Sulfate
Topic: Chemistry
 From HandWiki - Reading time: 14 min
From HandWiki - Reading time: 14 min
The sulfate or sulphate ion is a polyatomic anion with the empirical formula SO
42-
. Salts, acid derivatives, and peroxides of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salts of sulfuric acid and many are prepared from that acid.
Spelling
"Sulfate" is the spelling recommended by IUPAC, but "sulphate" is traditionally used in British English.
Structure
The sulfate anion consists of a central sulfur atom surrounded by four equivalent oxygen atoms in a tetrahedral arrangement. The symmetry of the isolated anion is the same as that of methane. The sulfur atom is in the +6 oxidation state while the four oxygen atoms are each in the −2 state. The sulfate ion carries an overall charge of −2 and it is the conjugate base of the bisulfate (or hydrogensulfate) ion, HSO
4-
, which is in turn the conjugate base of H
2SO
4, sulfuric acid. Organic sulfate esters, such as dimethyl sulfate, are covalent compounds and esters of sulfuric acid. The tetrahedral molecular geometry of the sulfate ion is as predicted by VSEPR theory.
Bonding

1 with polar covalent bonds only; 2 with an ionic bond
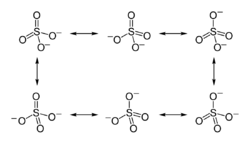
The first description of the bonding in modern terms was by Gilbert Lewis in his groundbreaking paper of 1916, where he described the bonding in terms of electron octets around each atom. There are two double bonds, and there is a formal charge of 2 on the sulfur atom and -1 on each oxygen atom.[1][lower-alpha 1]
Later, Linus Pauling used valence bond theory to propose that the most significant resonance canonicals had two pi bonds involving d orbitals. His reasoning was that the charge on sulfur was thus reduced, in accordance with his principle of electroneutrality.[2] The S−O bond length of 149 pm is shorter than the bond lengths in sulfuric acid of 157 pm for S−OH. The double bonding was taken by Pauling to account for the shortness of the S−O bond.
Pauling's use of d orbitals provoked a debate on the relative importance of pi bonding and bond polarity (electrostatic attraction) in causing the shortening of the S−O bond. The outcome was a broad consensus that d orbitals play a role, but are not as significant as Pauling had believed.[3][4]
A widely accepted description involving pπ – dπ bonding was initially proposed by Durward William John Cruickshank. In this model, fully occupied p orbitals on oxygen overlap with empty sulfur d orbitals (principally the dz2 and dx2–y2).[5] However, in this description, despite there being some π character to the S−O bonds, the bond has significant ionic character. For sulfuric acid, computational analysis (with natural bond orbitals) confirms a clear positive charge on sulfur (theoretically +2.45) and a low 3d occupancy. Therefore, the representation with four single bonds is the optimal Lewis structure rather than the one with two double bonds (thus the Lewis model, not the Pauling model).[6]
In this model, the structure obeys the octet rule and the charge distribution is in agreement with the electronegativity of the atoms. The discrepancy between the S−O bond length in the sulfate ion and the S−OH bond length in sulfuric acid is explained by donation of p-orbital electrons from the terminal S=O bonds in sulfuric acid into the antibonding S−OH orbitals, weakening them resulting in the longer bond length of the latter.
However, Pauling's representation for sulfate and other main group compounds with oxygen is still a common way of representing the bonding in many textbooks.[5][7] The apparent contradiction can be clarified if one realizes that the covalent double bonds in the Lewis structure actually represent bonds that are strongly polarized by more than 90% towards the oxygen atom. On the other hand, in the structure with a dipolar bond, the charge is localized as a lone pair on the oxygen.[6]
Preparation
Typically metal sulfates are prepared by treating metal oxides, metal carbonates, or the metal itself with sulfuric acid:[7]
- Zn + H
2SO
4 → ZnSO
4 + H
2 - Cu(OH)
2 + H
2SO
4 → CuSO
4 + 2 H
2O - CdCO
3 + H
2SO
4 → CdSO
4 + H
2O + CO
2
Although written with simple anhydrous formulas, these conversions generally are conducted in the presence of water. Consequently the product sulfates are hydrated, corresponding to zinc sulfate ZnSO
4 · 7H2O, copper(II) sulfate CuSO
4 · 5H2O, and cadmium sulfate CdSO
4 · H2O.
