Vanadium
Topic: Chemistry
 From HandWiki - Reading time: 30 min
From HandWiki - Reading time: 30 min
Vanadium is a chemical element; it has symbol V and atomic number 23. It is a hard, silvery-grey, malleable transition metal. The elemental metal is rarely found in nature, but once isolated artificially, the formation of an oxide layer (passivation) somewhat stabilizes the free metal against further oxidation.
Spanish-Mexican scientist Andrés Manuel del Río discovered compounds of vanadium in 1801 by analyzing a new lead-bearing mineral he called "brown lead". Though he initially presumed its qualities were due to the presence of a new element, he was later erroneously convinced by French chemist Hippolyte Victor Collet-Descotils that the element was just chromium. Then in 1830, Nils Gabriel Sefström generated chlorides of vanadium, thus proving there was a new element, and named it "vanadium" after the Scandinavian goddess of beauty and fertility, Vanadís (Freyja). The name was based on the wide range of colors found in vanadium compounds. Del Río's lead mineral was ultimately named vanadinite for its vanadium content. In 1867, Henry Enfield Roscoe obtained the pure element.
Vanadium occurs naturally in about 65 minerals and fossil fuel deposits. It is produced in China and Russia from steel smelter slag. Other countries produce it either from magnetite directly, flue dust of heavy oil, or as a byproduct of uranium mining. It is mainly used to produce specialty steel alloys such as high-speed tool steels, and some aluminium alloys. The most important industrial vanadium compound, vanadium pentoxide, is used as a catalyst for the production of sulfuric acid. The vanadium redox battery for energy storage may be an important application in the future.
Large amounts of vanadium ions are found in a few organisms, possibly as a toxin. The oxide and some other salts of vanadium have moderate toxicity. Particularly in the ocean, vanadium is used by some life forms as an active center of enzymes, such as the vanadium bromoperoxidase of some ocean algae.
History
Vanadium was discovered in Mexico in 1801 by the Spanish mineralogist Andrés Manuel del Río. Del Río extracted the element from a sample of Mexican "brown lead" ore, later named vanadinite. He found that its salts exhibit a wide variety of colors, and as a result, he named the element panchromium (Greek: παγχρώμιο "all colors"). Later, del Río renamed the element erythronium (Greek: ερυθρός "red") because most of the salts turned red upon heating.[1][2] In 1805, French chemist Hippolyte Victor Collet-Descotils, backed by del Río's friend Baron Alexander von Humboldt, incorrectly declared that del Río's new element was an impure sample of chromium. Del Río accepted Collet-Descotils' statement and retracted his claim.[3]
In 1831 Swedish chemist Nils Gabriel Sefström rediscovered the element in a new oxide he found while working with iron ores. Later that year, Friedrich Wöhler confirmed that this element was identical to that found by del Río and hence confirmed del Río's earlier work.[4][5] Sefström chose a name beginning with V, which had not yet been assigned to any element. He called the element vanadium after Old Norse Vanadís (another name for the Norse Vanir goddess Freyja, whose attributes include beauty and fertility), because of the many beautifully colored chemical compounds it produces.[4] On learning of Wöhler's findings, del Río began to passionately argue that his old claim be recognized, but the element kept the name vanadium.[6] In 1831, the geologist George William Featherstonhaugh suggested that vanadium should be renamed "rionium" after del Río, but this suggestion was not followed.[7]

As vanadium is usually found combined with other elements, the isolation of vanadium metal was difficult.[8] In 1831, Berzelius reported the production of the metal, but Henry Enfield Roscoe showed that Berzelius had produced the nitride, vanadium nitride (VN). Roscoe eventually produced the metal in 1867 by reduction of vanadium(II) chloride, VCl2, with hydrogen.[9] In 1927, pure vanadium was produced by reducing vanadium pentoxide with calcium.[10]
The first large-scale industrial use of vanadium was in the steel alloy chassis of the Ford Model T, inspired by French race cars. Vanadium steel allowed reduced weight while increasing tensile strength (c. 1905).[11] For the first decade of the 20th century, most vanadium ore were mined by the American Vanadium Company from the Minas Ragra in Peru. Later, the demand for uranium rose, leading to increased mining of that metal's ores. One major uranium ore was carnotite, which also contains vanadium. Thus, vanadium became available as a by-product of uranium production. Eventually, uranium mining began to supply a large share of the demand for vanadium.[12][13]
In 1911, German chemist Martin Henze discovered vanadium in the hemovanadin proteins found in blood cells (or coelomic cells) of Ascidiacea (sea squirts).[14][15]
Characteristics

Vanadium is an average-hard, ductile, steel-blue metal. Vanadium is usually described as "soft", because it is ductile, malleable, and not brittle.[16][17] Vanadium is harder than most metals and steels (see Hardnesses of the elements (data page) and iron). It has good resistance to corrosion and it is stable against alkalis and sulfuric and hydrochloric acids.[18] It is oxidized in air at about 933 K (660 °C, 1220 °F), although an oxide passivation layer forms even at room temperature.[19] It also reacts with hydrogen peroxide.
Isotopes
Naturally occurring vanadium is composed of one stable isotope, 51V, and one radioactive isotope, 50V. The latter has a half-life of 2.71×1017 years and a natural abundance of 0.25%. 51V has a nuclear spin of 7⁄2 and can be used for NMR spectroscopy.[20] Twenty-five artificial radioisotopes have been characterized, ranging in mass number from 42 to 68. The most stable of these isotopes are 49V with a half-life of 330 days, and 48V with a half-life of 15.97 days. The remaining radioactive isotopes have half-lives shorter than an hour, most below 10 seconds.[21]
Electron capture is the main decay mode for isotopes lighter than 51V. For the heavier ones, the most common mode is beta decay.[21] The electron capture reactions lead to the formation of element 22 (titanium) isotopes, while beta decay leads to element 24 (chromium) isotopes.
Compounds

The chemistry of vanadium is noteworthy for the accessibility of the four adjacent oxidation states 2–5. In an aqueous solution, vanadium forms metal aquo complexes of which the colors are lilac [V(H2O)6]2+, green [V(H2O)6]3+, blue [VO(H2O)5]2+, yellow-orange oxides [VO(H2O)5]3+, the formula for which depends on pH. Vanadium(II) compounds are reducing agents, and vanadium(V) compounds are oxidizing agents. Vanadium(IV) compounds often exist as vanadyl derivatives, which contain the VO2+ center.[18]
Ammonium vanadate(V) (NH4VO3) can be successively reduced with elemental zinc to obtain the different colors of vanadium in these four oxidation states. Lower oxidation states occur in compounds such as V(CO)6, [V(CO)6]− and substituted derivatives.[18]
Vanadium pentoxide is a commercially important catalyst for the production of sulfuric acid, a reaction that exploits the ability of vanadium oxides to undergo redox reactions.[18]
The vanadium redox battery utilizes all four oxidation states: one electrode uses the +5/+4 couple and the other uses the +3/+2 couple. Conversion of these oxidation states is illustrated by the reduction of a strongly acidic solution of a vanadium(V) compound with zinc dust or amalgam. The initial yellow color characteristic of the pervanadyl ion [VO2(H2O)4]+ is replaced by the blue color of [VO(H2O)5]2+, followed by the green color of [V(H2O)6]3+ and then the violet color of [V(H2O)6]2+.[18] Another potential vanadium battery based on VB2 uses multiple oxidation state to allow for 11 electrons to be released per VB2, giving it higher energy capacity by order of compared to Li-ion and gasoline per unit volume.[22] VB2 batteries can be further enhanced as air batteries, allowing for even higher energy density and lower weight than lithium battery or gasoline, even though recharging remains a challenge.[22]
Oxyanions
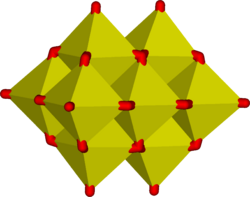
In an aqueous solution, vanadium(V) forms an extensive family of oxyanions as established by 51V NMR spectroscopy.[20] The interrelationships in this family are described by the predominance diagram, which shows at least 11 species, depending on pH and concentration.[23] The tetrahedral orthovanadate ion, VO3−4, is the principal species present at pH 12–14. Similar in size and charge to phosphorus(V), vanadium(V) also parallels its chemistry and crystallography. Orthovanadate VO3−4 is used in protein crystallography[24] to study the biochemistry of phosphate.[25] Besides that, this anion also has been shown to interact with the activity of some specific enzymes.[26][27] The tetrathiovanadate [VS4]3− is analogous to the orthovanadate ion.[28]
At lower pH values, the monomer [HVO4]2− and dimer [V2O7]4− are formed, with the monomer predominant at a vanadium concentration of less than c. 10−2M (pV > 2, where pV is equal to the minus value of the logarithm of the total vanadium concentration/M). The formation of the divanadate ion is analogous to the formation of the dichromate ion.[29][30] As the pH is reduced, further protonation and condensation to polyvanadates occur: at pH 4–6 [H2VO4]− is predominant at pV greater than ca. 4, while at higher concentrations trimers and tetramers are formed.[31] Between pH 2–4 decavanadate predominates, its formation from orthovanadate is represented by this condensation reaction:
- 10 [VO4]3− + 24 H+ → [V10O28]6− + 12 H2O

In decavanadate, each V(V) center is surrounded by six oxide ligands.[18] Vanadic acid, H3VO4, exists only at very low concentrations because protonation of the tetrahedral species [H2VO4]− results in the preferential formation of the octahedral [VO2(H2O)4]+ species.[32] In strongly acidic solutions, pH < 2, [VO2(H2O)4]+ is the predominant species, while the oxide V2O5 precipitates from solution at high concentrations. The oxide is formally the acid anhydride of vanadic acid. The structures of many vanadate compounds have been determined by X-ray crystallography.

