Substituted benzofuran
Topic: Chemistry
 From HandWiki - Reading time: 21 min
From HandWiki - Reading time: 21 min

The substituted benzofurans are a class of chemical compounds based on the heterocyclic and polycyclic compound benzofuran. Many medicines use the benzofuran core as a scaffold,[1][2][3] but most commonly the term is used to refer to the simpler compounds in this class which include numerous psychoactive drugs, including stimulants, psychedelics and empathogens. In general, these compounds have a benzofuran core to which a 2-aminoethyl group is attached (at any position), and combined with a range of other substituents.[4][5][6][7] Some psychoactive derivatives from this family have been sold under the name Benzofury.[8]
Use and effects
| Compound | Chemical name | Dose | Duration | |
|---|---|---|---|---|
| 2-APB | 2-(2-Aminopropyl)benzofuran | Unknown | Unknown | |
| 3-APB | 3-(2-Aminopropyl)benzofuran | Unknown | Unknown | |
| 4-APB | 4-(2-Aminopropyl)benzofuran | Unknown | Unknown | |
| 5-APB | 5-(2-Aminopropyl)benzofuran | 60–80 mg (20–100 mg+) | 3–8 hours | |
| 5-MAPB | 5-(2-(Methylamino)propyl)benzofuran | 30–70 mg (10–120 mg+) | 4–8 hours | |
| 5-EAPB | 5-(2-(Ethylamino)propyl)benzofuran | 80 mg (50–150 mg+) | Unknown | |
| 5-MBPB | 6-(2-(Methylamino)butyl)benzofuran | Unknown | Unknown | |
| BK-5-MAPB | 5-[2-(Methylamino)propanoyl]benzofuran | Unknown | Unknown | |
| 5-APDB | 5-(2-Aminopropyl)-2,3-dihydrobenzofuran | 100 mg (50–200 mg+) | Unknown | |
| 5-MAPDB | 5-(2-(Methylamino)propyl)-2,3-dihydrobenzofuran | 100 mg (50–150 mg+) | Unknown | |
| 6-APB | 6-(2-Aminopropyl)benzofuran | 80–100 mg (15–125 mg+) | 6–9 hours | |
| 6-MAPB | 6-(2-(Methylamino)propyl)benzofuran | 50–100 mg (20–130 mg+) | 6–8 hours | |
| 6-EAPB | 6-(2-(Ethylamino)propyl)benzofuran | Unknown | Unknown | |
| 6-MBPB | 6-(2-(Methylamino)butyl)benzofuran | Unknown | Unknown | |
| BK-6-MAPB | 6-[2-(Methylamino)propanoyl]benzofuran | Unknown | Unknown | |
| 6-APDB | 6-(2-Aminopropyl)-2,3-dihydrobenzofuran | 70 mg (20–130 mg+) | 6–8 hours | |
| 6-MAPDB | 6-(2-(Methylamino)propyl)-2,3-dihydrobenzofuran | Unknown | Unknown | |
| 7-APB | 7-(2-Aminopropyl)benzofuran | Unknown | Unknown | |
| Refs: [9][10][11][8] | ||||
Benzofurans like 5-APB and 6-APB are said to have relatively minor psychedelic effects.[12][13]
Interactions
Pharmacology
Pharmacodynamics
| Compound | Monoamine release (EC50, nM) | Ref | ||
|---|---|---|---|---|
| 5-HT | NE | DA | ||
| 5-APB | 19 | 21 | 31 | [14] |
| 6-APB | 36 | 14 | 10 | [14] |
| 5-MAPB | 64–90 | 24 | 41–459 | [14][15] |
| (S)-5-MAPB | 67 | ND | 258 | [15] |
| (R)-5-MAPB | 184 | ND | 1,951 | [15] |
| 6-MAPB | 33 | 14 | 20 | [14] |
| 5-MABB (5-MBPB) | ND | ND | ND | [16][17] |
| (S)-5-MABB | 31 | 158 | 210 | [16][17] |
| (R)-5-MABB | 49 | 850 | IA | [16][17] |
| 6-MABB (6-MBPB) | ND | ND | ND | [16][17] |
| (R)-6-MABB | 172 | 227 | IA | [16][17] |
| (S)-6-MABB | 54 | 77 | 41 | [16][17] |
| BK-5-MAPB | ND | ND | ND | ND |
| BK-6-MAPB | ND | ND | ND | ND |
| MDA | 160–162 | 47–108 | 106–190 | [18][19][14] |
| MDMA | 50–85 | 54–110 | 51–278 | [20][21][22][18][14] |
