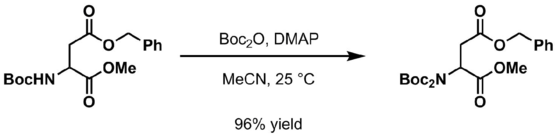Tert-Butyloxycarbonyl protecting group
Topic: Chemistry
 From HandWiki - Reading time: 4 min
From HandWiki - Reading time: 4 min
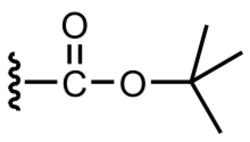
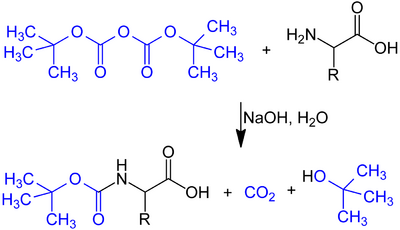
The tert-butyloxycarbonyl protecting group or tert-butoxycarbonyl protecting group[1] (BOC group) is a protecting group used in organic synthesis.
The BOC group can be added to amines under aqueous conditions using di-tert-butyl dicarbonate in the presence of a base such as sodium carbonate:
Protection of amines can also be accomplished in acetonitrile solution using 4-dimethylaminopyridine (DMAP) as the base.
Removal of the BOC group in amino acids can be accomplished with strong acids such as trifluoroacetic acid in dichloromethane, or with HCl in methanol.[2][3][4] A complication may be the tendency of the t-butyl cation intermediate to alkylate other nucleophiles; scavengers such as anisole or thioanisole may be used.[5][6] Selective cleavage of the N-Boc group in the presence of other protecting groups is possible when using AlCl3.
Sequential treatment with trimethylsilyl iodide then methanol can also be used for Boc deprotection,[7][8] especially where other deprotection methods are too harsh for the substrate.[9] The mechanism involves silylation of the carbonyl oxygen and elimination of tert-butyl iodide (1), methanolysis of the silyl ester to the carbamic acid (2) and finally decarboxylation to the amine (3).[10]
-
R2NCO2tBu + Me3SiI → R2NCO2SiMe3 + tBuI
()
-
R2NCO2SiMe3 + MeOH → R2NCO2H + MeOSiMe3
()
-
R2NCO2H → R2NH + CO2
()
Amine protection
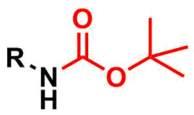
The tert-butyloxycarbonyl (Boc) group is used as a protecting group for amines in organic synthesis.
Common amine protection methods
- Simple rapid stirring of a mixture of the amine and di-tert-butyl dicarbonate (Boc2O) suspended in water at ambient temperature, an example of an on-water reaction.[11]
- Heating a mixture of the amine to be protected and di-tert-butyl dicarbonate in tetrahydrofuran (THF) at 40 °C[12]
- Add the amine to sodium hydroxide and di-tert-butyl dicarbonate in water and THF at 0 °C then warm to ambient temperature.[13]
- Heating a mixture of the amine to be protected and di-tert-butyl dicarbonate in a biphasic mixture of chloroform and aqueous sodium bicarbonate at reflux for 90 minutes.[14]
- Add the amine to di-tert-butyl dicarbonate, 4-dimethylaminopyridine (DMAP), and acetonitrile (MeCN) at ambient temperature[15]
BOC-protected amines are prepared using the reagent di-tert-butyl-iminodicarboxylate. Upon deprotonation, this reagent affords a doubly BOC-protected source of NH−2, which can be N-alkylated. The approach is complementary to the Gabriel synthesis of amines.
Common amine deprotection methods
- Mix the protected carbamate to be deprotected with 3 M hydrochloric acid (HCl) in ethyl acetate for 30 min at ambient temperature[16]
- Heat the carbamate in a mixture of aqueous hydrochloric acid and toluene at 65 °C[17]
- Dissolving desired protected compound in a 50/50 mix of dichloromethane and trifluoroacetic acid[18]
References
- ↑ "Rules for abbreviation of protecting groups". iupac.org. 2013. http://iupac.org/publications/pac/pdf/2013/pdf/8501x0307.pdf.
- ↑ Robert M. Williams; Peter J. Sinclair; Duane E. DeMong; Daimo Chen; Dongguan Zhai (2003). "Asymmetric Synthesis of N-tert-Butoxycarbonyl α-Amino Acids. Synthesis of (5S,6R)-4-tert-Butoxycarbonyl-5,6-diphenylmorpholin-2-one (4-Morpholinecarboxylic acid, 6-oxo-2,3-diphenyl-, 1,1-dimethylethyl ester, (2S,3R)-)"]. Organic Syntheses 80: 18. doi:10.15227/orgsyn.080.0018. http://www.orgsyn.org/demo.aspx?prep=v80p0018.
- ↑ E. A. Englund; H. N. Gopi; D. H. Appella (2004). "An Efficient Synthesis of a Probe for Protein Function: 2,3-Diaminopropionic Acid with Orthogonal Protecting Groups". Org. Lett. 6 (2): 213–215. doi:10.1021/ol0361599. PMID 14723531.
