Xenon isotope geochemistry
Topic: Chemistry
 From HandWiki - Reading time: 16 min
From HandWiki - Reading time: 16 min
Xenon isotope geochemistry uses the abundance of xenon (Xe) isotopes and total xenon to investigate how Xe has been generated, transported, fractionated, and distributed in planetary systems. Xe has nine stable or very long-lived isotopes. Radiogenic 129Xe and fissiogenic 131,132,134,136Xe isotopes are of special interest in geochemical research.[1] The radiogenic and fissiogenic properties can be used in deciphering the early chronology of Earth.[2][3] Elemental Xe in the atmosphere is depleted and isotopically enriched in heavier isotopes relative to estimated solar abundances.[1] The depletion and heavy isotopic enrichment can be explained by hydrodynamic escape to space that occurred in Earth's early atmosphere. Differences in the Xe isotope distribution between the deep mantle (from Ocean Island Basalts, or OIBs), shallower Mid-ocean Ridge Basalts (MORBs), and the atmosphere can be used to deduce Earth's history of formation and differentiation of the solid Earth into layers.
Background
Xe is the heaviest noble gas in the Earth's atmosphere. It has seven stable isotopes (126Xe,128Xe,129Xe,130Xe,131Xe, 132Xe, 134Xe) and two isotopes (124Xe, 136Xe) with long-lived half-lives. Xe has four synthetic radioisotopes with very short half-lives, usually less than one month. Xenon-129 can be used to examine the early history of the Earth. 129Xe was derived from the extinct nuclide of iodine, iodine-129 or 129I (with a half-life of 15.7 Million years, or Myr), which can be used in iodine-xenon (I-Xe) dating. The production of 129Xe stopped within about 100 Myr after the start of the Solar System because 129I became extinct.[1] In the modern atmosphere, about 6.8%[4] of atmospheric 129Xe originated from the decay 129I in the first ~100 Myr of the Solar System's history, i.e., during and immediately following Earth's accretion.
Fissiogenic Xe isotopes were generated mainly from the extinct nuclide, plutonium-244 or 244Pu (half-life of 80 Myr), and also the extant nuclide, uranium-238 or 238U (half-life of 4468 Myr). Spontaneous fission of 238U has generated ~5%[5] as much fissiogenic Xe as 244Pu. Pu and U fission produce the four fissiogenic isotopes, 136Xe, 134Xe, 132Xe, and 131Xe in distinct proportions. A reservoir that remains an entirely closed system over Earth's history has a ratio of Pu- to U-derived fissiogenic Xe reaching to ~27.[6] Accordingly, the isotopic composition of the fissiogenic Xe for a closed-system reservoir would largely resemble that produced from pure 244Pu fission.[7] Loss of Xe from a reservoir after 244Pu becomes extinct (500 Myr) would lead to a greater contribution of 238U fission to the fissiogenic Xe.[1]
Notation
Differences in the abundance of isotopes among natural samples are extremely small (almost always below 0.1% or 1 per mille). Nevertheless, these very small differences can record meaningful geological processes.[8] To compare these tiny but meaningful differences, isotope abundances in natural materials are often reported relative to isotope abundances in designated standards, with the delta (δ) notation. The absolute values of Xe isotopes are normalized to atmospheric 130Xe.[9] Define where i = 124, 126, 128, 129, 131, 132, 134, 136.
Applications
The age of Earth
Iodine-129 decays with a half-life of 15.7 Ma into 129Xe, resulting in excess 129Xe in primitive meteorites relative to primordial Xe isotopic compositions. The property of 129I can be used in radiometric chronology. However, as detailed below, the age of Earth's formation cannot be deduced directly from I-Xe dating. The major problem is the Xe closure time, or the time when the early Earth system stopped gaining substantial new material from space. When the Earth became closed for the I-Xe system, Xe isotope evolution began to obey a simple radioactive decay law as shown below and became predictable.