| MBDB | 540 | 3,300 | >100,000 | [23] |
| Methylone | 234–708 | 140–270 | 117–220 | [21][24][25][26][27] |
| Notes: The smaller the value, the more strongly the compound produces the effect. The assays were done in rat brain synaptosomes and human potencies may be different. See also Monoamine releasing agent § Activity profiles for a larger table with more compounds. | ||||
Benzofurans like 5-APB and 6-APB act as serotonin–norepinephrine–dopamine releasing agents and as serotonin 5-HT2 receptor agonists.[9][11][28] In addition, some benzofurans, including 5-MAPB, 6-MAPB, BK-5-MAPB, and BK-6-MAPB, have unexpectedly been found to be potent serotonin 5-HT1B receptor agonists.[29] Along with serotonin release and other actions, this property may be involved may be involved in their entactogenic effects.[29] Conversely, MDMA is much less potent as an agonist of the serotonin 5-HT1B receptor.[29]
Chemistry
-
O-DMT (1-oxa-DMT, DMBF)
-
MiPBF (1-oxa-MiPT)
-
Dimemebfe (5-MeO-BFE; 1-oxa-5-MeO-DMT)
-
5-MeO-DiBF (1-oxa-5-MeO-DiPT)
-
3-APB (1-oxa-AMT)
-
Mebfap (5-MeO-3-APB; 1-oxa-5-MeO-AMT)
History
The 2,3-dihydrobenzofurans 5-APDB and 6-APDB were described by David E. Nichols and colleagues at Purdue University as MDMA analogues in 1993.[14][8][13][28][30][31] Subsequently, the benzofurans 5-APB and 6-APB emerged as novel designer drugs in 2010.[13][8][28] Prior to this, they had been patented by Eli Lilly and Company as serotonin 5-HT2C receptor agonists for potential treatment of eating disorders and seizures in 2000 and 2006.[14][8][13] 5-APB and 6-APB are often confused with 5-APDB and 6-APDB.[8] The pharmacology of various benzofurans and 2,3-dihydrobenzofurans was further clarified in the mid-2010s and thereafter.[32][33][28][14]
Society and culture
Legal status
Substituted benzofurans saw widespread use as recreational drugs by being sold as research chemicals making them exempt from drug legislation. Many of the more common compounds were banned in the UK in June 2013 as temporary class drugs, while others have been made permanently illegal in various jurisdictions.[34][35][36]
List of substituted benzofurans
The derivatives may be produced by substitutions at six locations of the benzofuran molecule, as well as saturation of the 2,3- double bond.
The following table displays notable derivatives that have been reported:[37][38][39][40][41][42][43][44][45][46][47]
| Structure | Compound | CAS # | R2 | R3 | R4 | R5 | R6 | R7 | Other |
|---|---|---|---|---|---|---|---|---|---|
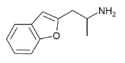 |
2-APB | 30455-73-3 | 2-aminopropyl | H | H | H | H | H | - |
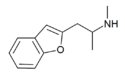 |
2-MAPB | 806596-15-6 | 2-(methylamino)propyl | H | H | H | H | H | - |
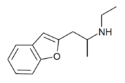 |
2-EAPB | 2-(ethylamino)propyl | H | H | H | H | H | - | |
| 2-MABB[48][49] | 2-(methylamino)butyl | H | H | H | H | H | - | ||
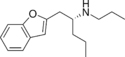 |
BPAP | 260550-89-8 | 2-(propylamino)pentyl | H | H | H | H | H | - |
| File:3-F-BPAP.svg | 3-F-BPAP | 501901-68-4 | 2-(3,3,3-trifluoropropylamino)pentyl | H | H | H | H | H | - |
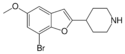 |
