Transition metal complexes of aldehydes and ketones
Topic: Chemistry
 From HandWiki - Reading time: 6 min
From HandWiki - Reading time: 6 min
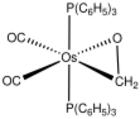
Transition metal complexes of aldehydes and ketones describes coordination complexes with aldehyde (RCHO) and ketone (R
2CO) ligands. Because aldehydes and ketones are common, the area is of fundamental interest. Some reactions that are useful in organic chemistry involve such complexes.
Structure and bonding
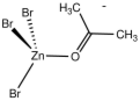
3(η1−acetone)]−.
In monometallic complexes, aldehydes and ketones can bind to metals in either of two modes, η1-O-bonded and η2-C,O-bonded. These bonding modes are sometimes referred to sigma- and pi-bonded. These forms may sometimes interconvert.
The sigma bonding mode is more common for higher valence, Lewis-acidic metal centers (e.g., Zn2+).[2] The pi-bonded mode is observed for low valence, electron-rich metal centers (e.g., Fe(0) and Os(0)).[3]
For the purpose of electron-counting, O-bonded ligands count as 2-electron "L ligands": they are Lewis bases. η2-C,O ligands are described as analogues of alkene ligands, i.e. the Dewar-Chatt-Duncanson model.[4]
η2-C,O ketones and aldehydes can function as bridging ligands, utilizing a lone pair of electrons on oxygen. One such complex is [(C
5H
5)
2Zr(CH
2O)]
3, which features a Zr
3O
3 ring.[5]
Formaldehyde complexes
Formaldehyde, being the simplest organic carbonyl and being an important industrial chemical, holds a special position as a ligand. Commonly it binds as η2-CH2O, i.e. "side-on bonded", comparable to ethylene. The first example was Os(η2-CH2O)(CO)2(PPh3)2 (Ph = phenyl, C6H5).[6] By virtue of the lone pairs of electrons on oxygen, M(η2-CH2O) entity can bridge to other metals.
Complexes are also known for vanadium,[7] rhenium,[8] zirconium (Zr),[9] ruthenium (Ru),[10] and niobium (Nb).[11]
Synthesis
Usually formaldehyde complexes are prepared by treating low-valence metal complexes with formaldehyde or one of its oligomers such as paraformaldehyde:
- Os(CO)2(PPh3)3 + CH2O → Os(η2-CH2O)(CO)2(PPh3)2 + PPh3
More exotic routes have been demonstrated such as the addition of CO to metal hydride complexes.[12] Such reactions are proposed to resemble steps in Fischer-Tropsch hydrogenation of CO.[13]
The complex W(PMe3)4(η2-CH2O)H2results from the addition of methanol to W(PMe3)4(η2-CH2PMe2)H.
Compounds in which metals replace the aldehydic hydrogen, instead of enolizing the carbonyl, are transition metal acyl complexes.
Reactions
The reactivity of metal-formaldehyde complexes has been well investigated. W(PMe3)4(η2-CH2O)H2 can be hydrogenated to give W(PMe3)4(CH3O)H3 and then methanol to generate W(PMe3)4H4.[14]
In some cases, alkenes can inert into the M-C bond of the M(η2-CH2O) entity.[15]

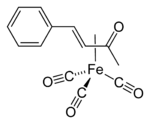
Some η2-aldehyde complexes insert alkenes to give five-membered metallacycles.[16]
η1-Complexes of alpha-beta unsaturated carbonyls exhibit enhanced reactivity toward dienes. This interaction is the basis of Lewis-acid catalyzed Diels-Alder reactions.
The hydroformylation of formaldehyde gives glycolaldehyde:[17]
- CH
2O + H
2 + CO → HOCH
2CHO
References
- ↑ Brown, Kevin L.; Clark, George R.; Headford, Christine E. L.; Marsden, Karen; Roper, Warren R. (1979). "Organometallic models for possible Fischer-Tropsch intermediates. Synthesis, structure, and reactions of a formaldehyde complex of osmium". Journal of the American Chemical Society 101 (2): 503–505. doi:10.1021/ja00496a050. Bibcode: 1979JAChS.101..503B.
- ↑ Andreev, V. P.; Sobolev, P. S.; Tafeenko, V. A. (2017). "Coordination of Zinc Tetraphenylporphyrin with Pyridine Derivatives in Chloroform Solution and in the Solid Phase". Russian Journal of General Chemistry 87 (7): 1572–1579. doi:10.1134/S1070363217070210.
- ↑ Berke, Heinz; Huttner, Gottfried; Weiler, Gertrud; Zsolnai, Laszlo (1981). "Struktur und Reaktivität eines Formaldehydeisen-Komplexes". Journal of Organometallic Chemistry 219 (3): 353–362. doi:10.1016/S0022-328X(00)90020-2.
- ↑ Huang, Yo Hsin; Gladysz, J. A. (1988). "Aldehyde and Ketone Ligands in organometallic complexes and catalysis". Journal of Chemical Education 65 (4): 298. doi:10.1021/ed065p298. Bibcode: 1988JChEd..65..298H.
- ↑ Kropp, Kurt; Skibbe, Volker; Erker, Gerhard; Krueger, Carl (1983). "Fischer-Tropsch intermediates: Tris[(.eta.2-formaldehyde)zirconocene] from the carbonylation of a zirconium hydride". Journal of the American Chemical Society 105 (10): 3353–3354. doi:10.1021/ja00348a075. Bibcode: 1983JAChS.105.3353K.