The principle of radiogenic chronology is,[5] if at time t1 the quantity of a radioisotope is P1 while at some previous time this quantity was P0, the interval between t1 and t0 is given by the law of radioactive decay as
Here is the decay constant of the radioisotope, which is the probability of decay per nucleus per unit time. The decay constant is related to the half life t1/2, by t1/2= ln(2)/
Calculations
The I-Xe system was first applied in 1975 to estimate the age of the Earth.[10] For all Xe isotopes, the initial isotope composition of iodine in the Earth is given by
where is the isotopic ratios of iodine at the time that Earth primarily formed, is the isotopic ratio of iodine at the end of stellar nucleosynthesis, and is the time interval between the end of stellar nucleosynthesis and the formation of the Earth. The estimated iodine-127 concentration in the Bulk Silicate Earth (BSE) (= crust + mantle average) ranges from 7 to 10 parts per billion (ppb) by mass.[11][12] If the BSE represents Earth's chemical composition,[2] the total 127I in the BSE ranges from 2.26×1017 to 3.23×1017 moles. The meteorite Bjurböle is 4.56 billion years old[13] with an initial 129I/127I ratio of 1.1×10−4,[14] so an equation can be derived as
where is the interval between the formation of the Earth and the formation of meteorite Bjurböle. Given the half life of 129I of 15.7 Myr, and assuming that all the initial 129I has decayed to 129Xe, the following equation can be derived:
129Xe in the modern atmosphere is 3.63×1013 grams. The iodine content for BSE lies between 10 and 12 ppb by mass. Consequently, should be 108 Myr,[2][3][10] i.e., the Xe-closure age is 108 Myr younger than the age of meteorite Bjurböle. The estimated Xe closure time was ~4.45 billion years ago when the growing Earth started to retain Xe in its atmosphere, which is coincident with ages derived from other geochronology dating methods.[2][3][15]
Xe closure age problem
There are some disputes about using I-Xe dating to estimate the Xe closure time. First, in the early solar system, planetesimals collided and grew into larger bodies that accreted to form the Earth. But there could be a 107 to 108 years time gap in Xe closure time between the Earth's inner and outer regions.[16] Some research support 4.45 Ga probably represents the time when the last giant impactor (Martian-size) hit Earth,[3] but some regard it as the time of core-mantle differentiation.[2] The second problem is that the total inventory of 129Xe on Earth may be larger than that of the atmosphere since the lower mantle hadn't been entirely mixed, which may underestimate 129Xe in the calculation. Last but not least, if Xe gas not been lost from the atmosphere during a long interval of early Earth's history, the chronology based on 129I-129Xe would need revising[17] since 129Xe and 127Xe could be greatly altered.
Loss of earth's earliest atmosphere
Compared with solar xenon, Earth's atmospheric Xe is enriched in heavy isotopes by 3 to 4% per atomic mass unit (amu).[18] However, the total abundance of xenon gas is depleted by one order of magnitude relative to other noble gases.[15] The elemental depletion while relative enrichment in heavy isotopes is called the "Xenon paradox". A possible explanation is that some processes can specifically diminish xenon rather than other light noble gases (e.g. Krypton) and preferentially remove lighter Xe isotopes.
In the last 2 decades, two categories of models have been proposed to solve the xenon paradox. The first assumes that the Earth accreted from porous planetesimals, and isotope fractionation happened due to gravitational separation.[19] However, this model cannot reproduce the abundance and isotopic composition of light noble gases in the atmosphere. The second category supposes a massive impact resulted in an aerodynamic drag on heavier gases.[20] Both the aerodynamic drag and the downward gravitational effect lead to a mass-dependent loss of Xe gases. But following research suggested that Xe isotope mass fractionation shouldn't be a rapid, single event.[21]
Research published since 2018 on noble gases preserved in Archean (3.5–3.0 Ga old) samples may provide a solution to the Xe paradox.[21][22] Isotopically mass fractionated Xe is found in tiny inclusions of ancient seawater in Archean barite[23] and hydrothermal quartz.[24] The distribution of Xe isotopes lies between the primordial solar and the modern atmospheric Xe isotope patterns. The isotopic fractionation gradually increases relative to the solar distribution as Earth evolves over its first 2 billion years.[21] This two billion-year history of evolving Xe fractionation coincides with early solar system conditions including high solar extreme ultraviolet (EUV) radiation[15][25][26] and large impacts that could energize large rates of hydrogen escape to space that are big enough to drag out xenon. However, models of neutral xenon atoms escaping cannot resolve the problem that other lighter noble gas elements don't show the signal of depletion or mass-dependent fractionation. For example, because Kr is lighter than Xe, Kr should also have escaped in a neutral wind. Yet the isotopic distribution of atmospheric Kr on Earth is significantly less fractionated than atmospheric Xe.[16]

A current explanation[28][29] is that hydrodynamic escape can preferentially remove lighter atmospheric species and lighter isotopes of Xe in the form of charged ions instead of neutral atoms. Hydrogen is liberated from hydrogen-bearing gases (H2 or CH4) by photolysis in the early Earth atmosphere. Hydrogen is light and can be abundant at the top of the atmosphere and escape. In the polar regions where there are open magnetic field lines, hydrogen ions can drag ionized Xe out from the atmosphere to space even though neutral Xe cannot escape.[27]
The mechanism is summarized as below.