Vanadium(V) forms various peroxo complexes, most notably in the active site of the vanadium-containing bromoperoxidase enzymes. The species VO(O2)(H2O)4+ is stable in acidic solutions. In alkaline solutions, species with 2, 3 and 4 peroxide groups are known; the last forms violet salts with the formula M3V(O2)4 nH2O (M= Li, Na, etc.), in which the vanadium has an 8-coordinate dodecahedral structure.[34][35]
Halide derivatives
Twelve binary halides, compounds with the formula VXn (n=2..5), are known.[36] VI4, VCl5, VBr5, and VI5 do not exist or are extremely unstable. In combination with other reagents, VCl4 is used as a catalyst for the polymerization of dienes. Like all binary halides, those of vanadium are Lewis acidic, especially those of V(IV) and V(V).[36] Many of the halides form octahedral complexes with the formula VXnL6−n (X= halide; L= other ligand).
Many vanadium oxyhalides (formula VOmXn) are known.[37] The oxytrichloride and oxytrifluoride (VOCl3 and VOF3) are the most widely studied. Akin to POCl3, they are volatile,[38] adopt tetrahedral structures in the gas phase, and are Lewis acidic.[39]
Coordination compounds
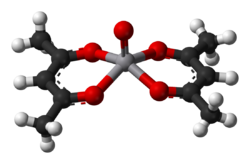
Complexes of vanadium(II) and (III) are reducing, while those of V(IV) and V(V) are oxidants. The vanadium ion is rather large and some complexes achieve coordination numbers greater than 6, as is the case in [V(CN)7]4−. Oxovanadium(V) also forms 7 coordinate coordination complexes with tetradentate ligands and peroxides and these complexes are used for oxidative brominations and thioether oxidations. The coordination chemistry of V4+ is dominated by the vanadyl center, VO2+, which binds four other ligands strongly and one weakly (the one trans to the vanadyl center). An example is vanadyl acetylacetonate (V(O)(O2C5H7)2). In this complex, the vanadium is 5-coordinate, distorted square pyramidal, meaning that a sixth ligand, such as pyridine, may be attached, though the association constant of this process is small. Many 5-coordinate vanadyl complexes have a trigonal bipyramidal geometry, such as VOCl2(NMe3)2.[40] The coordination chemistry of V5+ is dominated by the relatively stable dioxovanadium coordination complexes[41] which are often formed by aerial oxidation of the vanadium(IV) precursors indicating the stability of the +5 oxidation state and ease of interconversion between the +4 and +5 states.[42]
Organometallic compounds
The organometallic chemistry of vanadium is well–developed. Vanadocene dichloride is a versatile starting reagent and has applications in organic chemistry.[43] Vanadium carbonyl, V(CO)6, is a rare example of a paramagnetic metal carbonyl. Reduction yields V(CO)−6 (isoelectronic with Cr(CO)6), which may be further reduced with sodium in liquid ammonia to yield V(CO)3−5 (isoelectronic with Fe(CO)5).[44][45]
Occurrence

Metallic vanadium is rare in nature (known as native vanadium),[46][47] having been found among fumaroles of the Colima Volcano, but vanadium compounds occur naturally in about 65 different minerals.
Vanadium began to be used in the manufacture of special steels in 1896. At that time, very few deposits of vanadium ores were known. Between 1899 and 1906, the main deposits exploited were the mines of Santa Marta de los Barros (Badajoz), Spain. Vanadinite was extracted from these mines.[48] At the beginning of the 20th century, a large deposit of vanadium ore was discovered near Junín, Cerro de Pasco, Peru (now the Minas Ragra vanadium mine).[49][50][51] For several years this patrónite (VS4)[52] deposit was an economically significant source for vanadium ore. In 1920 roughly two-thirds of the worldwide production was supplied by the mine in Peru.[53] With the production of uranium in the 1910s and 1920s from carnotite (K2(UO2)2(VO4)2·3H2O) vanadium became available as a side product of uranium production. Vanadinite (Pb5(VO4)3Cl) and other vanadium bearing minerals are only mined in exceptional cases. With the rising demand, much of the world's vanadium production is now sourced from vanadium-bearing magnetite found in ultramafic gabbro bodies. If this titanomagnetite is used to produce iron, most of the vanadium goes to the slag and is extracted from it.[54][51]
Vanadium is mined mostly in China, South Africa and eastern Russia. In 2022 these three countries mined more than 96% of the 100,000 tons of produced vanadium, with China providing 70%.[55]
Fumaroles of Colima are known of being vanadium-rich, depositing other vanadium minerals, that include shcherbinaite (V2O5) and colimaite (K3VS4).[56][57][58]
Vanadium is also present in bauxite and deposits of crude oil, coal, oil shale, and tar sands. In crude oil, concentrations up to 1200 ppm have been reported. When such oil products are burned, traces of vanadium may cause corrosion in engines and boilers.[59] An estimated 110,000 tons of vanadium per year are released into the atmosphere by burning fossil fuels.[60] Black shales are also a potential source of vanadium. During WWII some vanadium was extracted from alum shales in the south of Sweden.[61]
In the universe, the cosmic abundance of vanadium is 0.0001%, making the element nearly as common as copper or zinc.[62] Vanadium is the 19th most abundant element in the crust.[63] It is detected spectroscopically in light from the Sun and sometimes in the light from other stars.[64] The vanadyl ion is also abundant in seawater, having an average concentration of 30 nM (1.5 mg/m3).[62] Some mineral water springs also contain the ion in high concentrations. For example, springs near Mount Fuji contain as much as 54 μg per liter.[62]
Production


Vanadium metal is obtained by a multistep process that begins with roasting crushed ore with NaCl or Na2CO3 at about 850 °C to give sodium metavanadate (NaVO3). An aqueous extract of this solid is acidified to produce "red cake", a polyvanadate salt, which is reduced with calcium metal. As an alternative for small-scale production, vanadium pentoxide is reduced with hydrogen or magnesium. Many other methods are also used, in all of which vanadium is produced as a byproduct of other processes.[65] Purification of vanadium is possible by the crystal bar process developed by Anton Eduard van Arkel and Jan Hendrik de Boer in 1925. It involves the formation of the metal iodide, in this example vanadium(III) iodide, and the subsequent decomposition to yield pure metal:[66]
- 2 V + 3 I2 ⇌ 2 VI3

Most vanadium is used as a steel alloy called ferrovanadium. Ferrovanadium is produced directly by reducing a mixture of vanadium oxide, iron oxides and iron in an electric furnace. The vanadium ends up in pig iron produced from vanadium-bearing magnetite. Depending on the ore used, the slag contains up to 25% of vanadium.[65]
Applications

Alloys
Approximately 85% of the vanadium produced is used as ferrovanadium or as a steel additive.[65] The considerable increase of strength in steel containing small amounts of vanadium was discovered in the early 20th century. Vanadium forms stable nitrides and carbides, resulting in a significant increase in the strength of steel.[67] From that time on, vanadium steel was used for applications in axles, bicycle frames, crankshafts, gears, and other critical components. There are two groups of vanadium steel alloys. Vanadium high-carbon steel alloys contain 0.15–0.25% vanadium, and high-speed tool steels (HSS) have a vanadium content of 1–5%. For high-speed tool steels, a hardness above HRC 60 can be achieved. HSS steel is used in surgical instruments and tools.[68] Powder-metallurgic alloys contain up to 18% percent vanadium. The high content of vanadium carbides in those alloys increases wear resistance significantly. One application for those alloys is tools and knives.[69]