Some metal sulfides can be oxidized to give metal sulfates.
Properties
There are numerous examples of ionic sulfates, many of which are highly soluble in water. Exceptions include calcium sulfate, strontium sulfate, lead(II) sulfate, barium sulfate, silver sulfate, and mercury sulfate, which are poorly soluble. Radium sulfate is the most insoluble sulfate known. The barium derivative is useful in the gravimetric analysis of sulfate: if one adds a solution of most barium salts, for instance barium chloride, to a solution containing sulfate ions, barium sulfate will precipitate out of solution as a whitish powder. This is a common laboratory test to determine if sulfate anions are present.
The sulfate ion can act as a ligand attaching either by one oxygen (monodentate) or by two oxygens as either a chelate or a bridge.[7] An example is the complex [Co(en)
2(SO
4)]+
Br−
[7] or the neutral metal complex [PtSO
4(PPh
3)
2] where the sulfate ion is acting as a bidentate ligand. The metal–oxygen bonds in sulfate complexes can have significant covalent character.
Uses and occurrence
Commercial applications

Sulfates are widely used industrially. Major compounds include:
- Gypsum, the natural mineral form of hydrated calcium sulfate, is used to produce plaster. About 100 million tonnes per year are used by the construction industry.
- Copper sulfate, a common fungicide, the more stable pentahydrate form (CuSO
4 · 5H2O) is used for Bordeaux mixture in agriculture, galvanic cells as electrolyte and pigment. - Iron(II) sulfate, a common form of iron in mineral supplements for humans, animals, and soil for plants.
- Magnesium sulfate (commonly known as Epsom salts), used in therapeutic baths.
- Lead(II) sulfate, produced on both plates during the discharge of a lead–acid battery.
- Sodium laureth sulfate, or SLES, a common detergent in shampoo formulations.
- Polyhalite, K
2Ca
2Mg(SO
4)
4 · 2H2O, used as fertiliser.
Occurrence in nature
Sulfate-reducing bacteria, some anaerobic microorganisms, such as those living in sediment or near deep sea thermal vents, use the reduction of sulfates coupled with the oxidation of organic compounds or hydrogen as an energy source for chemosynthesis.
History
Various forms of calcium sulphate have been used as building materials since the Bronze Age, and other sulphates have been utilized since at least antiquity. However, the first salts of this group were chemically described only in the 17th century. The production of sulphuric acid esters, as well as their biological significance and natural occurrence, has been known only since the 19th century.
In the eastern Mediterranean and the Middle East, the use of mortar was widespread thousands of years ago. In ancient Egypt, its application is documented as early as the third millennium BC. Archaeological sites demonstrating its use include the Old Palace of Aššur and the ruins of Amarna. In the Parthian Empire, plaster mortar was employed in the construction of vaults. From the period of the Roman Empire until the 19th century, lime mortar (containing calcium carbonate) was far more widely used, although gypsum mortar continued to be applied to a limited extent during the Middle Ages, for example in France.[8] The use of calcium sulphate in cement has been investigated since the late 19th century and has been widely adopted since the 1930s.[9] During the Bronze Age, alabaster was extensively used for decorative architectural elements in the Minoan culture. Toward the end of the Bronze Age, gypsum alabaster from Cretan quarries was exported and used elsewhere, for example for benches in Mycenae. In the ruins of Akrotiri on Santorinii, it was used for floor tiles.[10] Alabaster is easily worked and became a common and highly valued material for sculptures and monuments during the Middle Ages and the modern period. It was quarried primarily in central England, northern Spain, and the French Alps, and traded over long distances. In 1550, religious sculptures were banned in England (see Reformation iconoclasm), leading to the large-scale export of alabaster figures to France.[11]
Some sulfates were known to alchemists. The vitriol salts, from the Latin vitreolum, glassy, were so-called because they were some of the first transparent crystals known.[12] Green vitriol is iron(II) sulfate heptahydrate, FeSO
4 · 7H2O; blue vitriol is copper(II) sulfate pentahydrate, CuSO
4 · 5H2O and white vitriol is zinc sulfate heptahydrate, ZnSO
4 · 7H2O. Alum, a double sulfate of potassium and aluminium with the formula K
2Al
2(SO
4)
4 · 24H2O, figured in the development of the chemical industry.
