on ammonia emissions in animal feeding.[9] In the United States, researchers are exploring the use of saponins derived from plants to control invasive worm species, including the jumping worm.[10][11]
Decoction
The principal historical use of these plants was boiling down to make soap. Saponaria officinalis is most suited for this procedure, but other related species also work. The greatest concentration of saponin occurs during flowering, with the most saponin found in the woody stems and roots, but the leaves also contain some.
Biological sources
Saponins have historically been plant-derived, but they have also been isolated from marine organisms such as sea cucumber.[1][12] They derive their name from the soapwort plant (genus Saponaria, family Caryophyllaceae), the root of which was used historically as a soap.[1][13][2] Saponins are also found in the botanical family Sapindaceae, including its defining genus Sapindus (soapberry or soapnut) and the horse chestnut, and in the closely related families Aceraceae (maples) and Hippocastanaceae. It is also found heavily in Gynostemma pentaphyllum (Cucurbitaceae) in a form called gypenosides, and ginseng or red ginseng (Panax, Araliaceae) in a form called ginsenosides. Saponins are also found in the unripe fruit of Manilkara zapota (also known as sapodillas), resulting in highly astringent properties. Nerium oleander (Apocynaceae), also known as White Oleander, is a source of the potent cardiac toxin oleandrin. Within these families, this class of chemical compounds is found in various parts of the plant: leaves, stems, roots, bulbs, blossom and fruit.[14] Commercial formulations of plant-derived saponins, e.g., from the soap bark tree, Quillaja saponaria, and those from other sources are available via controlled manufacturing processes, which make them of use as chemical and biomedical reagents.[15] Soyasaponins are a group of structurally complex oleanane-type triterpenoid saponins that include soyasapogenol (aglycone) and oligosaccharide moieties biosynthesized on soybean tissues. Soyasaponins were previously associated to plant-microbe interactions[16] from root exudates and abiotic stresses, as nutritional deficiency.[17]
Role in plant ecology and impact on animal foraging
In plants, saponins may serve as anti-feedants,[2][18] and to protect the plant against microbes and fungi.[citation needed] Some plant saponins (e.g., from oat and spinach) may enhance nutrient absorption and aid in animal digestion. However, saponins are often bitter to taste, and so can reduce plant palatability (e.g., in livestock feeds), or even imbue them with life-threatening animal toxicity.[18] Some saponins are toxic to cold-blooded organisms and insects at particular concentrations.[18] Further research is needed to define the roles of these natural products in their host organisms, which have been described as "poorly understood" to date.[18]
Ethnobotany
Most saponins, which readily dissolve in water, are poisonous to fish.[19] Therefore, in ethnobotany, they are known for their use by indigenous people in obtaining aquatic food sources. Since prehistoric times, cultures throughout the world have used fish-killing plants, typically containing saponins, for fishing.[20][21][22]
Although prohibited by law, fish-poison plants are still widely used by indigenous tribes in Guyana.[23]
On the Indian subcontinent, the Gondi people use poison-plant extracts in fishing.[24]
Many of California's Native American tribes traditionally used soaproot, (genus Chlorogalum) and/or the root of various yucca species, which contain saponin, as a fish poison. They would pulverize the roots, mix with water to generate a foam, then put the suds into a stream. This would kill or incapacitate the fish, which could be gathered easily from the surface of the water. Among the tribes using this technique were the Lassik, the Luiseño, and the Mattole.[25]
Chemical structure
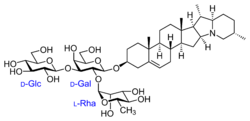
The vast heterogeneity of structures underlying this class of compounds makes generalizations difficult; they're a subclass of terpenoids, oxygenated derivatives of terpene hydrocarbons. Terpenes in turn are formally made up of five-carbon isoprene units. (The alternate steroid base is a terpene missing a few carbon atoms.) Derivatives are formed by substituting other groups for some of the hydrogen atoms of the base structure. In the case of most saponins, one of these substituents is a sugar, so the compound is a glycoside of the base molecule.[1]
More specifically, the lipophilic base structure of a saponin can be a triterpene, a steroid (such as spirostanol or furostanol) or a steroidal alkaloid (in which nitrogen atoms replace one or more carbon atoms). Alternatively, the base structure may be an acyclic carbon chain rather than the ring structure typical of steroids. One or two (rarely three) hydrophilic monosaccharide (simple sugar) units bind to the base structure via their hydroxyl (OH) groups. In some cases other substituents are present, such as carbon chains bearing hydroxyl or carboxyl groups. Such chain structures may be 1-11 carbon atoms long, but are usually 2–5 carbons long; the carbon chains themselves may be branched or unbranched.[1]
The most commonly encountered sugars are monosaccharides like glucose and galactose, though a wide variety of sugars occurs naturally. Other kinds of molecules such as organic acids may also attach to the base, by forming esters via their carboxyl (COOH) groups. Of particular note among these are sugar acids such as glucuronic acid and galacturonic acid, which are oxidized forms of glucose and galactose.[1]
See also
- Cardenolide
- Cardiac glycoside
- Phytochemical
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Hostettmann, K.; A. Marston (1995). Saponins. Cambridge: Cambridge University Press. p. 3ff. ISBN 978-0-521-32970-5. OCLC 29670810.