Vanadium stabilizes the beta form of titanium and increases the strength and temperature stability of titanium. Mixed with aluminium in titanium alloys, it is used in jet engines, high-speed airframes and dental implants. The most common alloy for seamless tubing is Titanium 3/2.5 containing 2.5% vanadium, the titanium alloy of choice in the aerospace, defense, and bicycle industries.[70] Another common alloy, primarily produced in sheets, is Titanium 6AL-4V, a titanium alloy with 6% aluminium and 4% vanadium.[71]
Several vanadium alloys show superconducting behavior. The first A15 phase superconductor was a vanadium compound, V3Si, which was discovered in 1952.[72] Vanadium-gallium tape is used in superconducting magnets (17.5 teslas or 175,000 gauss). The structure of the superconducting A15 phase of V3Ga is similar to that of the more common Nb3Sn and Nb3Ti.[73]
It has been found that a small amount, 40 to 270 ppm, of vanadium in Wootz steel significantly improved the strength of the product, and gave it distinctive patterning. The source of the vanadium in the original Wootz steel ingots remains unknown.[74]
Vanadium can be used as a substitute for molybdenum in armor steel, though the alloy produced is far more brittle and prone to spalling on non-penetrating impacts.[75] Nazi Germany was one of the most prominent users of such alloys, in armored vehicles like Tiger II or Jagdtiger.[76]
Catalysts

Vanadium compounds are used extensively as catalysts;[77] Vanadium pentoxide V2O5, is used as a catalyst in manufacturing sulfuric acid by the contact process[78] In this process sulfur dioxide (SO2) is oxidized to the trioxide (SO3):[18] In this redox reaction, sulfur is oxidized from +4 to +6, and vanadium is reduced from +5 to +4:
- V2O5 + SO2 → 2 VO2 + SO3
The catalyst is regenerated by oxidation with air:
- 4 VO2 + O2 → 2 V2O5
Similar oxidations are used in the production of maleic anhydride:
- C4H10 + 3.5 O2 → C4H2O3 + 4 H2O
Phthalic anhydride and several other bulk organic compounds are produced similarly. These green chemistry processes convert inexpensive feedstocks to highly functionalized, versatile intermediates.[79][80]
Vanadium is an important component of mixed metal oxide catalysts used in the oxidation of propane and propylene to acrolein, acrylic acid or the ammoxidation of propylene to acrylonitrile.[81]
Other uses
The vanadium redox battery, a type of flow battery, is an electrochemical cell consisting of aqueous vanadium ions in different oxidation states.[82][83] Batteries of this type were first proposed in the 1930s and developed commercially from the 1980s onwards. Cells use +5 and +2 formal oxidization state ions. Vanadium redox batteries are used commercially for grid energy storage.[84]
Vanadate can be used for protecting steel against rust and corrosion by conversion coating.[85] Vanadium foil is used in cladding titanium to steel because it is compatible with both iron and titanium.[86] The moderate thermal neutron-capture cross-section and the short half-life of the isotopes produced by neutron capture makes vanadium a suitable material for the inner structure of a fusion reactor.[87][88]
Vanadium can be added in small quantities < 5% to LFP battery cathodes to increase ionic conductivity.[89]
Proposed
Lithium vanadium oxide has been proposed for use as a high-energy-density anode for lithium-ion batteries, at 745 Wh/L when paired with a lithium cobalt oxide cathode.[90] Vanadium phosphates have been proposed as the cathode in the lithium vanadium phosphate battery, another type of lithium-ion battery.[91]
Sodium vanadium phosphate has also been proposed as an alternative to lithium-ion batteries. The new material is proposed by the Canepa Research Laboratory[92] at the University of Houston, who claim that since sodium is around 50 times cheaper than lithium, a sodium-ion alternative with similar energy density could lead to cheaper batteries with a simpler resource chain.
Biological role
Vanadium has a more significant role in marine environments than terrestrial ones.[93]


Vanadoenzymes
Several species of marine algae produce vanadium bromoperoxidase as well as the closely related chloroperoxidase (which may use a heme or vanadium cofactor) and iodoperoxidases. The bromoperoxidase produces an estimated 1–2 million tons of bromoform and 56,000 tons of bromomethane annually.[94] Most naturally occurring organobromine compounds are produced by this enzyme,[95] catalyzing the following reaction (R-H is hydrocarbon substrate):
A vanadium nitrogenase is used by some nitrogen-fixing micro-organisms, such as Azotobacter. In this role, vanadium serves in place of the more common molybdenum or iron, and gives the nitrogenase slightly different properties.[96]
Vanadium accumulation in tunicates
Vanadium is essential to tunicates, where it is stored in the highly acidified vacuoles of certain blood cell types, designated vanadocytes. Vanabins (vanadium-binding proteins) have been identified in the cytoplasm of such cells. The concentration of vanadium in the blood of ascidian tunicates is as much as ten million times higher [97][98] than the surrounding seawater, which normally contains 1 to 2 μg/L.[99][100] The function of this vanadium concentration system and these vanadium-bearing proteins is still unknown, but the vanadocytes are later deposited just under the outer surface of the tunic, where they may deter predation.[101]
Fungi
Amanita muscaria and related species of macrofungi accumulate vanadium (up to 500 mg/kg in dry weight). Vanadium is present in the coordination complex amavadin[102] in fungal fruit-bodies. The biological importance of the accumulation is unknown.[103][104] Toxic or peroxidase enzyme functions have been suggested.[105]
Mammals
Deficiencies in vanadium result in reduced growth in rats.[106] The U.S. Institute of Medicine has not confirmed that vanadium is an essential nutrient for humans, so neither a Recommended Dietary Intake nor an Adequate Intake have been established. Dietary intake is estimated at 6 to 18 μg/day, with less than 5% absorbed. The Tolerable Upper Intake Level (UL) of dietary vanadium, beyond which adverse effects may occur, is set at 1.8 mg/day.[107]
Research
Vanadyl sulfate as a dietary supplement has been researched as a means of increasing insulin sensitivity or otherwise improving glycemic control in people who are diabetic. Some of the trials had significant treatment effects but were deemed as being of poor study quality. The amounts of vanadium used in these trials (30 to 150 mg) far exceeded the safe upper limit.[108][109] The conclusion of the systemic review was "There is no rigorous evidence that oral vanadium supplementation improves glycaemic control in type 2 diabetes. The routine use of vanadium for this purpose cannot be recommended."[108]
In astrobiology, it has been suggested that discrete vanadium accumulations on Mars could be a potential microbial biosignature when used in conjunction with Raman spectroscopy and morphology.[110][111]
Safety
All vanadium compounds should be considered toxic.[112] Tetravalent VOSO4 has been reported to be at least 5 times more toxic than trivalent V2O3.[113] The US Occupational Safety and Health Administration (OSHA) has set an exposure limit of 0.05 mg/m3 for vanadium pentoxide dust and 0.1 mg/m3 for vanadium pentoxide fumes in workplace air for an 8-hour workday, 40-hour work week.[114] The US National Institute for Occupational Safety and Health (NIOSH) has recommended that 35 mg/m3 of vanadium be considered immediately dangerous to life and health, that is, likely to cause permanent health problems or death.[114] Vanadium has been identified as a carcinogenic component of PM2.5 pollution, explaining a proportion of cancers and death caused by it.[115]
Vanadium compounds are poorly absorbed through the gastrointestinal system. Inhalation of vanadium and vanadium compounds results primarily in adverse effects on the respiratory system.[116][117][118] Quantitative data are, however, insufficient to derive a subchronic or chronic inhalation reference dose. Other effects have been reported after oral or inhalation exposures on blood parameters,[119][120] liver,[121] neurological development,[122] and other organs[123] in rats.