Sulphates were historically used as dyes and pigments. In Ancient Egypt, anhydrite screed and jarosite, KFe3[(OH)6(SO4)2], were employed in wall decoration.[13] The pigment jarosite has also been identified in Central America in vessels from a burial site in Teotihuacán.[14] The use of iron gall ink was widespread during the Middle Ages and continued into the modern era. It was prepared from plant gall and iron(II) sulphate.[15] Alum (potassium aluminum sulfate) was formerly used in leather production (tanning), possibly as early as ancient Egypt.[16] It was certainly known in classical antiquity in Rome and Greece.[17] Alum tanning was widespread in antiquity and the Middle Ages; however, the effect was not permanent, as the alum could be washed out of the leather.[16] Throughout the Middle Ages, alum was an important industrial product. In addition to its use in leather processing, it served as a mordant in wool dyeing. From the mid-19th century onward, it was gradually replaced by other compounds, particularly aluminum sulfate, and is now of only minor significance.[17]
The first sulphuric acid salts were identified as such in the 17th century. Sodium sulphate, specifically its decahydrate known as Glauber's salt, was described around 1625 by Johann Rudolph Glauber. He analyzed water from a medicinal spring near Naples and isolated sodium sulphate, which he named Sal mirabile. Several years later, he determined that sodium sulphate could be produced from rock salt (sodium chloride) and sulphuric acid.[18] Magnesium sulphate obtained from a mineral spring in Epsom, England, was scientifically described at the end of the 17th century. Both the spring water and the isolated salt exhibited medicinal properties and were used as a laxative and for the treatment of headaches.[19]
The medical application of plaster for the treatment of bone fracture began in the early 19th century. Initially, wooden boxes filled with cast plaster were used. Although widespread in Europe, this method was impractical because the heavy plaster constructions confined patients to bed. Plaster-impregnated bandages for plaster casts were introduced in the mid-19th century and were initially prepared fresh in hospitals. Ready-to-use plaster bandages did not become commercially available until the 1930s.[20] Another important medical development was the use of magnesium sulphate for the treatment of seizures during pregnancy (eclampsia). This application was first reported in 1916. By 1930, magnesium sulphate had largely replaced less suitable treatments, such as opioids, and contributed significantly to the reduction of maternal mortality.[21]
Environmental effects
Sulfates occur as microscopic particles (aerosols) resulting from fossil fuel and biomass combustion. They increase the acidity of the atmosphere and form acid rain. The anaerobic sulfate-reducing bacteria Desulfovibrio desulfuricans and D. vulgaris can remove the black sulfate crust that often tarnishes buildings.[22]
Main effects on climate
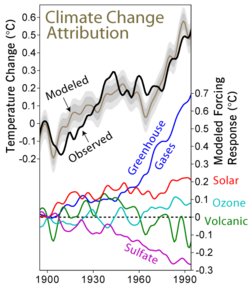

Reversal and accelerated warming
Hydrological cycle
Solar geoengineering
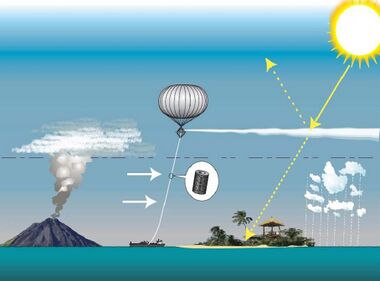
As the real world had shown the importance of sulfate aerosol concentrations to the global climate, research into the subject accelerated. Formation of the aerosols and their effects on the atmosphere can be studied in the lab, with methods like ion-chromatography and mass spectrometry[23] Samples of actual particles can be recovered from the stratosphere using balloons or aircraft,[24] and remote satellites were also used for observation.[25] This data is fed into the climate models,[26] as the necessity of accounting for aerosol cooling to truly understand the rate and evolution of warming had long been apparent, with the IPCC Second Assessment Report being the first to include an estimate of their impact on climate, and every major model able to simulate them by the time IPCC Fourth Assessment Report was published in 2007.