Xe can be directly photo-ionized by UV radiation in range of [28]
Or Xe can be ionized by change exchange with H2 and CO2 through
where H+ and CO2+ can come from EUV dissociation. Xe+ is chemically inert in H, H2, or CO2 atmospheres.[30] As a result, Xe+ tends to persist. These ions interact strongly with each other through the Coulomb force and are finally dragged away by strong ancient polar wind. Isotope mass fractionation accumulates as lighter isotopes of Xe+ preferentially escape from the Earth. A preliminary model suggests that Xe can escape in the Archean if the atmosphere contains >1% H2 or >0.5% methane.[28]
When O2 levels increased in the atmosphere, Xe+ could exchange positive charge with O2 though[30]
From this reaction, Xe escape stopped when the atmosphere became enriched in O2. As a result, Xe isotope fractionation may provide insights into the long history of hydrogen escape that ended with the Great Oxidation Event (GOE).[28] Understanding Xe isotopes is promising to reconstruct hydrogen or methane escape history that irreversibly oxidized the Earth and drove biological evolution toward aerobic ecological systems.[21][31][32] Other factors, such as the hydrogen (or methane) concentration becoming too low or EUV radiation from the aging Sun becoming too weak, can also cease the hydrodynamic escape of Xe,[28] but are not mutually exclusive.
Organic hazes on Archean Earth could also scavenge isotopically heavy Xe.[25] Ionized Xe can be chemically incorporated into organic materials,[33] going through the terrestrial weathering cycle on the surface. The trapped Xe is mass fractionated by about 1% per amu in heavier isotopes[28] but they may be released again and recover the original unfractionated composition, making them not sufficient to totally resolve Xe paradox.
Comparison between Kr and Xe in the atmosphere
Observed atmospheric Xe is depleted relative to Chondritic meteorites by a factor of 4 to 20 when compared to Kr.[27] In contrast, the stable isotopes of Kr are barely fractionated. This mechanism is unique to Xe since Kr+ ions are quickly neutralized via[34]
Therefore, Kr can be rapidly returned to neutral and wouldn't be dragged away by the charged ion wind in the polar region. Hence Kr is retained in the atmosphere.
Relation with Mass Independent Fraction of Sulfur Isotopes (MIF-S)
The signal of mass-independent fractionation of sulfur isotopes, known as MIF-S, correlates with the end of Xe isotope fractionation. During the Great Oxidation Event (GOE), the ozone layer formed when O2 rose, accounting for the end of the MIF-S signature. The disappearance of the MIF-S signal has been regarded as changing the redox ratio of Earth's surface reservoirs.[35][36] However, potential memory effects of MIF-S due to oxidative weathering can lead to large uncertainty on the process and chronology of GOE.[37] Compared to the MIF-S signals, hydrodynamic escape of Xe is not affected by the ozone formation and may be even more sensitive to O2 availability,[32] promising to provide more details about the oxidation history of Earth.[32]
Xe Isotopes as mantle tracers
Xe isotopes are also promising in tracing mantle dynamics in Earth's evolution. The first explicit recognition of non-atmospheric Xe in terrestrial samples came from the analysis of CO2-well gas in New Mexico, displaying an excess of 129I-derived or primitive source 129Xe and high content in 131-136Xe due to the decay of 238U.[38] At present, the excess of 129Xe and 131-136Xe has been widely observed in mid-ocean ridge basalt (MORBs)[39][40][41] and Oceanic island basalt (OIBs).[42] Because 136Xe receives more fissiogenic contribution than other heavy Xe isotopes, 129Xe (decay of 129I) and 136Xe are usually normalized to 130Xe when discussing Xe isotope trends of different mantle sources.[11] MORBs' 129Xe/130Xe and 136Xe/130Xe ratios lie on a trend from atmospheric ratios to higher values[39] and seemingly contaminated by the air. Oceanic island basalt (OIBs) data lies lower than those in MORBs, implying different Xe sources for OIBs and MORBs.[42]
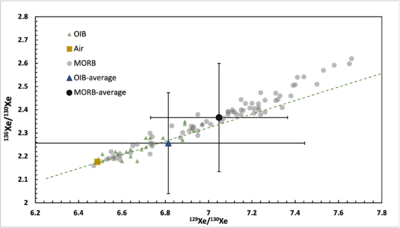