There is little evidence that vanadium or vanadium compounds are reproductive toxins or teratogens. Vanadium pentoxide was reported to be carcinogenic in male rats and in male and female mice by inhalation in an NTP study,[117] although the interpretation of the results has been disputed a few years after the report.[124] The carcinogenicity of vanadium has not been determined by the United States Environmental Protection Agency.[125]
Vanadium traces in diesel fuels are the main fuel component in high temperature corrosion. During combustion, vanadium oxidizes and reacts with sodium and sulfur, yielding vanadate compounds with melting points as low as 530 °C (986 °F), which attack the passivation layer on steel and render it susceptible to corrosion. The solid vanadium compounds also abrade engine components.[126][127]
See also
- Chemistry:Flow battery – Type of electrochemical cell
- Engineering:Grid energy storage – Large scale electricity supply management
- Chemistry:Vanadium carbide – Extremely hard refractory ceramic material
- Chemistry:Vanadium redox battery – Type of rechargeable flow battery
- Chemistry:Vanadium tetrachloride – Chemical reagent used to produce other vanadium compounds
- Chemistry:Vanadium(V) oxide – Precursor to vanadium alloys and industrial catalyst
- Chemistry:International Vanadium Symposium – Biennial interdisciplinary event
- Biology:Vanadium cycle – Exchange of vanadium between continental crust and seawater
References
- ↑ "The Vivid Element Vanadium". 2023-11-29. https://www.labxchange.org/library/items/lb:LabXchange:24e9407f:html:1.
- ↑ Uribe Salas, J.A. (2020-11-17). "Historia del vanadio, 1801-1831. Disputa por la autoria del descubrimiento.". Asclepio 72 (2): 5. https://doi.org/10.3989/asclepio.2020.23.
- ↑ Cintas, Pedro (12 November 2004). "The Road to Chemical Names and Eponyms: Discovery, Priority, and Credit". Angewandte Chemie International Edition 43 (44): 5888–5894. doi:10.1002/anie.200330074. PMID 15376297. Bibcode: 2004ACIE...43.5888C.
- ↑ 4.0 4.1 Sefström, N. G. (1831). "Ueber das Vanadin, ein neues Metall, gefunden im Stangeneisen von Eckersholm, einer Eisenhütte, die ihr Erz von Taberg in Småland bezieht". Annalen der Physik und Chemie 97 (1): 43–49. doi:10.1002/andp.18310970103. Bibcode: 1831AnP....97...43S. https://zenodo.org/record/1423544. Retrieved 27 August 2019.
- ↑ Hijmans, Sarah (2020-09-21). "Andrés Manuel del Rio and the Two Discoveries of Vanadium" (in en). https://www.jargonium.com/post/andr%C3%A9s-manuel-del-rio-and-the-two-discoveries-of-vanadium.
- ↑ Marshall, James L.; Marshall, Virginia R. (2004). "Rediscovery of the Elements: The "Undiscovery" of Vanadium". The Hexagon. p. 45. https://digital.library.unt.edu/ark:/67531/metadc111200/m2/1/high_res_d/metadc111200.pdf.
- ↑ Featherstonhaugh, George William (1831). "New Metal, provisionally called Vanadium". The Monthly American Journal of Geology and Natural Science: 69. https://archive.org/stream/monthlyamericanj11831phil#page/68/mode/2up/search/rionium.
- ↑ Habashi, Fathi (January 2001). "Historical Introduction to Refractory Metals". Mineral Processing and Extractive Metallurgy Review 22 (1): 25–53. doi:10.1080/08827509808962488. Bibcode: 2001MPEMR..22...25H.
- ↑ Roscoe, Henry Enfield (31 December 1870). "XIX. Researches on vanadium". Proceedings of the Royal Society of London 18 (114–122): 37–42. doi:10.1098/rspl.1869.0012. https://zenodo.org/record/1432055. Retrieved 27 August 2019.
- ↑ Marden, J. W.; Rich, M. N. (July 1927). "Vanadium 1". Industrial & Engineering Chemistry 19 (7): 786–788. doi:10.1021/ie50211a012.
- ↑ Betz, Frederick (2003). Managing Technological Innovation: Competitive Advantage from Change. Wiley-IEEE. pp. 158–159. ISBN 978-0-471-22563-8. https://books.google.com/books?id=KnpGtu-R77UC&pg=PA158.
- ↑ Busch, Phillip Maxwell (1961). Vanadium: A Materials Survey. U.S. Department of the Interior, Bureau of Mines. p. 65. OCLC 934517147. http://digital.library.unt.edu/ark:/67531/metadc170746/. Retrieved 19 April 2023.
- ↑ Wise, James M. (May 2018). "Remarkable folded dacitic dikes at Mina Ragra, Peru". https://www.southamericatotheworld.com/remarkable-folded-dacitic-dikes-at-mina-ragra-peru/.
- ↑ Henze, M. (1911). "Untersuchungen über das Blut der Ascidien. I. Mitteilung". Z. Physiol. Chem. 72 (5–6): 494–50. doi:10.1515/bchm2.1911.72.5-6.494. https://books.google.com/books?id=x5g8AAAAIAAJ.
- ↑ Michibata, H.; Uyama, T.; Ueki, T.; Kanamori, K. (2002). "Vanadocytes, cells hold the key to resolving the highly selective accumulation and reduction of vanadium in ascidians". Microscopy Research and Technique 56 (6): 421–434. doi:10.1002/jemt.10042. PMID 11921344. http://ir.lib.hiroshima-u.ac.jp/files/public/0/22/20141016115442843522/MicroscopResTech_56_421-434_2002.pdf. Retrieved 27 August 2019.
- ↑ George F. Vander Voort (1984). Metallography, principles and practice. ASM International. pp. 137–. ISBN 978-0-87170-672-0. https://books.google.com/books?id=GRQC8zYqtBIC&pg=PA137. Retrieved 17 September 2011.
- ↑ Cardarelli, François (2008). Materials handbook: a concise desktop reference. Springer. pp. 338–. ISBN 978-1-84628-668-1. https://books.google.com/books?id=PvU-qbQJq7IC&pg=PA338. Retrieved 17 September 2011.
- ↑ 18.0 18.1 18.2 18.3 18.4 18.5 18.6 Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (1985). "Vanadium" (in de). Lehrbuch der Anorganischen Chemie (91–100 ed.). Walter de Gruyter. pp. 1071–1075. ISBN 978-3-11-007511-3.