Brofaromine | 63638-91-5 | 4-piperidinyl | H | H | methoxy | H | bromo | - |
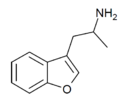 |
3-APB | 105909-13-5 | H | 2-aminopropyl | H | H | H | H | - |
| File:O-DMT chemical structure.svg | O-DMT | H | 2-(dimethyl-amino)ethyl | H | H | H | H | - | |
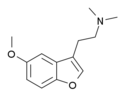 |
Dimemebfe | 140853-58-3 | H | 2-(dimethyl-amino)ethyl | H | methoxy | H | H | - |
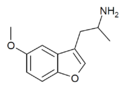 |
Mebfap | 140853-59-4 | H | 2-aminopropyl | H | methoxy | H | H | - |
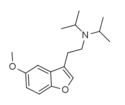 |
5-MeO-DiBF | H | 2-(diisopropyl-amino)ethyl | H | methoxy | H | H | - | |
| Error creating thumbnail: | MiPBF | H | 2-(methylisopropyl-amino)ethyl | H | H | H | H | - | |
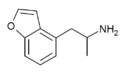 |
4-APB | 286834-82-0 | H | H | 2-aminopropyl | H | H | H | - |
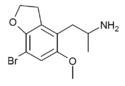 |
DOB-5-HEMIFLY (5-MeO-7-Br-4-APDB) |
H | H | 2-aminopropyl | methoxy | H | bromo | 2,3-dihydro | |
| File:5-APB chemical structure.svg | 5-APB | 286834-81-9 | H | H | H | 2-aminopropyl | H | H | - |
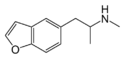 |
5-MAPB | 1354631-77-8 | H | H | H | 2-(methylamino)propyl | H | H | - |
| 5-EAPB | 1445566-01-7 | H | H | H | 2-(ethylamino)propyl | H | H | - | |
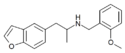 |
5-APB-NBOMe | H | H | H | 2-[(2-methoxybenzyl)-amino]propyl | H | H | - | |
| 6-APB | 286834-85-3 | H | H | H | H | 2-aminopropyl | H | - | |
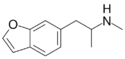 |
6-MAPB | 1354631-79-0 | H | H | H | H | 2-(methylamino)propyl | H | - |
| 6-EAPB | 1632539-47-9 | H | H | H | H | 2-(ethylamino)propyl | H | - | |
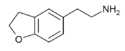 |
5-AEDB | H | H | H | 2-aminoethyl | H | H | 2,3-dihydro | |
| 5-APDB | 152624-03-8 | H | H | H | 2-aminopropyl | H | H | 2,3-dihydro | |
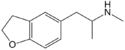 |
5-MAPDB | 1354631-78-9 | H | H | H | 2-(methylamino)propyl | H | H | 2,3-dihydro |
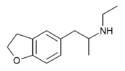 |
5-EAPDB | H | H | H | 2-(ethylamino)propyl | H | H | 2,3-dihydro | |
| 6-APDB | 1354631-78-9 | H | H | H | H | 2-aminopropyl | H | 2,3-dihydro | |
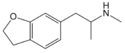 |
6-MAPDB | 1354631-81-4 | H | H | H | H | 2-(methylamino)propyl | H | 2,3-dihydro |
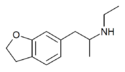 |
6-EAPDB | H | H | H | H | 2-(ethylamino)propyl | H | 2,3-dihydro | |
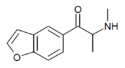 |
bk-5-MAPB | H | H | H | 1-oxo-2-(methylamino)propyl | H | H | - | |
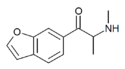 |
bk-6-MAPB | H | H | H | H | 1-oxo-2-(methylamino)propyl | H | - | |
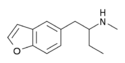 |
5-MBPB | H | H | H | 2-(methylamino)butyl | H | H | - | |
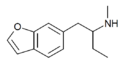 |
6-MBPB | H | H | H | H | 2-(methylamino)butyl | H | - | |
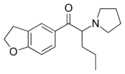 |
5-DBFPV | 2117405-32-8 | H | H | H | 1-oxo-2-(pyrrolidin-1-yl)pentyl | H | H | 2,3-dihydro |
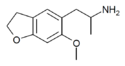 |
6-MeO-5-APDB | H | H | H | 2-aminopropyl | methoxy | H | 2,3-dihydro | |
| File:Semi-fly.svg | F (F-1) | 99355-77-8 | H | H | H | methoxy | 2-aminopropyl | H | 2,3-dihydro |