The deviations in 129Xe/130Xe ratio between air and MORBs show that mantle degassing occurred before 129I was extinct, otherwise 129Xe/130Xe in the air would be the same as in the mantle.[45][46] The differences in the 129Xe/130Xe ratio between MORBs and OIBs may indicate that the mantle reservoirs are still not thoroughly mixed. The chemical differences between OIBs and MORBs still await discovery.[11]
To obtain mantle Xe isotope ratios, it is necessary to remove contamination by atmospheric Xe, which could start before 2.5 billion years ago.[47][48] Theoretically, the many non-radiogenic isotopic ratios (124Xe/130Xe, 126Xe/130Xe, and 128Xe/130Xe) can be used to accurately correct for atmospheric contamination if slight differences between air and mantle can be precisely measured. Still, we cannot reach such precision with current techniques.[11]
Xe in other planets
Mars
On Mars, Xe isotopes in the present atmosphere are mass fractionated relative to their primordial composition from in situ measurement of the Curiosity Rover at Gale Crater, Mars.[49] Paleo-atmospheric Xe trapped in the Martian regolith breccia NWA 11220 is mass-dependently fractionated relative to solar Xe by ~16.2‰.[29] The extent of fractionation is comparable for Mars and Earth, which may be compelling evidence that hydrodynamic escape also occurred in the Mars history. The regolith breccia NWA7084 and the >4 Ga orthopyroxene ALH84001 Martian meteorites trap ancient Martian atmospheric gases with little if any Xe isotopic fractionation relative to modern Martian atmospheric Xe.[50] Alternative models for Mars consider that the isotopic fractionation and escape of Mars atmospheric Xe occurred very early in the planet's history and ceased around a few hundred million years after planetary formation rather than continuing during its evolutionary history[21][51]
Venus
Xe has not been detected in Venus's atmosphere. 132Xe has an upper limit of 10 parts per billion by volume.[51][52] The absence of data on the abundance of Xe precludes us from evaluating if the abundance of Xe is close to solar values or if there is Xe paradox on Venus. The lack also prevents us from checking if the isotopic composition has been mass dependently fractionated, as in the case of Earth and Mars.
Jupiter
Jupiter's atmosphere has 2.5 ± 0.5 times the solar abundance values for Xenon and similarly elevated argon and krypton (2.1 ± 0.5 and 2.7 ± 0.5 times solar values separately). These signals of enrichment are due to these elements coming to Jupiter in very cold (T<30K) icy planetesimals.[53]
See also
References
- ↑ 1.0 1.1 1.2 1.3 Mukhopadhyay, Sujoy (2018), Bobrowsky, Peter; Marker, Brian, eds. (in en), Xenon Isotopes, Encyclopedia of Earth Sciences Series, Cham: Springer International Publishing, pp. 1–8, doi:10.1007/978-3-319-39193-9_203-1, ISBN 978-3-319-12127-7, http://link.springer.com/10.1007/978-3-319-39193-9_203-1, retrieved 2022-06-03
- ↑ 2.0 2.1 2.2 2.3 2.4 Allègre, Claude J. (1995). "The age of the Earth". Geochimica et Cosmochimica Acta 59 (8): 1445–1456. doi:10.1016/0016-7037(95)00054-4. Bibcode: 1995GeCoA..59.1445A. https://dx.doi.org/10.1016/0016-7037%2895%2900054-4.
- ↑ 3.0 3.1 3.2 3.3 Zhang, Youxue (1998). "The young age of Earth" (in en). Geochimica et Cosmochimica Acta 62 (18): 3185–3189. doi:10.1016/S0016-7037(98)00211-7. Bibcode: 1998GeCoA..62.3185Z. https://linkinghub.elsevier.com/retrieve/pii/S0016703798002117.
- ↑ Pepin, Robert O. (1991). "On the origin and early evolution of terrestrial planet atmospheres and meteoritic volatiles" (in en). Icarus 92 (1): 2–79. doi:10.1016/0019-1035(91)90036-S. Bibcode: 1991Icar...92....2P. https://linkinghub.elsevier.com/retrieve/pii/001910359190036S.
- ↑ 5.0 5.1 Dickin, Alan P. (2018-02-08). Radiogenic Isotope Geology (3 ed.). Cambridge University Press. doi:10.1017/9781316163009. ISBN 978-1-316-16300-9. https://www.cambridge.org/core/product/identifier/9781316163009/type/book.
- ↑ Tolstikhin, I.N.; Kramers, J.D.; Hofmann, A.W. (2006). "A chemical Earth model with whole mantle convection: The importance of a core–mantle boundary layer (D") and its early formation" (in en). Chemical Geology 226 (3–4): 79–99. doi:10.1016/j.chemgeo.2005.09.015. Bibcode: 2006ChGeo.226...79T. https://linkinghub.elsevier.com/retrieve/pii/S0009254105004213.