- ↑ Klinser, Gregor; Zettl, Roman; Wilkening, Martin; Krenn, Heinz; Hanzu, Ilie; Würschum, Roland (2019). "Redox processes in sodium vanadium phosphate cathodes – insights from operando magnetometry" (in en). Physical Chemistry Chemical Physics 21 (36): 20151–20155. doi:10.1039/C9CP04045E. ISSN 1463-9076. PMID 31482877. Bibcode: 2019PCCP...2120151K.
- ↑ 20.0 20.1 Rehder, D.; Polenova, T.; Bühl, M. (2007). Vanadium-51 NMR. Annual Reports on NMR Spectroscopy. 62. pp. 49–114. doi:10.1016/S0066-4103(07)62002-X. ISBN 978-0-12-373919-3.
- ↑ 21.0 21.1 Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties". Chinese Physics C 45 (3). doi:10.1088/1674-1137/abddae. https://www-nds.iaea.org/amdc/ame2020/NUBASE2020.pdf.
- ↑ 22.0 22.1 Licht, Stuart; Wu, Huiming; Yu, Xingwen; Wang, Yufei (2008-07-11). "Renewable highest capacity VB2/air energy storage" (in en). Chemical Communications (28): 3257–3259. doi:10.1039/B807929C. ISSN 1364-548X. PMID 18622436. https://pubs.rsc.org/en/content/articlelanding/2008/cc/b807929c.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 984. ISBN 978-0-08-037941-8.
- ↑ Sinning, Irmgard; Hol, Wim G. J. (2004). "The power of vanadate in crystallographic investigations of phosphoryl transfer enzymes". FEBS Letters 577 (3): 315–21. doi:10.1016/j.febslet.2004.10.022. PMID 15556602. Bibcode: 2004FEBSL.577..315D.
- ↑ Seargeant, L E; Stinson, R A (1 July 1979). "Inhibition of human alkaline phosphatases by vanadate". Biochemical Journal 181 (1): 247–250. doi:10.1042/bj1810247. PMID 486156.
- ↑ Crans, Debbie C.; Simone, Carmen M. (9 July 1991). "Nonreductive interaction of vanadate with an enzyme containing a thiol group in the active site: glycerol-3-phosphate dehydrogenase". Biochemistry 30 (27): 6734–6741. doi:10.1021/bi00241a015. PMID 2065057.
- ↑ Karlish, S. J. D.; Beaugé, L. A.; Glynn, I. M. (November 1979). "Vanadate inhibits (Na+ + K+)ATPase by blocking a conformational change of the unphosphorylated form". Nature 282 (5736): 333–335. doi:10.1038/282333a0. PMID 228199. Bibcode: 1979Natur.282..333K.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 988. ISBN 978-0-08-037941-8.
- ↑ Crans, Debbie C. (18 December 2015). "Antidiabetic, Chemical, and Physical Properties of Organic Vanadates as Presumed Transition-State Inhibitors for Phosphatases". The Journal of Organic Chemistry 80 (24): 11899–11915. doi:10.1021/acs.joc.5b02229. PMID 26544762.
- ↑ Jung, Sabrina (2018). Speciation of molybdenum- and vanadium-based polyoxometalate species in aqueous medium and gas-phase and its consequences for M1 structured MoV oxide synthesis (Thesis). doi:10.14279/depositonce-7254.
- ↑ Cruywagen, J. J. (1999-01-01), Sykes, A. G., ed. (in en), Protonation, Oligomerization, and Condensation Reactions of Vanadate(V), Molybdate(vi), and Tungstate(vi), Advances in Inorganic Chemistry, 49, Academic Press, pp. 127–182, doi:10.1016/S0898-8838(08)60270-6, ISBN 978-0-12-023649-7, https://www.sciencedirect.com/science/article/pii/S0898883808602706, retrieved 2023-04-16
- ↑ Tracey, Alan S.; Willsky, Gail R.; Takeuchi, Esther S. (2007-03-19) (in en). Vanadium: Chemistry, Biochemistry, Pharmacology and Practical Applications. CRC Press. ISBN 978-1-4200-4614-4. https://books.google.com/books?id=vkMGP3PiuyYC&dq=+protonation+of+the+tetrahedral+species+%5BH2VO4%5D%E2%88%92+results+in+the+preferential+formation+of+the+octahedral+%5BVO2(H2O)4%5D++species&pg=PP1.
- ↑ Al-Kharafi, F.M.; Badawy, W.A. (January 1997). "Electrochemical behaviour of vanadium in aqueous solutions of different pH". Electrochimica Acta 42 (4): 579–586. doi:10.1016/S0013-4686(96)00202-2.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8., p994.
- ↑ Strukul, Giorgio (1992). Catalytic oxidations with hydrogen peroxide as oxidant. Springer. p. 128. ISBN 978-0-7923-1771-5. https://books.google.com/books?id=Lmt3x9CyfLgC&pg=PA128.
- ↑ 36.0 36.1 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 989. ISBN 978-0-08-037941-8.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 993. ISBN 978-0-08-037941-8.
- ↑ Flesch, Gerald D.; Svec, Harry J. (1 August 1975). "Thermochemistry of vanadium oxytrichloride and vanadium oxytrifluoride by mass spectrometry". Inorganic Chemistry 14 (8): 1817–1822. doi:10.1021/ic50150a015.
- ↑ Iqbal, Javed; Bhatia, Beena; Nayyar, Naresh K. (March 1994). "Transition Metal-Promoted Free-Radical Reactions in Organic Synthesis: The Formation of Carbon-Carbon Bonds". Chemical Reviews 94 (2): 519–564. doi:10.1021/cr00026a008.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 995. ISBN 978-0-08-037941-8.
- ↑ Geiser, Jan Nicholas (2019). Development of an improved state-of-charge sensor for the all-vanadium redox flow battery (Thesis). doi:10.22028/D291-29229.
- ↑ Nica, Simona; Rudolph, Manfred; Görls, Helmar; Plass, Winfried (April 2007). "Structural characterization and electrochemical behavior of oxovanadium(V) complexes with N-salicylidene hydrazides". Inorganica Chimica Acta 360 (5): 1743–1752. doi:10.1016/j.ica.2006.09.018.
- ↑ Wilkinson, G.; Birmingham, J. M. (September 1954). "Bis-cyclopentadienyl Compounds of Ti, Zr, V, Nb and Ta". Journal of the American Chemical Society 76 (17): 4281–4284. doi:10.1021/ja01646a008. Bibcode: 1954JAChS..76.4281W.
- ↑ Bellard, S.; Rubinson, K. A.; Sheldrick, G. M. (15 February 1979). "Crystal and molecular structure of vanadium hexacarbonyl". Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry 35 (2): 271–274. doi:10.1107/S0567740879003332. Bibcode: 1979AcCrB..35..271B.
- ↑ Elschenbroich, C.; Salzer A. (1992). Organometallics: A Concise Introduction. Wiley-VCH. ISBN 978-3-527-28165-7.
- ↑ Ostrooumov, M.; Taran, Y. (2015). "Discovery of Native Vanadium, a New Mineral from the Colima Volcano, State of Colima (Mexico)". Revista de la Sociedad Española de Mineralogía 20: 109–110. https://www.uhu.es/fexp/sem2015/arc/macla/macla_20_109-110.pdf. Retrieved 7 February 2023.
- ↑ "Vanadium: Vanadium mineral information and data". https://www.mindat.org/min-43604.html.
- ↑ Calvo Rebollar, Miguel (2019) (in es). Construyendo la Tabla Periódica. Zaragoza, Spain: Prames. pp. 161–165. ISBN 978-84-8321-908-9.
- ↑ Hillebrand, W. F. (1907). "The Vanadium Sulphide, Patronite, and ITS Mineral Associates from Minasragra, Peru". Journal of the American Chemical Society 29 (7): 1019–1029. doi:10.1021/ja01961a006. Bibcode: 1907JAChS..29.1019H. https://zenodo.org/record/1450154. Retrieved 6 September 2020.