| File:F-2.svg | F-2 | 99355-74-5 | methyl | H | H | methoxy | 2-aminopropyl | H | 2,3-dihydro |
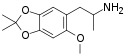 |
F-22 | 952016-51-2 | dimethyl | H | H | methoxy | 2-aminopropyl | H | 2,3-dihydro |
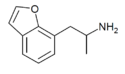 |
7-APB | 286834-86-4 | H | H | H | H | H | 2-aminopropyl | - |
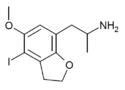 |
DOI-2-HEMIFLY (4-I-5-MeO-7-APDB) |
H | H | iodo | methoxy | H | 2-aminopropyl | 2,3-dihydro | |
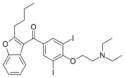
|
Amiodarone | 1951-25-3 | propyl | 3,5-diiodo-4-(2-diethylamino-ethoxy)benzoyl | H | H | H | H | - |
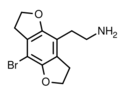 |
2C-B-FLY | 733720-95-1 | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,6-f] | - | bromo | 2,3-dihydro |
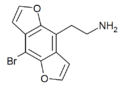 |
2C-B-DRAGONFLY | 260809-98-1 | H | H | 2-aminoethyl | furo[5,6-f] | - | bromo | - |
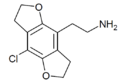 |
2C-C-FLY | 1354633-83-2 | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,6-f] | - | chloro | 2,3-dihydro |
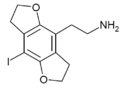 |
2C-I-FLY | 1354633-88-7 | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,6-f] | - | iodo | 2,3-dihydro |
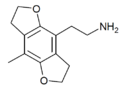 |
2C-D-FLY | 1354634-07-3 | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,6-f] | - | methyl | 2,3-dihydro |
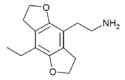 |
2C-E-FLY | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,6-f] | - | ethyl | 2,3-dihydro | |
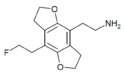 |
2C-EF-FLY | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,6-f] | - | 2-fluoroethyl | 2,3-dihydro | |
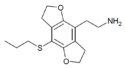 |
2C-T-7-FLY | 1354633-05-8 | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,6-f] | - | n-propylthio | 2,3-dihydro |
| File:2C-MeTriox structure.png | 2C-MeTriox | H | H | 2-aminoethyl | 5,6-methylenedioxy[5,6-f] | - | methyl | 2,3-dihydro | |
| File:MeTriox structure.png | MeTriox | H | H | 2-aminopropyl | 5,6-methylenedioxy[5,6-f] | - | methyl | 2,3-dihydro | |
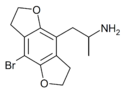 |
DOB-FLY | 219986-75-1 | H | H | 2-aminopropyl | 5,6-dihydrofuro[5,6-f] | - | bromo | 2,3-dihydro |
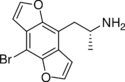 |
Bromo-DragonFLY | 502759-67-3 | H | H | 2-aminopropyl | furo[5,6-f] | - | bromo | - |
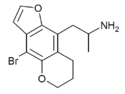 |
DOB-2-DRAGONFLY-5-BUTTERFLY | 1043541-82-7 | H | H | 2-aminopropyl | 5,6-dihydropyrano | - | bromo | - |
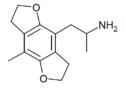 |
DOM-FLY | 748748-08-5 | H | H | 2-aminopropyl | 5,6-dihydrofuro[5,6-f] | - | methyl | 2,3-dihydro |
| File:DOMOM-Fly structure.png | DOMOM-FLY [50] | H | H | 2-aminopropyl | 5,6-dihydrofuro[5,6-f] | - | methoxymethyl | 2,3-dihydro | |
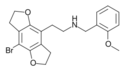 |
2C-B-FLY-NBOMe | 1335331-42-4 | H | H | 2-[(2-methoxybenzyl)-amino]ethyl | 5,6-dihydrofuro[5,6-f] | - | bromo | 2,3-dihydro |
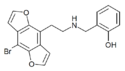 |
2C-B-DRAGONFLY-NBOH | 1335331-45-7 | H | H | 2-[(2-hydroxybenzyl)-amino]ethyl | furo[5,6-f] | - | bromo | - |
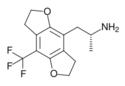 |