- ↑ Mukhopadhyay, Sujoy; Parai, Rita (2019-05-30). "Noble Gases: A Record of Earth's Evolution and Mantle Dynamics" (in en). Annual Review of Earth and Planetary Sciences 47 (1): 389–419. doi:10.1146/annurev-earth-053018-060238. ISSN 0084-6597. Bibcode: 2019AREPS..47..389M. https://www.annualreviews.org/doi/10.1146/annurev-earth-053018-060238.
- ↑ Sharp, Zachary (2017). Principles of Stable Isotope Geochemistry (2nd ed.). pp. 2–4.
- ↑ Basford, JR; Dragon, JC; Pepin, RO (1973). "Krypton and Xenon in lunar fines". Proceedings of the Lunar Science Conference 4 (1915).
- ↑ 10.0 10.1 Wetherill, G W (1975). "Radiometric Chronology of the Early Solar System" (in en). Annual Review of Nuclear Science 25 (1): 283–328. doi:10.1146/annurev.ns.25.120175.001435. ISSN 0066-4243. Bibcode: 1975ARNPS..25..283W. https://www.annualreviews.org/doi/10.1146/annurev.ns.25.120175.001435.
- ↑ 11.0 11.1 11.2 11.3 11.4 Heinrich D. Holland, ed (2014). Treatise on geochemistry (Second ed.). Amsterdam. ISBN 978-0-08-098300-4. OCLC 864682251.
- ↑ McDonough, W.F.; Sun, S.-s. (1995). "The composition of the Earth" (in en). Chemical Geology 120 (3–4): 223–253. doi:10.1016/0009-2541(94)00140-4. Bibcode: 1995ChGeo.120..223M. https://linkinghub.elsevier.com/retrieve/pii/0009254194001404.
- ↑ Hudson, GB (1989). "The early solar system abundance of 244Pu as inferred from the St. Severin chondrite". pp. 547–557.
- ↑ Swindle, TD (1988). Iodine-xenon dating IN: Meteorites and the early solar system (A89-27476 10-91). Tucson, AZ, University of Arizona Press. pp. 1127–1146.
- ↑ 15.0 15.1 15.2 15.3 Avice, G.; Marty, B (2014-09-13). "The iodine–plutonium–xenon age of the Moon–Earth system revisited" (in en). Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 372 (2024): 20130260. doi:10.1098/rsta.2013.0260. ISSN 1364-503X. PMID 25114317. Bibcode: 2014RSPTA.37230260A.
- ↑ 16.0 16.1 Ozima, M.; Podosek, F. A.; Igarashi, G. (1985). "Terrestrial xenon isotope constraints on the early history of the Earth" (in en). Nature 315 (6019): 471–474. doi:10.1038/315471a0. ISSN 0028-0836. Bibcode: 1985Natur.315..471O. http://www.nature.com/articles/315471a0.
- ↑ Turner, G. (2003). "M. Ozima and F.A. Podosek Noble Gas Geochemistry" (in en). Mineralogical Magazine (Cambridge: Cambridge University Press) 67 (2): 418–419. doi:10.1180/S0026461X00011725. ISBN 05-2180-366-7. ISSN 0026-461X. https://www.cambridge.org/core/product/identifier/S0026461X00011725/type/journal_article.
- ↑ Ozima, M.; Podosek, F. A. (1999-11-10). "Formation age of Earth from 129 I/ 127 I and 244 Pu/ 238 U systematics and the missing Xe" (in en). Journal of Geophysical Research: Solid Earth 104 (B11): 25493–25499. doi:10.1029/1999JB900257. Bibcode: 1999JGR...10425493O.
- ↑ Ozima, Minoru; Zahnle, Kevin (1993). "Mantle degassing and atmospheric evolution: Noble gas view.". Geochemical Journal 27 (4): 185–200. doi:10.2343/geochemj.27.185. ISSN 0016-7002. Bibcode: 1993GeocJ..27..185O. http://www.jstage.jst.go.jp/article/geochemj1966/27/4-5/27_4-5_185/_article.
- ↑ Dauphas, Nicolas (2003). "The dual origin of the terrestrial atmosphere" (in en). Icarus 165 (2): 326–339. doi:10.1016/S0019-1035(03)00198-2. Bibcode: 2003Icar..165..326D. https://linkinghub.elsevier.com/retrieve/pii/S0019103503001982.
- ↑ 21.0 21.1 21.2 21.3 21.4 Avice, G.; Marty, B.; Burgess, R.; Hofmann, A.; Philippot, P.; Zahnle, K.; Zakharov, D. (2018). "Evolution of atmospheric xenon and other noble gases inferred from Archean to Paleoproterozoic rocks" (in en). Geochimica et Cosmochimica Acta 232: 82–100. doi:10.1016/j.gca.2018.04.018. Bibcode: 2018GeCoA.232...82A.