[27] Many scientists also see the other side of this research, which is learning how to cause the same effect artificially.[28] While discussed around the 1990s, if not earlier,[29] stratospheric aerosol injection as a solar geoengineering method is best associated with Paul Crutzen's detailed 2006 proposal.[30] Deploying in the stratosphere ensures that the aerosols are at their most effective, and that the progress of clean air measures would not be reversed: more recent research estimated that even under the highest-emission scenario RCP 8.5, the addition of stratospheric sulfur required to avoid 4 °C (7.2 °F) relative to now (and 5 °C (9.0 °F) relative to the preindustrial) would be effectively offset by the future controls on tropospheric sulfate pollution, and the amount required would be even less for less drastic warming scenarios.[31] This spurred a detailed look at its costs and benefits,[32] but even with hundreds of studies into the subject completed by the early 2020s, some notable uncertainties remain.[33]
Hydrogensulfate (bisulfate)
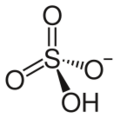
| |
| Names | |
|---|---|
| IUPAC name
Hydrogensulfate[34]
| |
| Other names
Bisulfate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| 2121 | |
PubChem CID
|
|
| |
| |
| Properties | |
| HSO− 4 | |
| Molar mass | 97.071 g/mol |
| Conjugate acid | Sulfuric acid |
| Conjugate base | Sulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
The hydrogensulfate ion (HSO−
4), also called the bisulfate ion, is the conjugate base of sulfuric acid (H
2SO
4).[35][lower-alpha 2] Sulfuric acid is classified as a strong acid; in aqueous solutions it ionizes completely to form hydronium (H
3O+
) and hydrogensulfate (HSO−
4) ions. In other words, the sulfuric acid behaves as a Brønsted–Lowry acid and is deprotonated to form hydrogensulfate ion. Hydrogensulfate has a valency of 1. An example of a salt containing the HSO−
4 ion is sodium bisulfate, NaHSO
4. In dilute solutions the hydrogensulfate ions also dissociate, forming more hydronium ions and sulfate ions (SO2−
4).
Other sulfur oxyanions
| Molecular formula | Name |
|---|---|
| SO2− 5 |
Peroxomonosulfate |
| SO2− 4 |
Sulfate |
| SO2− 3 |
Sulfite |
| S 2O2− 8 |
Peroxydisulfate |
| S 2O2− 7 |
Pyrosulfate |
| S 2O2− 6 |
Dithionate |
| S 2O2− 5 |
Metabisulfite |
| S 2O2− 4 |
Dithionite |
| S 2O2− 3 |
Thiosulfate |
| S 3O2− 6 |
Trithionate |
| S 4O2− 6 |
Tetrathionate |
See also
- Sulfonate
- Sulfation and desulfation of lead–acid batteries
- Sulfate-reducing microorganism
Notes
- ↑ Lewis assigned to sulfur a negative charge of two, starting from six own valence electrons and ending up with eight electrons shared with the oxygen atoms. In fact, sulfur donates two electrons to the oxygen atoms.
- ↑ The prefix "bi" in "bisulfate" comes from an outdated naming system and is based on the observation that there is twice as much sulfate (SO2−
4) in sodium bisulfate (NaHSO
4) and other bisulfates as in sodium sulfate (Na
2SO
4) and other sulfates. See also bicarbonate.
References
- ↑ Lewis, Gilbert N. (1916). "The Atom and the Molecule". J. Am. Chem. Soc. 38 (4): 762–785. doi:10.1021/ja02261a002. http://osulibrary.oregonstate.edu/specialcollections/coll/pauling/bond/papers/corr216.3-lewispub-19160400-18-large.html. (See page 778.)
- ↑ Pauling, Linus (1948). "The modern theory of valency". J. Chem. Soc. 17: 1461–1467. doi:10.1039/JR9480001461. PMID 18893624. https://authors.library.caltech.edu/59671/.
- ↑ Coulson, C. A. (1969). "d Electrons and Molecular Bonding". Nature 221 (5186): 1106. doi:10.1038/2211106a0. Bibcode: 1969Natur.221.1106C.
- ↑ Mitchell, K. A. R. (1969). "Use of outer d orbitals in bonding". Chem. Rev. 69 (2): 157. doi:10.1021/cr60258a001.
- ↑ 5.0 5.1 Cotton, F. Albert; Wilkinson, Geoffrey (1966). Advanced Inorganic Chemistry (2nd ed.). New York, NY: Wiley.
- ↑ 6.0 6.1 Stefan, Thorsten; Janoschek, Rudolf (Feb 2000). "How relevant are S=O and P=O Double Bonds for the Description of the Acid Molecules H2SO3, H2SO4, and H3PO4, respectively?". J. Mol. Modeling 6 (2): 282–288. doi:10.1007/PL00010730.