- ↑ Hewett, F. (1906). "A New Occurrence of Vanadium in Peru". The Engineering and Mining Journal 82 (9): 385.
- ↑ 51.0 51.1 Steinberg, W.S.; Geyser, W.; Nell, J. (2011). "The history and development of the pyrometallurgical processes at Evraz Highveld Steel & Vanadium". The Journal of the Southern African Institute of Mining and Metallurgy 111: 705–710. http://www.scielo.org.za/pdf/jsaimm/v111n10/v111n10a09.pdf. Retrieved 17 December 2018.
- ↑ "mineralogical data about Patrónite". mindata.org. https://www.mindat.org/min-3131.html.
- ↑ Allen, M. A.; Butler, G. M. (1921). "Vanadium". University of Arizona. https://repository.arizona.edu/bitstream/handle/10150/630042/b-115_vanadium.pdf. Retrieved 20 January 2020.
- ↑ Hukkanen, E.; Walden, H. (1985). "The production of vanadium and steel from titanomagnetites". International Journal of Mineral Processing 15 (1–2): 89–102. doi:10.1016/0301-7516(85)90026-2. Bibcode: 1985IJMP...15...89H.
- ↑ Polyak, Désirée E.. "Mineral Commodity Summaries 2023: Vanadium". United States Geological Survey. https://pubs.usgs.gov/periodicals/mcs2023/mcs2023-vanadium.pdf.
- ↑ Ostrooumov, M., and Taran, Y., 2015. Discovery of Native Vanadium, a New Mineral from the Colima Volcano, State of Colima (Mexico). Revista de la Sociedad Española de Mineralogía 20, 109-110
- ↑ "Vanadium: Vaandium mineral information and data". http://www.mindat.org/min-43604.html. Retrieved 2016-03-02.
- ↑ "Colima volcano (Volcan de Fuego; Volcan de Colima), Colima volcanic complex, Jalisco, Mexico". http://www.mindat.org/min-43604.html. Retrieved 2016-03-02.
- ↑ Pearson, C. D.; Green, J. B. (1 May 1993). "Vanadium and nickel complexes in petroleum resid acid, base, and neutral fractions". Energy & Fuels 7 (3): 338–346. doi:10.1021/ef00039a001. Bibcode: 1993EnFue...7..338P. https://digital.library.unt.edu/ark:/67531/metadc1198139/. Retrieved 10 August 2018.
- ↑ Anke, Manfred (2004). "Vanadium: An element both essential and toxic to plants, animals and humans?". Anales de la Real Academia Nacional de Farmacia 70 (4): 961–999. https://analesranf.com/wp-content/uploads/2004/70_04/7004_06.pdf. Retrieved 19 April 2023.
- ↑ Dyni, John R. (2006). "Geology and resources of some world oil-shale deposits". Scientific Investigations Report. p. 22. doi:10.3133/sir29955294.
- ↑ 62.0 62.1 62.2 Rehder, Dieter (2008). Bioinorganic Vanadium Chemistry. Inorganic Chemistry (1st ed.). Hamburg, Germany: John Wiley & Sons, Ltd. pp. 5 & 9–10. doi:10.1002/9780470994429. ISBN 978-0-470-06509-9.
- ↑ Emsley, John (2003) (in en). Nature's Building Blocks: An A-Z Guide to the Elements. Oxford University Press. ISBN 978-0-19-850340-8. https://books.google.com/books?id=j-Xu07p3cKwC&dq=%2219th+most+abundant+element%22&pg=PA486.
- ↑ Cowley, C. R.; Elste, G. H.; Urbanski, J. L. (October 1978). "Vanadium abundances in early A stars". Publications of the Astronomical Society of the Pacific 90: 536. doi:10.1086/130379. Bibcode: 1978PASP...90..536C.
- ↑ 65.0 65.1 65.2 Moskalyk, R.R; Alfantazi, A.M (September 2003). "Processing of vanadium: a review". Minerals Engineering 16 (9): 793–805. doi:10.1016/S0892-6875(03)00213-9. Bibcode: 2003MiEng..16..793M.
- ↑ Carlson, O. N.; Owen, C. V. (1961). "Preparation of High-Purity Vanadium Metalb by the Iodide Refining Process". Journal of the Electrochemical Society 108 (1): 88. doi:10.1149/1.2428019.
- ↑ Chandler, Harry (1998). Metallurgy for the Non-metallurgist. ASM International. pp. 6–7. ISBN 978-0-87170-652-2. https://books.google.com/books?id=arupok8PTBEC.
- ↑ Davis, Joseph R. (1995). Tool Materials: Tool Materials. ASM International. ISBN 978-0-87170-545-7. https://books.google.com/books?id=Kws7x68r_aUC&pg=PA11.
- ↑ Oleg D. Neikov; Naboychenko, Stanislav; Mourachova, Irina; Victor G. Gopienko; Irina V. Frishberg; Dina V. Lotsko (2009-02-24). Handbook of Non-Ferrous Metal Powders: Technologies and Applications. Elsevier. p. 490. ISBN 978-0-08-055940-7. https://books.google.com/books?id=6aP3te2hGuQC&pg=PA490. Retrieved 17 October 2013.
- ↑ "Technical Supplement: Titanium". http://www.sevencycles.com/buildingbike/techsupplement/ti.php.
- ↑ Zwicker, Ulrich (1974). "Herstellung des Metalls". Titan und Titanlegierungen. pp. 4–29. doi:10.1007/978-3-642-80587-5_2. ISBN 978-3-642-80588-2.
- ↑ Hardy, George F.; Hulm, John K. (15 February 1953). "Superconducting Silicides and Germanides". Physical Review 89 (4): 884. doi:10.1103/PhysRev.89.884. Bibcode: 1953PhRv...89Q.884H.
- ↑ Markiewicz, W.; Mains, E.; Vankeuren, R.; Wilcox, R.; Rosner, C.; Inoue, H.; Hayashi, C.; Tachikawa, K. (January 1977). "A 17.5 Tesla superconducting concentric Nb3Sn and V3Ga magnet system". IEEE Transactions on Magnetics 13 (1): 35–37. doi:10.1109/TMAG.1977.1059431.
- ↑ Verhoeven, J. D.; Pendray, A. H.; Dauksch, W. E. (September 1998). "The key role of impurities in ancient damascus steel blades". JOM 50 (9): 58–64. doi:10.1007/s11837-998-0419-y. Bibcode: 1998JOM....50i..58V.
- ↑ Rohrmann, B. (1985). "Vanadium in South Africa (Metal Review Series no. 2)". Journal of the Southern African Institute of Mining and Metallurgy 85 (5): 141–150.
- ↑ Overy, R. J. (1973). "Transportation and Rearmament in the Third Reich". The Historical Journal 16 (2): 389–409. doi:10.1017/s0018246x00005926.
- ↑ Langeslay, Ryan R.; Kaphan, David M.; Marshall, Christopher L.; Stair, Peter C.; Sattelberger, Alfred P.; Delferro, Massimiliano (8 October 2018). "Catalytic Applications of Vanadium: A Mechanistic Perspective". Chemical Reviews 119 (4): 2128–2191. doi:10.1021/acs.chemrev.8b00245. PMID 30296048.
- ↑ Eriksen, K.M.; Karydis, D.A.; Boghosian, S.; Fehrmann, R. (August 1995). "Deactivation and Compound Formation in Sulfuric-Acid Catalysts and Model Systems". Journal of Catalysis 155 (1): 32–42. doi:10.1006/jcat.1995.1185.
- ↑ Bauer, Günter; Güther, Volker; Hess, Hans; Otto, Andreas; Roidl, Oskar; Roller, Heinz; Sattelberger, Siegfried (2000). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a27_367.
