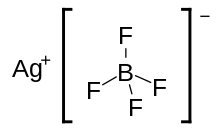
Silver tetrafluoroborate
Topic: Chemistry
 From HandWiki - Reading time: 4 min
From HandWiki - Reading time: 4 min
| |
| Names | |
|---|---|
| IUPAC name
Silver tetrafluoridoborate(1–)
| |
| Other names
Borate(1-), tetrafluoro-, silver(1+)
Argentous tetrafluoroborate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| |
| |
| Properties | |
| AgBF4 | |
| Molar mass | 194.673 g/mol |
| Appearance | Off-white powder |
| Odor | almost odorless |
| Density | 4.16 g/cm3 |
| Melting point | 71.5 °C (160.7 °F; 344.6 K) (monohydrate) |
| soluble | |
| Hazards | |
| Safety data sheet | External MSDS |
| GHS pictograms | 
|
| GHS Signal word | Danger |
| H314 | |
| P260, P264, P280, P301+330+331, P303+361+353, P304+340, P305+351+338, P310, P321, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Silver tetrafluoroborate is an inorganic compound with the molecular formula AgBF4. It is a white solid, although commercial samples often are gray, that dissolves in polar organic solvents as well as water.[2]
Structure
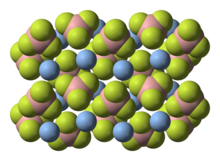
According to X-ray crystallography, the solid compound consists of Ag+ centers bound to four fluoride sites of the BF4−.[3]
Preparation
Silver tetrafluoroborate can be prepared by several methods. A simple route entails dissolving silver carbonate in aqueous tetrafluoroboric acid.[4] It can also be produced by treating silver(I) fluoride with boron trifluoride in nitromethane solution. The reaction between boron trifluoride and a benzene suspension of silver oxide is yet another route, one that exploits the solubility of the compound in benzene. This method however affords silver fulminate, a sensitive explosive.[5]
Laboratory uses
In the inorganic and organometallic chemistry laboratory, silver tetrafluoroborate, sometimes referred to "silver BF-4", is a used as a reagent to remove halide ligands and to oxidize electron-rich complexes. In dichloromethane, silver tetrafluoroborate is a moderately strong oxidant.[6] Similar to silver hexafluorophosphate, it is commonly used to replace halide anions or ligands with the weakly coordinating tetrafluoroborate anions. The abstraction of the halide is driven by the precipitation of the corresponding silver halide.
References
- ↑ "Silver tetrafluoroborate" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/159722#section=Safety-and-Hazards.
- ↑ Wistrand, Lars-G.; Kerekes, Angela D.; Sannigrahi, Mousumi (2005). "Silver Tetrafluoroborate". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rs028.pub2. ISBN 0-471-93623-5.
- ↑ Evgeny Goreshnik, Zoran Mazej, "X-ray single crystal structure and vibrational spectra of AgBF4" Solid State Sciences 2005, Volume 7, pp. 1225–1229. doi:10.1016/j.solidstatesciences.2005.06.007
- ↑ Chalker, Justin M.; Thompson, Amber L.; Davis, Benjamin (2010). "Safe and Scalable Preparation of Barluenga's Reagent". Organic Syntheses 87: 288. doi:10.15227/orgsyn.087.0288.
- ↑ Lemal, D. M; Fry, A. J. (1961-01-01). "The preparation of silver fluoroborate: a warning". Tetrahedron Letters 2 (21): 775–776. doi:10.1016/S0040-4039(01)99266-4. ISSN 0040-4039. https://www.sciencedirect.com/science/article/pii/S0040403901992664.
- ↑ N. G. Connelly, W. E. Geiger (1996). "Chemical Redox Agents for Organometallic Chemistry". Chemical Reviews 96 (2): 877–910. doi:10.1021/cr940053x. PMID 11848774.
 |
 KSF
KSF
