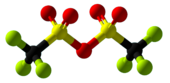
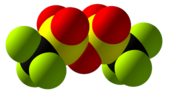
Trifluoromethanesulfonic anhydride
Topic: Chemistry
 From HandWiki - Reading time: 3 min
From HandWiki - Reading time: 3 min
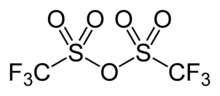
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Trifluoromethanesulfonic anhydride | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C2F6O5S2 | |||
| Molar mass | 282.13 g·mol−1 | ||
| Appearance | colourless liquid | ||
| Density | 1.6770 g/mL | ||
| Boiling point | 82[1] °C (180 °F; 355 K) | ||
| Reacts to form Triflic acid | |||
| Hazards | |||
| Safety data sheet | Fisher MSDS | ||
| GHS pictograms |   
| ||
| GHS Signal word | Danger | ||
| H272, H302, H314, H335 | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Trifluoromethanesulfonic anhydride, also known as triflic anhydride, is the chemical compound with the formula (CF3SO2)2O. It is the acid anhydride derived from triflic acid. This compound is a strong electrophile, useful for introducing the triflyl group, CF3SO2. Abbreviated Tf2O, triflic anhydride is the acid anhydride of the superacid triflic acid, CF3SO2OH.[2][3]
Preparation and uses
Triflic anhydride is prepared by dehydration of triflic acid using P4O10.[2]
Triflic anhydride is useful for converting ketones into enol triflates.[4]
In a representative application, is used to convert an imine into a NTf group.[5] It will convert phenols into a triflic ester, which enables cleavage of the C-O bond.[6][7]
Assay
The typical impurity in triflic anhydride is triflic acid, which is also a colorless liquid. Samples of triflic anhydride can be assayed by 19F NMR spectroscopy: −72.6 ppm[8] vs. −77.3 for TfOH (std CFCl3).
Safety
It is an aggressive electrophile and readily hydrolyzes to the strong acid triflic acid. It is very harmful to skin and eyes.[9]
See also
References
- ↑ Bloodworth, A.J.; Curtis, Richard J.; Spencer, Michael D.; Tallant, Neil A. (March 1993). "Oxymetallation. Part 24. Preparation of cyclic peroxides by cycloperoxymercuriation of unsaturated hydroperoxides.". Tetrahedron 49 (13): 2729–2750. doi:10.1016/S0040-4020(01)86350-X.
- ↑ 2.0 2.1 Martínez, A. G.; Subramanian, L. R.; Hanack, M. (2016). "Trifluoromethanesulfonic Anhydride". Encyclopedia of Reagents for Organic Synthesis: 1–17. doi:10.1002/047084289X.rt247.pub3. ISBN 9780470842898.
- ↑ Baraznenok, Ivan L.; Nenajdenko, Valentine G.; Balenkova, Elizabeth S. (May 2000). "Chemical Transformations Induced by Triflic Anhydride". Tetrahedron 56 (20): 3077–3119. doi:10.1016/S0040-4020(00)00093-4.
- ↑ Cacchi, Sandro; Morera, Enrico; Ortar, Giorgio (2011). "Discussion Addendum for: Palladium-Catalyzed Reduction of Vinyl Trifluoromethanesulfonates to Alkenes: Cholesta-3,5-diene". Organic Syntheses 88: 260. doi:10.15227/orgsyn.088.0260.
- ↑ Baker, T. J.; Tomioka, M.; Goodman, M. (2002). "Preparation and Use of N,N'-Di-BOC-N''-Triflylguanidine". Organic Syntheses 78: 91. doi:10.15227/orgsyn.078.0091.
- ↑ McWilliams, J. C.; Fleitz, F. J.; Zheng, N.; Armstrong, III, J. D.. "Preparation of n-Butyl 4-Chlorophenyl Sulfide". Organic Syntheses 79: 43. doi:10.15227/orgsyn.079.0043.
- ↑ Cai, D.; Payack, J. F.; Bender, D. R.; Hughes, D. L.; Verhoeven, T. R.; Reider, P. J. (1999). "(R)-(+)- and (S)-(−)-2,2'-Bis(Diphenylphosphino)-1,1'-Binaphthyl (BINAP)". Organic Syntheses 76: 6. doi:10.15227/orgsyn.076.0006.
- ↑ Dell'Amico, Daniela Belli; Boschi, Daniele; Calderazzo, Fausto; Labella, Luca; Marchetti, Fabio (28 February 2002). "Synthesis, and crystal and molecular structures of the triflato and trifluoroacetato complexes of zinc, Zn(O3SCF3)2(DME)2 and [Zn(O2CCF3)2(DME)n"]. Inorganica Chimica Acta 330 (1): 149–154. doi:10.1016/S0020-1693(01)00739-3. http://144.206.159.178/ft/458/52133/914984.pdf.
- ↑ "MSDS - 176176". https://www.sigmaaldrich.com/MSDS/MSDS/DisplayMSDSPage.do?country=US&language=en&productNumber=176176&brand=ALDRICH&PageToGoToURL=https://www.sigmaaldrich.com/catalog/product/aldrich/176176?lang=en.
 |
 KSF
KSF