- ↑ 2.0 2.1 2.2 "Saponins". Cornell University. 14 August 2008. https://www.ansci.cornell.edu/plants/toxicagents/saponin.html.
- ↑ 3.0 3.1 Rao, A. V.; Gurfinkel, D. M. (2000). "The bioactivity of saponins: triterpenoid and steroidal glycosides". Drug Metabolism and Drug Interactions 17 (1–4): 211–235. doi:10.1515/dmdi.2000.17.1-4.211. ISSN 0792-5077. PMID 11201296. https://pubmed.ncbi.nlm.nih.gov/11201296/#:~:text=Triterpenoid%20and%20steroidal%20glycosides,%20referred,in%20natural%20or%20synthetic%20form..
- ↑ 4.0 4.1 Lorent, Joseph H.; Quetin-Leclercq, Joëlle; Mingeot-Leclercq, Marie-Paule (2014-11-28). "The amphiphilic nature of saponins and their effects on artificial and biological membranes and potential consequences for red blood and cancer cells". Organic and Biomolecular Chemistry (Royal Society of Chemistry) 12 (44): 8803–8822. doi:10.1039/c4ob01652a. ISSN 1477-0520. PMID 25295776.
- ↑ 5.0 5.1 Sun, Hong-Xiang; Xie, Yong; Ye, Yi-Ping (2009). "Advances in saponin-based adjuvants". Vaccine 27 (12): 1787–1796. doi:10.1016/j.vaccine.2009.01.091. ISSN 0264-410X. PMID 19208455.
- ↑ 6.0 6.1 "Quillaja". Drugs.com. 2018. https://www.drugs.com/npp/quillaja.html.
- ↑ Skene, Caroline D.; Philip Sutton (1 September 2006). "Saponin-adjuvanted particulate vaccines for clinical use". Methods 40 (1): 53–9. doi:10.1016/j.ymeth.2006.05.019. PMID 16997713.
- ↑ Younes, Maged; Aquilina, Gabriele; Castle, Laurence; Engel, Karl‐Heinz; Fowler, Paul; Frutos Fernandez, Maria Jose; Fürst, Peter; Gürtler, Rainer et al. (2019). "Re‐evaluation of Quillaia extract (E 999) as a food additive and safety of the proposed extension of use". EFSA Journal 17 (3): e05622. doi:10.2903/j.efsa.2019.5622. PMID 32626248.
- ↑ Zentner, Eduard (July 2011). "Effects of phytogenic feed additives containing quillaja saponaria on ammonia in fattening pigs". https://www.delacon.com/phytogenics/articles/Effects-of-phytogenic-feed-additives-containing-quillaja-saponaria-on-ammonia-in-fattening-pigs.pdf.
- ↑ Roach, Margaret (2020-07-22). "As Summer Takes Hold, So Do the Jumping Worms" (in en-US). The New York Times. ISSN 0362-4331. https://www.nytimes.com/2020/07/22/realestate/invasive-jumping-worm-garden-summer.html.
- ↑ "Invasive 'Jumping' Worms Are Now Tearing Through Midwestern Forests" (in en). 2020-01-02. https://www.audubon.org/news/invasive-jumping-worms-are-now-tearing-through-midwestern-forests.