- ↑ Broadley, M.W.; Byrne, D.J.; Ardoin, L.; Almayrac, M.G.; Bekaert, D.V.; Marty, B. (2022). "High precision noble gas measurements of hydrothermal quartz reveal variable loss rate of Xe from the Archean atmosphere" (in en). Earth and Planetary Science Letters 588: 117577. doi:10.1016/j.epsl.2022.117577. Bibcode: 2022E&PSL.58817577B.
- ↑ Srinivasan, B. (1976). "Barites: anomalous xenon from spallation and neutron-induced reactions" (in en). Earth and Planetary Science Letters 31 (1): 129–141. doi:10.1016/0012-821X(76)90104-7. Bibcode: 1976E&PSL..31..129S. https://linkinghub.elsevier.com/retrieve/pii/0012821X76901047.
- ↑ Pujol, Magali; Marty, Bernard; Burgess, Ray (2011). "Chondritic-like xenon trapped in Archean rocks: A possible signature of the ancient atmosphere" (in en). Earth and Planetary Science Letters 308 (3–4): 298–306. doi:10.1016/j.epsl.2011.05.053. Bibcode: 2011E&PSL.308..298P. https://linkinghub.elsevier.com/retrieve/pii/S0012821X11003463.
- ↑ 25.0 25.1 Hébrard, E.; Marty, B. (2014). "Coupled noble gas–hydrocarbon evolution of the early Earth atmosphere upon solar UV irradiation" (in en). Earth and Planetary Science Letters 385: 40–48. doi:10.1016/j.epsl.2013.10.022. Bibcode: 2014E&PSL.385...40H. https://linkinghub.elsevier.com/retrieve/pii/S0012821X13005852.
- ↑ Avice, Guillaume; Marty, Bernard; Burgess, Ray (2017). "The origin and degassing history of the Earth's atmosphere revealed by Archean xenon" (in en). Nature Communications 8 (1): 15455. doi:10.1038/ncomms15455. ISSN 2041-1723. PMID 28516958. Bibcode: 2017NatCo...815455A.
- ↑ 27.0 27.1 27.2 Catling, David C.; Zahnle, Kevin J. (2020-02-28). "The Archean atmosphere" (in en). Science Advances 6 (9): eaax1420. doi:10.1126/sciadv.aax1420. ISSN 2375-2548. PMID 32133393. Bibcode: 2020SciA....6.1420C.
- ↑ 28.0 28.1 28.2 28.3 28.4 28.5 Zahnle, Kevin J.; Gacesa, Marko; Catling, David C. (2019). "Strange messenger: A new history of hydrogen on Earth, as told by Xenon" (in en). Geochimica et Cosmochimica Acta 244: 56–85. doi:10.1016/j.gca.2018.09.017. Bibcode: 2019GeCoA.244...56Z. https://linkinghub.elsevier.com/retrieve/pii/S0016703718305349.
- ↑ 29.0 29.1 Cassata, William S.; Zahnle, Kevin J.; Samperton, Kyle M.; Stephenson, Peter C.; Wimpenny, Josh (2022). "Xenon isotope constraints on ancient Martian atmospheric escape" (in en). Earth and Planetary Science Letters 580: 117349. doi:10.1016/j.epsl.2021.117349. Bibcode: 2022E&PSL.58017349C.
- ↑ 30.0 30.1 Anicich, V. G. (1993). "A survey of bimolecular ion-molecule reactions for use in modeling the chemistry of planetary atmospheres, cometary comae, and interstellar clouds - 1993 supplement" (in en). The Astrophysical Journal Supplement Series 84: 215. doi:10.1086/191752. ISSN 0067-0049. Bibcode: 1993ApJS...84..215A. http://adsabs.harvard.edu/doi/10.1086/191752.
- ↑ Catling, David C.; Zahnle, Kevin J.; McKay, Christopher P. (2001-08-03). "Biogenic Methane, Hydrogen Escape, and the Irreversible Oxidation of Early Earth" (in en). Science 293 (5531): 839–843. doi:10.1126/science.1061976. ISSN 0036-8075. PMID 11486082. Bibcode: 2001Sci...293..839C. https://www.science.org/doi/10.1126/science.1061976.