- ↑ D. M. Shendage; R. Fröhlich; G. Haufe (2004). "Highly Efficient Stereoconservative Amidation and Deamidation of α-Amino Acids". Org. Lett. 6 (21): 3675–3678. doi:10.1021/ol048771l. PMID 15469321.
- ↑ Lundt, Behrend F.; Johansen, Nils L.; Vølund, Aage; Markussen, Jan (1978). "Removal of t-Butyl and t-Butoxycarbonyl Protecting Groups with Trifluoroacetic acid". Int. J. Pept. Protein Res. 12 (5): 258–268. doi:10.1111/j.1399-3011.1978.tb02896.x. PMID 744685.
- ↑ Vommina V. Sureshbabu; Narasimhamurthy Narendra (2011). "Protection Reactions". in Andrew B. Hughes. Protection Reactions, Medicinal Chemistry, Combinatorial Synthesis. Amino Acids, Peptides and Proteins in Organic Chemistry. 4. Wiley-VCH. pp. XVIII – LXXXIV. doi:10.1002/9783527631827.ch1. ISBN 9783527641574. https://books.google.com/books?id=uZK2i-FkeAUC&pg=PP18.
- ↑ Richard S. Lott; Virander S. Chauhan; Charles H. Stammer (1979). "Trimethylsilyl iodide as a peptide deblocking agent". J. Chem. Soc., Chem. Commun. (11): 495–496. doi:10.1039/C39790000495.
- ↑ Olah, G; Narang, S. C. (1982). "Iodotrimethylsilane—a versatile synthetic reagent". Tetrahedron 38 (15): 2225–2277. doi:10.1016/0040-4020(82)87002-6.
- ↑ Zhijian Liu; Nobuyoshi Yasuda; Michael Simeone; Robert A. Reamer (2014). "N-Boc Deprotection and Isolation Method for Water-Soluble Zwitterionic Compounds". J. Org. Chem. 79 (23): 11792–11796. doi:10.1021/jo502319z. PMID 25376704.
- ↑ Michael E. Jung; Mark A. Lyster (1978). "Conversion of alkyl carbamates into amines via treatment with trimethylsilyl iodide". J. Chem. Soc., Chem. Commun. (7): 315–316. doi:10.1039/C39780000315.
- ↑ Chankeshwara, Sunay V.; Chakraborti, Asit K. (2006). "Catalyst-Free Chemoselective N-tert-Butyloxycarbonylation of Amines in Water". Org. Lett. 8 (15): 3259–3262. doi:10.1021/ol0611191. PMID 16836380.
- ↑ Wuts, Peter G. M.; Greene, Theodora W. (2006). "Protection for the Amino Group". Greene's Protective Groups in Organic Synthesis (4th ed.). John Wiley & Sons. pp. 696–926. doi:10.1002/9780470053485.ch7. ISBN 9780471697541. http://onlinelibrary.wiley.com/book/10.1002/0470053488.
- ↑ Fachao Yan; Hanbing Liang; Jian Song; Jie Cui; Qing Liu; Sheng Liu; Ping Wang; Yunhui Dong et al. (2017). "Palladium-Catalyzed Cyclization-Heck Reaction of Allenamides: An Approach to 3-Methylene-5-phenyl-1,2,3,4-tetrahydropyridine Derivatives". Org. Lett. 19 (1): 86–89. doi:10.1021/acs.orglett.6b03364. PMID 27966983.
- ↑ Tarbell, D. Stanley; Yamamoto, Yutaka; Pope, Barry M. (1972). "New Method to Prepare N-t-Butoxycarbonyl Derivatives and the Corresponding Sulfur Analogs from di-t-Butyl Dicarbonate or di-t-Butyl Dithiol Dicarbonates and Amino Acids". Proc. Natl. Acad. Sci. U.S.A. 69 (3): 730–732. doi:10.1073/pnas.69.3.730. PMID 16591972. Bibcode: 1972PNAS...69..730T.
- ↑ Englund, Ethan A.; Gopi, Hosahudya N.; Appella, Daniel H. (2004). "An Efficient Synthesis of a Probe for Protein Function: 2,3-Diaminopropionic Acid with Orthogonal Protecting Groups". Org. Lett. 6 (2): 213–215. doi:10.1021/ol0361599. PMID 14723531.
- ↑ Stahl, Glenn L.; Walter, Roderich; Smith, Clark W. (1978). "General Procedure for the Synthesis of Mono-N-Acylated 1,6-Diaminohexanes". J. Org. Chem. 43 (11): 2285–2286. doi:10.1021/jo00405a045.
- ↑ Prashad, Mahavir; Har, Denis; Hu, Bin; Kim, Hong-Yong; Girgis, Michael J.; Chaudhary, Apurva; Repič, Oljan; Blacklock, Thomas J. et al. (2004). "Process Development of a Large-Scale Synthesis of TKA731: A Tachykinin Receptor Antagonist". Org. Process Res. Dev. 8 (3): 330–340. doi:10.1021/op0341824.
- ↑ "Boc Deprotection - TFA". http://commonorganicchemistry.com/Rxn_Pages/Boc_Protection/Boc_Protection_TFA.htm.
 |
 KSF
KSF