TFMFly | 780744-19-6 | H | H | 2-aminopropyl | 5,6-dihydrofuro[5,6-f] | - | trifluoromethyl | 2,3-dihydro |
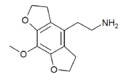 |
Mescaline-FLY | H | H | 2-aminoethyl | 5,6-dihydrofuro[5,4-b] | - | methoxy | 2,3-dihydro | |
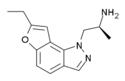 |
YM-348 | 372163-84-3 | ethyl | H | 1-(2-aminopropyl)-pyrazol[4,5-f] | - | H | H | - |
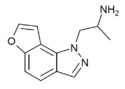 |
2-Desethyl-YM-348 | 748116-94-1 | H | H | 1-(2-aminopropyl)-pyrazol[4,5-f] | - | H | H | - |
See also
- Substituted methylenedioxyphenethylamine § Related compounds
- Substituted 2-aminoindane
- Substituted amphetamine
- Substituted benzothiophene
- Substituted cathinone
- Substituted methylenedioxyphenethylamine
- Substituted methoxyphenethylamine
- Substituted naphthylethylamine
- Substituted phenethylamine
- Substituted tryptamine
References
- ↑ "Benzofuran derivatives: a patent review". Expert Opinion on Therapeutic Patents 23 (9): 1133–56. September 2013. doi:10.1517/13543776.2013.801455. PMID 23683135.
- ↑ "Biological and medicinal significance of benzofuran". European Journal of Medicinal Chemistry 97: 561–81. June 2015. doi:10.1016/j.ejmech.2014.10.085. PMID 26015069.
- ↑ "Bioactive Benzofuran derivatives: A review". European Journal of Medicinal Chemistry 97: 483–504. June 2015. doi:10.1016/j.ejmech.2014.11.039. PMID 25482554.
- ↑ "Pharmacokinetics, pharmacodynamics and toxicology of new psychoactive substances (NPS): 2C-B, 4-fluoroamphetamine and benzofurans". Drug and Alcohol Dependence 157: 18–27. December 2015. doi:10.1016/j.drugalcdep.2015.10.011. PMID 26530501.
- ↑ "Identification of five substituted phenethylamine derivatives 5-MAPDB, 5-AEDB, MDMA methylene homolog, 6-Br-MDMA, and 5-APB-NBOMe". Drug Testing and Analysis 9 (2): 199–207. February 2017. doi:10.1002/dta.1955. PMID 26856255.
- ↑ "Pharmacology and Literature Review Based on Related Death and Non-Fatal Case Reports of the Benzofurans and Benzodifurans Designer Drugs". Current Pharmaceutical Design 23 (36): 5523–5529. 2017. doi:10.2174/1381612823666170714155140. PMID 28714411.
- ↑ "Comparison of the behavioral responses induced by phenylalkylamine hallucinogens and their tetrahydrobenzodifuran ("FLY") and benzodifuran ("DragonFLY") analogs". Neuropharmacology 144: 368–376. January 2019. doi:10.1016/j.neuropharm.2018.10.037. PMID 30385253.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 "Benzo fury: A new trend in the drug misuse scene". J Appl Toxicol 39 (8): 1083–1095. August 2019. doi:10.1002/jat.3774. PMID 30723925.
- ↑ 9.0 9.1 "Beyond ecstasy: Alternative entactogens to 3,4-methylenedioxymethamphetamine with potential applications in psychotherapy". J Psychopharmacol 35 (5): 512–536. May 2021. doi:10.1177/0269881120920420. PMID 32909493.
- ↑ "Monoamine Transporter and Receptor Interaction Profiles in Vitro Predict Reported Human Doses of Novel Psychoactive Stimulants and Psychedelics". Int J Neuropsychopharmacol 21 (10): 926–931. October 2018. doi:10.1093/ijnp/pyy047. PMID 29850881.
- ↑ 11.0 11.1 "Designer drugs: mechanism of action and adverse effects". Arch Toxicol 94 (4): 1085–1133. April 2020. doi:10.1007/s00204-020-02693-7. PMID 32249347.