- ↑ Riguera, Ricardo (August 1997). "Isolating bioactive compounds from marine organisms". Journal of Marine Biotechnology 5 (4): 187–193. https://www.springerlink.com/content/m9cclbrm1y0e5ge5/.[|permanent dead link|dead link}}]
- ↑ Liener, Irvin E (1980). "Toxic constituents of plant foodstuffs". The Proceedings of the Nutrition Society (New York City: Academic Press) 29 (1): 56–7. doi:10.1079/pns19700010. ISBN 978-0-12-449960-7. OCLC 5447168. PMID 5529217.[verification needed]
- ↑ "Species Information". Dr. Duke's Phytochemical and Ethnobotanical Databases. https://sun.ars-grin.gov:8080/npgspub/xsql/duke/plantdisp.xsql?taxon=691.
- ↑ "Saponin from quillaja bark". Sigma-Aldrich. http://www.sigmaaldrich.com/US/en/product/sigma/s4521.
- ↑ Tsuno, Yuhei; Fujimatsu, Teruhisa; Endo, Keiji; Sugiyama, Akifumi; Yazaki, Kazufumi (2018-02-01). "Soyasaponins: A New Class of Root Exudates in Soybean (Glycine max)". Plant & Cell Physiology 59 (2): 366–375. doi:10.1093/pcp/pcx192. ISSN 1471-9053. PMID 29216402. https://pubmed.ncbi.nlm.nih.gov/29216402.
- ↑ Cotrim, Gustavo dos Santos; Silva, Deivid Metzker da; Graça, José Perez da; Oliveira Junior, Adilson de; Castro, Cesar de; Zocolo, Guilherme Julião; Lannes, Lucíola Santos; Hoffmann-Campo, Clara Beatriz (2023). "Glycine max (L.) Merr. (Soybean) metabolome responses to potassium availability". Phytochemistry 205: 113472. doi:10.1016/j.phytochem.2022.113472. ISSN 0031-9422. PMID 36270412. https://www.sciencedirect.com/science/article/pii/S0031942222003880.
- ↑ 18.0 18.1 18.2 18.3 Foerster, Hartmut (22 May 2006). "MetaCyc Pathway: saponin biosynthesis I". https://BioCyc.org/META/NEW-IMAGE?type=PATHWAY&object=PWY-5203&detail-level=3.
- ↑ Howes, F. N. (1930), "Fish-poison plants", Bulletin of Miscellaneous Information (Royal Gardens, Kew) 1930 (4): 129–153, doi:10.2307/4107559
- ↑ Jonathan G. Cannon; Robert A. Burton; Steven G. Wood; Noel L. Owen (2004), "Naturally Occurring Fish Poisons from Plants", J. Chem. Educ. 81 (10): 1457, doi:10.1021/ed081p1457, Bibcode: 2004JChEd..81.1457C
- ↑ C. E. Bradley (1956), "Arrow and fish poison of the American southwest", Division of Biology, California Institute of Technology 10 (4): pp. 362–366, doi:10.1007/BF02859766
- ↑ Webb, L. J.; Tracey, J. G.; Haydock, K.P. (1959), An Australian phytochemical survey. III. Saponins in eastern Australian flowering plants, CSIRO, pp. 26, doi:10.25919/5xj5-7648, https://doi.org/10.25919/5xj5-7648
- ↑ Tinde Van Andel (2000), "The diverse uses of fish-poison plants in Northwest Guyana", Economic Botany 54 (4): 500–512, doi:10.1007/BF02866548
- ↑ Murthy, E N; Pattanaik, Chiranjibi; Reddy, C; Sudhakar, Raju V S (March 2010), "Piscicidal plants used by Gond tribe of Kawal wildlife sanctuary, Andhra Pradesh, India", Indian Journal of Natural Products and Resources 1 (1): 97–101, https://nopr.niscair.res.in/handle/123456789/7696, retrieved 22 September 2010
- ↑ Campbell, Paul (1999). Survival skills of native California. Gibbs Smith. pp. 433. ISBN 978-0-87905-921-7. https://books.google.com/books?id=qSRLW5ziVFAC&q=soaproot+fish+poison&pg=PA433. Retrieved 20 November 2020.
 |
 KSF
KSF