- ↑ 7.0 7.1 7.2 7.3 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Paolo Vitti (October 2021), "Mortars and masonry—structural lime and gypsum mortars in Antiquity and Middle Ages", Archaeological and Anthropological Sciences 13 (10), doi:10.1007/s12520-021-01408-y
- ↑ José da Silva Andrade Neto, Angeles G. De la Torre, Ana Paula Kirchheim (April 2021), "Effects of sulfates on the hydration of Portland cement – A review", Construction and Building Materials 279: 122428, doi:10.1016/j.conbuildmat.2021.122428
- ↑ Zofia Anna Stos-Gale (June 1992), "Isotope archaeology: reading the past in metals, minerals, and bone", Endeavour 16 (2): 85–90, doi:10.1016/0160-9327(92)90007-C
- ↑ W. Kloppmann, L. Leroux, P. Bromblet, P.-Y. Le Pogam, A. H. Cooper, N. Worley, C. Guerrot, A. T. Montech, A. M. Gallas, R. Aillaud (2017-11-07), "Competing English, Spanish, and French alabaster trade in Europe over five centuries as evidenced by isotope fingerprinting", Proceedings of the National Academy of Sciences 114 (45): 11856–11860, doi:10.1073/pnas.1707450114, PMID 29078309
- ↑ Taylor, F. Sherwood (1942). Inorganic and Theoretical Chemistry (6th ed.). William Heinemann.
- ↑ J. Ambers (August 2004), "Raman analysis of pigments from the Egyptian Old Kingdom", Journal of Raman Spectroscopy 35 (8–9): 768–773, doi:10.1002/jrs.1187
- ↑ María Teresa Doménech-Carbó, María Luisa Vázquez de Agredos-Pascual, Laura Osete-Cortina, Antonio Doménech-Carbó, Núria Guasch-Ferré, Linda R. Manzanilla, Cristina Vidal-Lorenzo (April 2012), "Characterization of prehispanic cosmetics found in a burial of the ancient city of Teotihuacan (Mexico)", Journal of Archaeological Science 39 (4): 1043–1062, doi:10.1016/j.jas.2011.12.001
- ↑ Alana S. Lee, Peter J. Mahon, Dudley C. Creagh (August 2006), "Raman analysis of iron gall inks on parchment", Vibrational Spectroscopy 41 (2): 170–175, doi:10.1016/j.vibspec.2005.11.006
- ↑ 16.0 16.1 Karina Grömer, Gabriela Russ-Popa, Konstantina Saliari (2017), "Products of animal skin from Antiquity to the Medieval Period", Annalen des Naturhistorischen Museums in Wien. Serie A für Mineralogie und Petrographie, Geologie und Paläontologie, Anthropologie und Prähistorie 119: 69–93
- ↑ 17.0 17.1 Fritz Ullmann (1985), Ullmanns Enzyklopädie der Technischen Chemie, 1 (5. ed.), Weinheim: VCH, pp. 527–533, ISBN 3-527-20101-7
- ↑ James C. Hill (September 1979), "Johann Glauber’s discovery of sodium sulfate – Sal Mirabile Glauberi", Journal of Chemical Education 56 (9): 593, doi:10.1021/ed056p593
- ↑ Fortes, A. D.. ""The Story of Epsomite."" (PDF). https://cdn.mineralogicalrecord.com/wp-content/uploads/2020/10/pdfs/Epsomite-Article-Figs.pdf.
- ↑ B. Szostakowski, P. Smitham, W.S. Khan (2017-04-17), "Plaster of Paris–Short History of Casting and Injured Limb Immobilzation", The Open Orthopaedics Journal 11 (1): 291–296, doi:10.2174/1874325001711010291, PMID 28567158
- ↑ Linda A. Hunter, Karen J. Gibbins (November 2011), "Magnesium Sulfate: Past, Present, and Future", Journal of Midwifery & Women’s Health 56 (6): 566–574, doi:10.1111/j.1542-2011.2011.00121.x
- ↑ Andrea Rinaldi (Nov 2006). "Saving a fragile legacy. Biotechnology and microbiology are increasingly used to preserve and restore the worlds cultural heritage". EMBO Reports 7 (11): 1075–1079. doi:10.1038/sj.embor.7400844. PMID 17077862.