- ↑ Abon, Michel; Volta, Jean-Claude (September 1997). "Vanadium phosphorus oxides for n-butane oxidation to maleic anhydride". Applied Catalysis A: General 157 (1–2): 173–193. doi:10.1016/S0926-860X(97)00016-1. Bibcode: 1997AppCA.157..173A.
- ↑ Fierro, J. G. L., ed (2006). Metal Oxides, Chemistry and Applications. CRC Press. pp. 415–455. ISBN 978-0-8247-2371-2.
- ↑ Joerissen, Ludwig; Garche, Juergen; Fabjan, Ch.; Tomazic, G. (March 2004). "Possible use of vanadium redox-flow batteries for energy storage in small grids and stand-alone photovoltaic systems". Journal of Power Sources 127 (1–2): 98–104. doi:10.1016/j.jpowsour.2003.09.066. Bibcode: 2004JPS...127...98J.
- ↑ Rychcik, M.; Skyllas-Kazacos, M. (January 1988). "Characteristics of a new all-vanadium redox flow battery". Journal of Power Sources 22 (1): 59–67. doi:10.1016/0378-7753(88)80005-3. Bibcode: 1988JPS....22...59R.
- ↑ Li, Liyu; Kim, Soowhan; Wang, Wei; Vijayakumar, M.; Nie, Zimin; Chen, Baowei; Zhang, Jianlu; Xia, Guanguang et al. (May 2011). "A Stable Vanadium Redox-Flow Battery with High Energy Density for Large-Scale Energy Storage". Advanced Energy Materials 1 (3): 394–400. doi:10.1002/aenm.201100008. Bibcode: 2011AdEnM...1..394L.
- ↑ Guan, H.; Buchheit, R. G. (1 March 2004). "Corrosion Protection of Aluminum Alloy 2024-T3 by Vanadate Conversion Coatings". Corrosion 60 (3): 284–296. doi:10.5006/1.3287733.
- ↑ Lositskii, N. T.; Grigor'ev, A. A.; Khitrova, G. V. (December 1966). "Welding of chemical equipment made from two-layer sheet with titanium protective layer (review of foreign literature)". Chemical and Petroleum Engineering 2 (12): 854–856. doi:10.1007/BF01146317. Bibcode: 1966CPE.....2..854L.
- ↑ Matsui, H.; Fukumoto, K.; Smith, D.L.; Chung, Hee M.; van Witzenburg, W.; Votinov, S.N. (October 1996). "Status of vanadium alloys for fusion reactors". Journal of Nuclear Materials 233-237: 92–99. doi:10.1016/S0022-3115(96)00331-5. Bibcode: 1996JNuM..233...92M. https://zenodo.org/record/1259631. Retrieved 10 August 2018.
- ↑ "Vanadium Data Sheet". ATI Wah Chang. http://www.wahchang.com/pages/products/data/pdf/Vanadium.pdf.
- ↑ , Michael R. & Chuanjing Xu"Electrode material with enhanced ionic transport properties" patent US7842420B2, issued 2010-11-30
- ↑ Kariatsumari, Koji (February 2008). "Li-Ion Rechargeable Batteries Made Safer". Nikkei Business Publications, Inc.. http://techon.nikkeibp.co.jp/article/HONSHI/20080129/146549/.
- ↑ Saıdi, M.Y.; Barker, J.; Huang, H.; Swoyer, J.L.; Adamson, G. (1 June 2003), "Performance characteristics of lithium vanadium phosphate as a cathode material for lithium-ion batteries", Journal of Power Sources 119–121: 266–272, doi:10.1016/S0378-7753(03)00245-3, Bibcode: 2003JPS...119..266S Selected papers presented at the 11th International Meeting on Lithium Batteries
- ↑ "Breakthrough New Material Brings Affordable, Sustainable Future Within Grasp" (in en). https://www.uh.edu/news-events/stories/2024/december/12202024-canepa-batteries.php.
- ↑ Sigel, Astrid; Sigel, Helmut, eds (1995). Vanadium and Its Role in Life. Metal Ions in Biological Systems. 31. CRC. ISBN 978-0-8247-9383-8.
- ↑ Gribble, Gordon W. (1999). "The diversity of naturally occurring organobromine compounds". Chemical Society Reviews 28 (5): 335–346. doi:10.1039/a900201d.
- ↑ Butler, Alison; Carter-Franklin, Jayme N. (2004). "The role of vanadium bromoperoxidase in the biosynthesis of halogenated marine natural products". Natural Product Reports 21 (1): 180–188. doi:10.1039/b302337k. PMID 15039842.
- ↑ Robson, R. L.; Eady, R. R.; Richardson, T. H.; Miller, R. W.; Hawkins, M.; Postgate, J. R. (1986). "The alternative nitrogenase of Azotobacter chroococcum is a vanadium enzyme". Nature 322 (6077): 388–390. doi:10.1038/322388a0. Bibcode: 1986Natur.322..388R.
- ↑ Smith, M. J. (1989). "Vanadium biochemistry: The unknown role of vanadium-containing cells in ascidians (sea squirts)". Experientia 45 (5): 452–7. doi:10.1007/BF01952027. PMID 2656286.
- ↑ MacAra, Ian G.; McLeod, G. C.; Kustin, Kenneth (1979). "Tunichromes and metal ion accumulation in tunicate blood cells". Comparative Biochemistry and Physiology B 63 (3): 299–302. doi:10.1016/0305-0491(79)90252-9.
- ↑ Trefry, John H.; Metz, Simone (1989). "Role of hydrothermal precipitates in the geochemical cycling of vanadium". Nature 342 (6249): 531–533. doi:10.1038/342531a0. Bibcode: 1989Natur.342..531T.
- ↑ Weiss, H.; Guttman, M. A.; Korkisch, J.; Steffan, I. (1977). "Comparison of methods for the determination of vanadium in sea-water". Talanta 24 (8): 509–11. doi:10.1016/0039-9140(77)80035-0. PMID 18962130.
- ↑ Ruppert, Edward E.; Fox, Richard, S.; Barnes, Robert D. (2004). Invertebrate Zoology (7th ed.). Cengage Learning. pp. 947. ISBN 978-81-315-0104-7.
- ↑ Kneifel, Helmut; Bayer, Ernst (June 1973). "Determination of the Structure of the Vanadium Compound, Amavadine, from Fly Agaric". Angewandte Chemie International Edition in English 12 (6): 508. doi:10.1002/anie.197305081.
- ↑ Falandysz, J.; Kunito, T.; Kubota, R.; Lipka, K.; Mazur, A.; Falandysz, Justyna J.; Tanabe, S. (31 August 2007). "Selected elements in fly agaric Amanita muscaria". Journal of Environmental Science and Health, Part A 42 (11): 1615–1623. doi:10.1080/10934520701517853. PMID 17849303. Bibcode: 2007JESHA..42.1615F.
- ↑ Berry, Robert E.; Armstrong, Elaine M.; Beddoes, Roy L.; Collison, David; Ertok, S. Nigar; Helliwell, Madeleine; Garner, C. David (15 March 1999). "The Structural Characterization of Amavadin". Angewandte Chemie 38 (6): 795–797. doi:10.1002/(SICI)1521-3773(19990315)38:6<795::AID-ANIE795>3.0.CO;2-7. PMID 29711812.
- ↑ da Silva, José A.L.; Fraústo da Silva, João J.R.; Pombeiro, Armando J.L. (August 2013). "Amavadin, a vanadium natural complex: Its role and applications". Coordination Chemistry Reviews 257 (15–16): 2388–2400. doi:10.1016/j.ccr.2013.03.010.
- ↑ Schwarz, Klaus; Milne, David B. (22 October 1971). "Growth Effects of Vanadium in the Rat". Science 174 (4007): 426–428. doi:10.1126/science.174.4007.426. PMID 5112000. Bibcode: 1971Sci...174..426S.