- ↑ "Serotonergic Psychedelics: Experimental Approaches for Assessing Mechanisms of Action". Handb Exp Pharmacol 252: 227–260. 2018. doi:10.1007/164_2018_107. PMID 29532180.
- ↑ 13.0 13.1 13.2 13.3 Greene, Shaun L (2013). "Benzofurans and Benzodifurans". Novel Psychoactive Substances. Elsevier. p. 383–392. doi:10.1016/b978-0-12-415816-0.00016-x. ISBN 978-0-12-415816-0. https://linkinghub.elsevier.com/retrieve/pii/B978012415816000016X. Retrieved 2 November 2025. "A patent granted to Eli Lilly and Company in 2006 classifies 5-APB and 6-APB as 5HT2C receptor agonists [15]. [...] Internet user reports of 5-APB and 6-APB date from late 2010 [13]. [...] Information regarding the desired clinical effects of 5-APB and 6-APB is limited to on-line user report forums. Limited user reports indicate that positive effects of 5-APB include increased empathy, variable euphoria, visual disturbances, appreciation for music and dancing and more general ‘stimulation’ as opposed to 6-APB [12,23–25]. Reported positive effects of 6-APB include increased tactile and visual stimulation, mild euphoria, and appreciation for music, visual hallucinations and increase in mood, feelings of peace, love and self-acceptance [10,11,13,14]."
- ↑ 14.0 14.1 14.2 14.3 14.4 14.5 14.6 14.7 14.8 "The psychoactive aminoalkylbenzofuran derivatives, 5-APB and 6-APB, mimic the effects of 3,4-methylenedioxyamphetamine (MDA) on monoamine transmission in male rats". Psychopharmacology (Berl) 237 (12): 3703–3714. December 2020. doi:10.1007/s00213-020-05648-z. PMID 32875347. "The synthetic preparation of both 5-APB and 6-APB was first published in 2000 as part of a research program designed for the development of selective 5-HT2C receptor agonists (Briner et al. 2000; Briner et al. 2006) [...] Briner K, Burkhart JP, Burkholder TP, Fisher MJ, Gritton WH, Kohlman DT, Liang SX, Miller SC, Mullaney JT, Xu Y-C, Xu Y (2000) Aminoalkylbenzofurans as serotonin (5-HT(2C)) agonists Patent No. WO2000044737A1, Eli Lilly and Company, Indianapolis, IN, USA, 2000. Briner K, Burkhart JP, Burkholder TP, Fisher MJ, Gritton WH, Kohlman DT, Liang SX, Miller SC, Mullaney JT, Xu YC (2006) Aminoalkylbenzofurans as serotonin (5-HT(2C)) agonists Patent No. US7045545B1, Eli Lilly and Company, Indianapolis, IN, USA, 2006.".
- ↑ 15.0 15.1 15.2 "Advantageous benzofuran compositions for mental disorders or enhancement". 8 December 2022. https://patents.google.com/patent/US11767305B2/.
- ↑ 16.0 16.1 16.2 16.3 16.4 16.5 "Novel Benzofuran Derivatives Induce Monoamine Release and Substitute for the Discriminative Stimulus Effects of 3,4-Methylenedioxymethamphetamine". J Pharmacol Exp Ther 391 (1): 22–29. September 2024. doi:10.1124/jpet.123.001837. PMID 38272669.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 "A "Furious" Effort to Develop Novel 3,4-Methylenedioxymethamphetamine-Like Therapeutics". J Pharmacol Exp Ther 391 (1): 18–21. September 2024. doi:10.1124/jpet.124.002183. PMID 39293859.
- ↑ 18.0 18.1 "3,4-methylenedioxymethamphetamine (MDMA, "Ecstasy") induces fenfluramine-like proliferative actions on human cardiac valvular interstitial cells in vitro". Molecular Pharmacology 63 (6): 1223–1229. June 2003. doi:10.1124/mol.63.6.1223. PMID 12761331.
- ↑ "Dopamine-releasing agents". Dopamine Transporters: Chemistry, Biology and Pharmacology. Hoboken [NJ]: Wiley. July 2008. pp. 305–320. ISBN 978-0-470-11790-3. OCLC 181862653. https://bitnest.netfirms.com/external/Books/Dopamine-releasing-agents_c11.pdf.