- ↑ Kobayashi, Yuya; Ide, Yu; Takegawa, Nobuyuki (3 April 2021). "Development of a novel particle mass spectrometer for online measurements of refractory sulfate aerosols". Aerosol Science and Technology 55 (4): 371–386. doi:10.1080/02786826.2020.1852168. ISSN 0278-6826. Bibcode: 2021AerST..55..371K. https://doi.org/10.1080/02786826.2020.1852168.
- ↑ Palumbo, P.; A. Rotundi; V. Della Corte; A. Ciucci; L. Colangeli; F. Esposito; E. Mazzotta Epifani; V. Mennella et al.. "The DUSTER experiment: collection and analysis of aerosol in the high stratosphere". Societa Astronomica Italiana. https://www.researchgate.net/publication/234296252_DUSTER_Aerosol_collection_in_the_stratosphere. Retrieved 19 February 2009.
- ↑ Myhre, Gunnar; Stordal, Frode; Berglen, Tore F.; Sundet, Jostein K.; Isaksen, Ivar S. A. (1 March 2004). "Uncertainties in the Radiative Forcing Due to Sulfate Aerosols" (in EN). Journal of the Atmospheric Sciences 61 (5): 485–498. doi:10.1175/1520-0469(2004)061<0485:UITRFD>2.0.CO;2. ISSN 0022-4928. Bibcode: 2004JAtS...61..485M.
- ↑ Zhang, Jie; Furtado, Kalli; Turnock, Steven T.; Mulcahy, Jane P.; Wilcox, Laura J.; Booth, Ben B.; Sexton, David; Wu, Tongwen et al. (22 December 2021). "The role of anthropogenic aerosols in the anomalous cooling from 1960 to 1990 in the CMIP6 Earth system models" (in en). Atmospheric Chemistry and Physics 21 (4): 18609–18627. doi:10.5194/acp-21-18609-2021. Bibcode: 2021ACP....2118609Z. https://acp.copernicus.org/articles/21/18609/2021/.
- ↑ "Aerosols and Incoming Sunlight (Direct Effects)". NASA. 2 November 2010. https://earthobservatory.nasa.gov/features/Aerosols/page3.php.
- ↑ "Stratospheric Injections Could Help Cool Earth, Computer Model Shows". ScienceDaily. 15 September 2006. https://www.sciencedaily.com/releases/2006/09/060914182715.htm.
- ↑ Launder B.; J.M.T. Thompson (1996). "Global and Arctic climate engineering: numerical model studies". Phil. Trans. R. Soc. A 366 (1882): 4039–56. doi:10.1098/rsta.2008.0132. PMID 18757275. Bibcode: 2008RSPTA.366.4039C.
- ↑ Crutzen, Paul J. (25 July 2006). "Albedo Enhancement by Stratospheric Sulfur Injections: A Contribution to Resolve a Policy Dilemma?" (in en). Climatic Change 77 (3-4). doi:10.1007/s10584-006-9101-y. ISSN 0165-0009. https://link.springer.com/10.1007/s10584-006-9101-y.
- ↑ Visioni, Daniele; Slessarev, Eric; MacMartin, Douglas G; Mahowald, Natalie M; Goodale, Christine L; Xia, Lili (1 September 2020). "What goes up must come down: impacts of deposition in a sulfate geoengineering scenario". Environmental Research Letters 15 (9): 094063. doi:10.1088/1748-9326/ab94eb. ISSN 1748-9326. Bibcode: 2020ERL....15i4063V.
- ↑ "Costs and benefits of geo-engineering in the Stratosphere". http://www.met.reading.ac.uk/pg-research/downloads/2009/pgr-charlton.pdf.
- ↑ Trisos, Christopher H.; Geden, Oliver; Seneviratne, Sonia I.; Sugiyama, Masahiro; van Aalst, Maarten; Bala, Govindasamy; Mach, Katharine J.; Ginzburg, Veronika et al. (2021). "Cross-Working Group Box SRM: Solar Radiation Modification". Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change 2021: 1238. doi:10.1017/9781009157896.007. Bibcode: 2021AGUFM.U13B..05K. https://www.ipcc.ch/report/ar6/wg2/downloads/report/IPCC_AR6_WGII_Chapter16.pdf.
- ↑ Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005, IUPAC, p. 129, http://old.iupac.org/publications/books/rbook/Red_Book_2005.pdf
- ↑ Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005, IUPAC, p. 129, http://old.iupac.org/publications/books/rbook/Red_Book_2005.pdf
External links
 |
 KSF
KSF