- ↑ Nickel. IN: Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Copper . National Academy Press. 2001, PP. 532–543.
- ↑ 108.0 108.1 Smith, D.M.; Pickering, R.M.; Lewith, G.T. (31 January 2008). "A systematic review of vanadium oral supplements for glycaemic control in type 2 diabetes mellitus". QJM 101 (5): 351–358. doi:10.1093/qjmed/hcn003. PMID 18319296.
- ↑ "Vanadium (vanadyl sulfate). Monograph". Altern Med Rev 14 (2): 177–80. 2009. PMID 19594227.
- ↑ Lynch, Brendan M. (21 September 2017). "Hope to discover sure signs of life on Mars? New research says look for the element vanadium". PhysOrg. https://phys.org/news/2017-09-life-mars-element-vanadium.html.
- ↑ Marshall, C. P; Olcott Marshall, A; Aitken, J. B; Lai, B; Vogt, S; Breuer, P; Steemans, P; Lay, P. A (2017). "Imaging of Vanadium in Microfossils: A New Potential Biosignature". Astrobiology 17 (11): 1069–1076. doi:10.1089/ast.2017.1709. PMID 28910135. Bibcode: 2017AsBio..17.1069M.
- ↑ Srivastava, A. K. (2000). "Anti-diabetic and toxic effects of vanadium compounds". Molecular and Cellular Biochemistry 206 (1–2): 177–182. doi:10.1023/A:1007075204494. PMID 10839208.
- ↑ Roschin, A. V. (1967). "Toksikologiia soedineniĭ vanadiia, primeneniaemykh v sovremennoĭ promyshlennosti" (in ru). Gigiena i Sanitariia (Water Res.) 32 (6): 26–32. PMID 5605589.
- ↑ 114.0 114.1 "Occupational Safety and Health Guidelines for Vanadium Pentoxide". Occupational Safety and Health Administration. http://www.osha.gov/SLTC/healthguidelines/vanadiumpentoxidedust/recognition.html.
- ↑ "Long-term exposure to PM2·5 constituents and incident cancer among Medicare beneficiaries in the USA: a national cohort study". The Lancet. https://www.thelancet.com/callback?red_uri=%2Fjournals%2Flanplh%2Farticle%2FPIIS2542-5196%2825%2900212-8%2Ffulltext%3Ffbclid%3DIwdGRleAOk2tdleHRuA2FlbQIxMQBzcnRjBmFwcF9pZA8xNzM4NDc2NDI2NzAzNzAAAR4enTLCqO_TgPUdAPjNj6oagf5EItjHwzncIL26C7EWKygKv3Kp_ZmBMNAEDA_aem_d9YygSXkhsuBJHqIiape2A&code=LG2oyADcpm_-JRzcRG2u697dkP1WsCksz9RAP0PQ&state=16632644460.
- ↑ Sax, N. I. (1984). Dangerous Properties of Industrial Materials (6th ed.). Van Nostrand Reinhold. pp. 2717–2720.
- ↑ 117.0 117.1 Ress, N. B.; Chou, B. J.; Renne, R. A.; Dill, J. A.; Miller, R. A.; Roycroft, J. H.; Hailey, J. R.; Haseman, J. K. et al. (1 August 2003). "Carcinogenicity of Inhaled Vanadium Pentoxide in F344/N Rats and B6C3F1 Mice". Toxicological Sciences 74 (2): 287–296. doi:10.1093/toxsci/kfg136. PMID 12773761.
- ↑ Wörle-Knirsch, Jörg M.; Kern, Katrin; Schleh, Carsten; Adelhelm, Christel; Feldmann, Claus; Krug, Harald F. (2007). "Nanoparticulate Vanadium Oxide Potentiated Vanadium Toxicity in Human Lung Cells". Environmental Science and Technology 41 (1): 331–336. doi:10.1021/es061140x. PMID 17265967. Bibcode: 2007EnST...41..331W.
- ↑ Ścibior, A.; Zaporowska, H.; Ostrowski, J. (2006). "Selected haematological and biochemical parameters of blood in rats after subchronic administration of vanadium and/or magnesium in drinking water". Archives of Environmental Contamination and Toxicology 51 (2): 287–295. doi:10.1007/s00244-005-0126-4. PMID 16783625. Bibcode: 2006ArECT..51..287S.
- ↑ González-Villalva, Adriana; Fortoul, Teresa I; Avila-Costa, Maria Rosa; Piñón-Zarate, Gabriela; Rodriguez-Lara, Vianey; Martínez-Levy, Gabriela; Rojas-Lemus, Marcela; Bizarro-Nevarez, Patricia et al. (April 2006). "Thrombocytosis induced in mice after subacute and subchronic V2O5 inhalation". Toxicology and Industrial Health 22 (3): 113–116. doi:10.1191/0748233706th250oa. PMID 16716040. Bibcode: 2006ToxIH..22..113G.
- ↑ Kobayashi, Kazuo; Himeno, Seiichiro; Satoh, Masahiko; Kuroda, Junji; Shibata, Nobuo; Seko, Yoshiyuki; Hasegawa, Tatsuya (2006). "Pentavalent vanadium induces hepatic metallothionein through interleukin-6-dependent and -independent mechanisms". Toxicology 228 (2–3): 162–170. doi:10.1016/j.tox.2006.08.022. PMID 16987576. Bibcode: 2006Toxgy.228..162K.
- ↑ Soazo, Marina; Garcia, Graciela Beatriz (2007). "Vanadium exposure through lactation produces behavioral alterations and CNS myelin deficit in neonatal rats". Neurotoxicology and Teratology 29 (4): 503–510. doi:10.1016/j.ntt.2007.03.001. PMID 17493788. Bibcode: 2007NTxT...29..503S.
- ↑ Barceloux, Donald G. (1999). "Vanadium". Clinical Toxicology 37 (2): 265–278. doi:10.1081/CLT-100102425. PMID 10382561.
- ↑ Duffus, J. H. (2007). "Carcinogenicity classification of vanadium pentoxide and inorganic vanadium compounds, the NTP study of carcinogenicity of inhaled vanadium pentoxide, and vanadium chemistry". Regulatory Toxicology and Pharmacology 47 (1): 110–114. doi:10.1016/j.yrtph.2006.08.006. PMID 17030368. Bibcode: 2007RToxP..47..110D.
- ↑ Opreskos, Dennis M. (1991). "Toxicity Summary for Vanadium". Oak Ridge National Laboratory. https://rais.ornl.gov/tox/profiles/vanadium_f_V1.html.
- ↑ Woodyard, Doug (2009-08-18). Pounder's Marine Diesel Engines and Gas Turbines. Butterworth-Heinemann. p. 92. ISBN 978-0-08-094361-9. https://books.google.com/books?id=RC_k4q6y-JIC&pg=PA92.
- ↑ Totten, George E.; Westbrook, Steven R.; Shah, Rajesh J. (2003-06-01). Fuels and Lubricants Handbook: Technology, Properties, Performance, and Testing. p. 152. ISBN 978-0-8031-2096-9. https://books.google.com/books?id=J_AkNu-Y1wQC&pg=PA152.
Further reading
- Slebodnick, Carla (1999). "Modeling the Biological Chemistry of Vanadium: Structural and Reactivity Studies Elucidating Biological Function". in Hill, Hugh A.O.. Metal sites in proteins and models: phosphatases, Lewis acids, and vanadium. Springer. ISBN 978-3-540-65553-4. https://books.google.com/books?id=ZF5aNOeHd5sC&pg=PA51.
External links
| Wikimedia Commons has media related to Vanadium. |
 "Vanadium". Encyclopædia Britannica. XXIV (9th ed.). 1875–1889. p. 54.
"Vanadium". Encyclopædia Britannica. XXIV (9th ed.). 1875–1889. p. 54.- Vanadium at The Periodic Table of Videos (University of Nottingham)
 |
 KSF
KSF