- ↑ 32.0 32.1 32.2 Ardoin, L.; Broadley, M.W.; Almayrac, M.; Avice, G.; Byrne, D.J.; Tarantola, A.; Lepland, A.; Saito, T. et al. (2022). "The end of the isotopic evolution of atmospheric xenon". Geochemical Perspectives Letters 20: 43–47. doi:10.7185/geochemlet.2207. Bibcode: 2022GChPL..20...43A. https://www.geochemicalperspectivesletters.org/article2207.
- ↑ Marrocchi, Yves; Marty, Bernard (2013-08-28). "Experimental determination of the xenon isotopic fractionation during adsorption: XENON ISOTOPIC FRACTIONATION" (in en). Geophysical Research Letters 40 (16): 4165–4170. doi:10.1002/grl.50845.
- ↑ Anicich, V. G. (1993). "A survey of bimolecular ion-molecule reactions for use in modeling the chemistry of planetary atmospheres, cometary comae, and interstellar clouds - 1993 supplement" (in en). The Astrophysical Journal Supplement Series 84: 215. doi:10.1086/191752. ISSN 0067-0049. Bibcode: 1993ApJS...84..215A. http://adsabs.harvard.edu/doi/10.1086/191752.
- ↑ Lyons, Timothy W.; Reinhard, Christopher T.; Planavsky, Noah J. (2014). "The rise of oxygen in Earth's early ocean and atmosphere" (in en). Nature 506 (7488): 307–315. doi:10.1038/nature13068. ISSN 0028-0836. PMID 24553238. Bibcode: 2014Natur.506..307L. http://www.nature.com/articles/nature13068.
- ↑ Farquhar, James; Wing, Boswell A (2003). "Multiple sulfur isotopes and the evolution of the atmosphere" (in en). Earth and Planetary Science Letters 213 (1–2): 1–13. doi:10.1016/S0012-821X(03)00296-6. Bibcode: 2003E&PSL.213....1F. https://linkinghub.elsevier.com/retrieve/pii/S0012821X03002966.
- ↑ Philippot, Pascal; Ávila, Janaína N.; Killingsworth, Bryan A.; Tessalina, Svetlana; Baton, Franck; Caquineau, Tom; Muller, Elodie; Pecoits, Ernesto et al. (2018). "Globally asynchronous sulphur isotope signals require re-definition of the Great Oxidation Event" (in en). Nature Communications 9 (1): 2245. doi:10.1038/s41467-018-04621-x. ISSN 2041-1723. PMID 29884814. Bibcode: 2018NatCo...9.2245P.
- ↑ Phinney, D.; Tennyson, J.; Frick, U. (1978). "Xenon in CO 2 well gas revisited" (in en). Journal of Geophysical Research 83 (B5): 2313. doi:10.1029/JB083iB05p02313. ISSN 0148-0227. Bibcode: 1978JGR....83.2313P. http://doi.wiley.com/10.1029/JB083iB05p02313.
- ↑ 39.0 39.1 Staudacher, Thomas; Allègre, Claude J. (1982). "Terrestrial xenology" (in en). Earth and Planetary Science Letters 60 (3): 389–406. doi:10.1016/0012-821X(82)90075-9. Bibcode: 1982E&PSL..60..389S. https://linkinghub.elsevier.com/retrieve/pii/0012821X82900759.
- ↑ 40.0 40.1 Kunz, Joachim; Staudacher, Thomas; Allègre, Claude J. (1998-05-08). "Plutonium-Fission Xenon Found in Earth's Mantle" (in en). Science 280 (5365): 877–880. doi:10.1126/science.280.5365.877. ISSN 0036-8075. PMID 9572726. Bibcode: 1998Sci...280..877K. https://www.science.org/doi/10.1126/science.280.5365.877.
- ↑ Marty, Bernard (1989). "Neon and xenon isotopes in MORB: implications for the earth-atmosphere evolution" (in en). Earth and Planetary Science Letters 94 (1–2): 45–56. doi:10.1016/0012-821X(89)90082-4. Bibcode: 1989E&PSL..94...45M. https://linkinghub.elsevier.com/retrieve/pii/0012821X89900824.
- ↑ 42.0 42.1 Broadley, Michael W.; Barry, Peter H.; Bekaert, David V.; Byrne, David J.; Caracausi, Antonio; Ballentine, Christopher J.; Marty, Bernard (2020-06-23). "Identification of chondritic krypton and xenon in Yellowstone gases and the timing of terrestrial volatile accretion" (in en). Proceedings of the National Academy of Sciences 117 (25): 13997–14004. doi:10.1073/pnas.2003907117. ISSN 0027-8424. PMID 32513744. Bibcode: 2020PNAS..11713997B.