- ↑ "Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin". Synapse 39 (1): 32–41. January 2001. doi:10.1002/1098-2396(20010101)39:1<32::AID-SYN5>3.0.CO;2-3. PMID 11071707.
- ↑ 21.0 21.1 "The designer methcathinone analogs, mephedrone and methylone, are substrates for monoamine transporters in brain tissue". Neuropsychopharmacology 37 (5): 1192–1203. April 2012. doi:10.1038/npp.2011.304. PMID 22169943.
- ↑ "The new psychoactive substances 5-(2-aminopropyl)indole (5-IT) and 6-(2-aminopropyl)indole (6-IT) interact with monoamine transporters in brain tissue". Neuropharmacology 101: 68–75. February 2016. doi:10.1016/j.neuropharm.2015.09.004. PMID 26362361.
- ↑ "The effects of non-medically used psychoactive drugs on monoamine neurotransmission in rat brain". European Journal of Pharmacology 559 (2–3): 132–137. March 2007. doi:10.1016/j.ejphar.2006.11.075. PMID 17223101.
- ↑ "Powerful cocaine-like actions of 3,4-methylenedioxypyrovalerone (MDPV), a principal constituent of psychoactive 'bath salts' products". Neuropsychopharmacology 38 (4): 552–562. March 2013. doi:10.1038/npp.2012.204. PMID 23072836.
- ↑ Sakloth, Farhana (11 December 2015). Psychoactive synthetic cathinones (or 'bath salts'): Investigation of mechanisms of action. VCU Scholars Compass (Thesis). doi:10.25772/AY8R-PW77. Retrieved 24 November 2024.
- ↑ "Eutylone and Its Structural Isomers Interact with Monoamine Transporters and Induce Locomotor Stimulation". ACS Chem Neurosci 12 (7): 1170–1177. April 2021. doi:10.1021/acschemneuro.0c00797. PMID 33689284.
- ↑ "Pharmacokinetic Profiles and Pharmacodynamic Effects for Methylone and Its Metabolites in Rats". Neuropsychopharmacology 42 (3): 649–660. February 2017. doi:10.1038/npp.2016.213. PMID 27658484.
- ↑ 28.0 28.1 28.2 28.3 "Pharmacological profile of novel psychoactive benzofurans". Br J Pharmacol 172 (13): 3412–3425. July 2015. doi:10.1111/bph.13128. PMID 25765500.
- ↑ 29.0 29.1 29.2 Matthew Baggott, "Advantageous benzofuran compositions for mental disorders or enhancement", US patent 20230150963, published 2023 May 18, assigned to Tactogen
- ↑ "Medicinal Chemistry and Structure–Activity Relationships". Amphetamine and Its Analogs: Psychopharmacology, Toxicology, and Abuse. Academic Press. 1994. pp. 3–41. ISBN 978-0-12-173375-9. https://bitnest.netfirms.com/external/Books/AmphetamineAndItsAnalogs3. "Very recently, the oxygen atoms in the dioxole ring of MDA were replaced individually with methylene units to give compounds 25, 26, and 27. In addition, the ring-expanded compound 28 was prepared for comparison. [...]"
- ↑ "Synthesis and pharmacological examination of benzofuran, indan, and tetralin analogues of 3,4-(methylenedioxy)amphetamine". J Med Chem 36 (23): 3700–3706. November 1993. doi:10.1021/jm00075a027. PMID 8246240.
- ↑ "Neurochemical profiles of some novel psychoactive substances". Eur J Pharmacol 700 (1-3): 147–151. January 2013. doi:10.1016/j.ejphar.2012.12.006. PMID 23261499.
- ↑ "The effects of benzofury (5-APB) on the dopamine transporter and 5-HT2-dependent vasoconstriction in the rat". Prog Neuropsychopharmacol Biol Psychiatry 48: 57–63. January 2014. doi:10.1016/j.pnpbp.2013.08.013. PMID 24012617.
- ↑ UK Home Office (2014-03-05). "The Misuse of Drugs Act 1971 (Ketamine etc.) (Amendment) Order 2014". UK Government. http://www.legislation.gov.uk/ukdsi/2014/9780111110904.
- ↑ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in zh). China Food and Drug Administration. 27 September 2015. http://www.sfda.gov.cn/WS01/CL0056/130753.html.