- ↑ Clark, G.R.; Headford, C.E.L.; Marsden, K.; Roper, W.R. (June 1982). "Synthesis, structure and reactions of a dihapto-formaldehyde complex, Os(η2-CH2O)(CO)2(PPh3)2". Journal of Organometallic Chemistry 231 (4): 335–360. doi:10.1016/s0022-328x(00)81212-7. ISSN 0022-328X. https://doi.org/10.1016/S0022-328X(00)81212-7.
- ↑ Gambarotta, S.; Floriani, C.; Chiesi-Villa, A.; Guastini, C. (April 1982). "Metal-formaldehyde chemistry: coordination, disproportionation and Lewis acid-promoted transformation to oxymethylene derivatives" (in en). Journal of the American Chemical Society 104 (7): 2019–2020. doi:10.1021/ja00371a038. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja00371a038.
- ↑ Buhro, William E.; Patton, Alan T.; Strouse, Charles E.; Gladysz, J. A.; McCormick, Fred B.; Etter, Margaret C. (February 1983). "Syntheses, chemical properties, and x-ray crystal structures of rhenium formaldehyde and thioformaldehyde complexes" (in en). Journal of the American Chemical Society 105 (4): 1056–1058. doi:10.1021/ja00342a070. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja00342a070.
- ↑ Kropp, Kurt; Skibbe, Volker; Erker, Gerhard; Krueger, Carl (May 1983). "Fischer-Tropsch intermediates: tris[(.eta.2-formaldehyde)zirconocene from the carbonylation of a zirconium hydride"] (in en). Journal of the American Chemical Society 105 (10): 3353–3354. doi:10.1021/ja00348a075. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja00348a075.
- ↑ Chaudret, Bruno N.; Cole-Hamilton, David J.; Nohr, Ronald S.; Wilkinson, Geoffrey (1977-01-01). "The reactions of chlorohydrido- and dichloro-tris(triphenylphosphine)ruthenium(II) with alkali hydroxides and alkoxides. Hydridohydroxobis(triphenylphosphine)ruthenium(II) monosolvates, their reactions and related compounds" (in en). Journal of the Chemical Society, Dalton Transactions (16): 1546–1557. doi:10.1039/DT9770001546. ISSN 1364-5447. https://pubs.rsc.org/en/content/articlelanding/1977/dt/dt9770001546.
- ↑ Wolczanski, Peter T.; Threlkel, Richard S.; Bercaw, John E. (January 1979). "Reduction of coordinated carbon monoxide to "zirconoxy" carbenes with permethylzirconocene dihydride" (in en). Journal of the American Chemical Society 101 (1): 218–220. doi:10.1021/ja00495a037. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja00495a037.
- ↑ Gambarotta, S.; Floriani, C.; Chiesi-Villa, A.; Guastini, C. (March 1983). "Genesis, bonding mode and reaction with carbon monoxide of an oxymethylene unit bridging two metal atoms" (in en). Journal of the American Chemical Society 105 (6): 1690–1691. doi:10.1021/ja00344a066. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja00344a066.
- ↑ Fachinetti, Giuseppe; Floriani, Carlo; Pucci, Sergio (1978-01-01). "Stoicheiometric reduction of CO and CO2 to methanol: evidence for carbon monoxide insertion into zirconium–hydrogen bond" (in en). Journal of the Chemical Society, Chemical Communications (6): 269–270. doi:10.1039/C39780000269. ISSN 0022-4936. https://pubs.rsc.org/en/content/articlelanding/1978/c3/c39780000269.
- ↑ Green, Malcolm L. H.; Parkin, Gerard; Moynihan, Kelly J.; Prout, Keith (1984-01-01). "Formation of an η2-formaldehyde compound from methanol and its hydrogenation giving methanol" (in en). Journal of the Chemical Society, Chemical Communications (22): 1540. doi:10.1039/C39840001540. ISSN 0022-4936. https://pubs.rsc.org/en/content/articlelanding/1984/c3/c39840001540.
- ↑ Green, Malcolm L. H.; Parkin, Gerard (1986-01-01). "Ethylene insertion into the W–C bond of the η2-formaldehyde ligand system of W(PMe3)4(η2-CH2O)H2 giving the oxometallacyclopentane derivative W(OCH2CH2CH2)(PMe3)2(C2H4)2 and related studies" (in en). Journal of the Chemical Society, Chemical Communications (1): 90–91. doi:10.1039/C39860000090. ISSN 0022-4936. https://pubs.rsc.org/en/content/articlelanding/1986/c3/c39860000090.
- ↑ Hoshimoto, Yoichi; Ohashi, Masato; Ogoshi, Sensuke (2015). "Catalytic Transformation of Aldehydes with Nickel Complexes through η2-Coordination and Oxidative Cyclization". Accounts of Chemical Research 48 (6): 1746–1755. doi:10.1021/acs.accounts.5b00061. PMID 25955708.
- ↑ Wei, Jie; Li, Maoshuai; Wang, Meiyan; Feng, Shixiang; Dai, Weikang; Yang, Qi; Feng, Yi; Yang, Wanxin et al. (2021). "Review Hydroformylation of formaldehyde to glycolaldehyde: An alternative synthetic route for ethylene glycol". Chinese Journal of Chemical Engineering 35: 3–16. doi:10.1016/j.cjche.2020.08.056.
 |
 KSF
KSF