- ↑ Trieloff, Mario; Kunz, Joachim; Allègre, Claude J. (2002). "Noble gas systematics of the Réunion mantle plume source and the origin of primordial noble gases in Earth's mantle" (in en). Earth and Planetary Science Letters 200 (3–4): 297–313. doi:10.1016/S0012-821X(02)00639-8. Bibcode: 2002E&PSL.200..297T. https://linkinghub.elsevier.com/retrieve/pii/S0012821X02006398.
- ↑ Trieloff, Mario; Kunz, Joachim; Clague, David A.; Harrison, Darrell; Allègre, Claude J. (2000-05-12). "The Nature of Pristine Noble Gases in Mantle Plumes" (in en). Science 288 (5468): 1036–1038. doi:10.1126/science.288.5468.1036. ISSN 0036-8075. PMID 10807571. Bibcode: 2000Sci...288.1036T. https://www.science.org/doi/10.1126/science.288.5468.1036.
- ↑ Allègre, Claude J.; Staudacher, Thomas; Sarda, Philippe; Kurz, Mark (1983). "Constraints on evolution of Earth's mantle from rare gas systematics" (in en). Nature 303 (5920): 762–766. doi:10.1038/303762a0. ISSN 0028-0836. Bibcode: 1983Natur.303..762A. http://www.nature.com/articles/303762a0.
- ↑ J. Allègre, Claude; Staudacher, Thomas; Sarda, Philippe (1987). "Rare gas systematics: formation of the atmosphere, evolution and structure of the Earth's mantle" (in en). Earth and Planetary Science Letters 81 (2–3): 127–150. doi:10.1016/0012-821X(87)90151-8. Bibcode: 1987E&PSL..81..127A. https://linkinghub.elsevier.com/retrieve/pii/0012821X87901518.
- ↑ Patterson, D. B.; Honda, M.; McDougall, I. (1990). "Atmospheric contamination: A possible source for heavy noble gases in basalts from Loihi Seamount, Hawaii" (in en). Geophysical Research Letters 17 (6): 705–708. doi:10.1029/GL017i006p00705. Bibcode: 1990GeoRL..17..705P. http://doi.wiley.com/10.1029/GL017i006p00705.
- ↑ Parai, Rita; Mukhopadhyay, Sujoy (2018). "Xenon isotopic constraints on the history of volatile recycling into the mantle" (in en). Nature 560 (7717): 223–227. doi:10.1038/s41586-018-0388-4. ISSN 0028-0836. PMID 30089920. Bibcode: 2018Natur.560..223P. http://www.nature.com/articles/s41586-018-0388-4.
- ↑ Conrad, P.G.; Malespin, C.A.; Franz, H.B.; Pepin, R.O.; Trainer, M.G.; Schwenzer, S.P.; Atreya, S.K.; Freissinet, C. et al. (2016). "In situ measurement of atmospheric krypton and xenon on Mars with Mars Science Laboratory" (in en). Earth and Planetary Science Letters 454: 1–9. doi:10.1016/j.epsl.2016.08.028. Bibcode: 2016E&PSL.454....1C. https://linkinghub.elsevier.com/retrieve/pii/S0012821X16304514.
- ↑ Cassata, William S. (2017). "Meteorite constraints on Martian atmospheric loss and paleoclimate" (in en). Earth and Planetary Science Letters 479: 322–329. doi:10.1016/j.epsl.2017.09.034. Bibcode: 2017E&PSL.479..322C.
- ↑ 51.0 51.1 Avice, Guillaume; Marty, Bernard (2020). "Perspectives on Atmospheric Evolution from Noble Gas and Nitrogen Isotopes on Earth, Mars & Venus" (in en). Space Science Reviews 216 (3): 36. doi:10.1007/s11214-020-00655-0. ISSN 0038-6308. Bibcode: 2020SSRv..216...36A. http://link.springer.com/10.1007/s11214-020-00655-0.
- ↑ Zahnle, KJ (2013). "Venus, Earth, Xenon". AGU Fall Meeting Abstracts 2013. Bibcode: 2013AGUFM.P34A..06Z. https://ui.adsabs.harvard.edu/abs/2013AGUFM.P34A..06Z.
- ↑ Owen, Tobias; Mahaffy, Paul; Niemann, H. B.; Atreya, Sushil; Donahue, Thomas; Bar-Nun, Akiva; de Pater, Imke (1999). "A low-temperature origin for the planetesimals that formed Jupiter" (in en). Nature 402 (6759): 269–270. doi:10.1038/46232. ISSN 0028-0836. PMID 10580497. Bibcode: 1999Natur.402..269O. http://www.nature.com/articles/46232.
 |
 KSF
KSF