- ↑ "指定薬物名称・構造式一覧(平成27年9月16日現在)" (in ja). 厚生労働省. 16 September 2015. http://www.mhlw.go.jp/seisakunitsuite/bunya/kenkou_iryou/iyakuhin/yakubuturanyou/dl/meisho.pdf.
- ↑ "Benzofuran bioisosteres of hallucinogenic tryptamines". Journal of Medicinal Chemistry 35 (11): 2061–4. May 1992. doi:10.1021/jm00089a017. PMID 1534585.
- ↑ "Synthesis and pharmacological examination of benzofuran, indan, and tetralin analogues of 3,4-(methylenedioxy)amphetamine". Journal of Medicinal Chemistry 36 (23): 3700–6. November 1993. doi:10.1021/jm00075a027. PMID 8246240.
- ↑ Karin Briner, Joseph Paul Burkhart, Timothy Paul Burkholder, Matthew Joseph Fisher, William Harlan Gritton, Daniel Timothy Kohlman, Sidney Xi Liang, Shawn Christopher Miller, Jeffrey Thomas Mullaney, Yao-Chang Xu, Yanping Xu, "Aminoalkylbenzofurans as serotonin (5-HT(2c)) agonists", US patent 7045545, published 19 January 2000, issued 16 May 2006
- ↑ "Temporary class drug order report on 5-6APB and NBOMe compounds". UK Home Office. 4 Jun 2013. https://www.gov.uk/government/publications/temporary-class-drug-order-report-on-benzofury-and-nbome-compounds.
- ↑ "Identification of (2-aminopropyl)benzofuran (APB) phenyl ring positional isomers in internet purchased products". Drug Testing and Analysis 5 (4): 270–6. April 2013. doi:10.1002/dta.1451. PMID 23349125.
- ↑ "Synthesis and evaluation of 2,3-dihydrobenzofuran analogues of the hallucinogen 1-(2,5-dimethoxy-4-methylphenyl)-2-aminopropane: drug discrimination studies in rats". Journal of Medicinal Chemistry 29 (2): 302–4. February 1986. doi:10.1021/jm00152a022. PMID 3950910.
- ↑ "2,3-Dihydrobenzofuran analogues of hallucinogenic phenethylamines". Journal of Medicinal Chemistry 34 (1): 276–81. January 1991. doi:10.1021/jm00105a043. PMID 1992127.
- ↑ "Dihydrobenzofuran analogues of hallucinogens. 3. Models of 4-substituted (2,5-dimethoxyphenyl)alkylamine derivatives with rigidified methoxy groups". Journal of Medicinal Chemistry 39 (15): 2953–61. July 1996. doi:10.1021/jm960199j. PMID 8709129.
- ↑ "Identification of five substituted phenethylamine derivatives 5-MAPDB, 5-AEDB, MDMA methylene homolog, 6-Br-MDMA, and 5-APB-NBOMe". Drug Testing and Analysis 9 (2): 199–207. February 2017. doi:10.1002/dta.1955. PMID 26856255.
- ↑ "Interactions of phenethylamine-derived psychoactive substances of the 2C-series with human monoamine oxidases". Drug Testing and Analysis 11 (2): 318–324. February 2019. doi:10.1002/dta.2494. PMID 30188017. http://researchonline.ljmu.ac.uk/id/eprint/9155/1/DTA-18-0224.R1.pdf.
- ↑ "Phenethylamine-derived new psychoactive substances 2C-E-FLY, 2C-EF-FLY, and 2C-T-7-FLY: Investigations on their metabolic fate including isoenzyme activities and their toxicological detectability in urine screenings". Drug Testing and Analysis 11 (10): 1507–1521. October 2019. doi:10.1002/dta.2675. PMID 31299701.
- ↑ "PiHKAL·info". 12 June 2025. https://isomerdesign.com/pihkal/explore/2981.
- ↑ "2-MABB" (in ru). https://aipsin.com/newsubstance/149/.
- ↑ "Novel benzodifuran analogs as potent 5-HT2A receptor agonists with ocular hypotensive activity". Bioorg Med Chem Lett 17 (11): 2998–3002. Jun 2007. doi:10.1016/j.bmcl.2007.03.073. PMID 17419053.
 |
 KSF
KSF










