Pathogenic bacteria
Topic: Medicine
 From HandWiki - Reading time: 24 min
From HandWiki - Reading time: 24 min
| Pathogenic bacteria | |
|---|---|
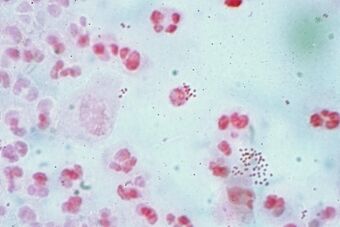 | |
| Neisseria gonorrhoeae (small red dots) in pus from a man with a urethral discharge (Gram stain) |
Pathogenic bacteria are bacteria that can cause disease.[1] This article focuses on the bacteria that are pathogenic to humans. Most species of bacteria are harmless and are often beneficial but others can cause infectious diseases. The number of these pathogenic species in humans is estimated to be fewer than a hundred.[2] By contrast, several thousand species are part of the gut flora present in the digestive tract.[citation needed]
The body is continually exposed to many species of bacteria, including beneficial commensals, which grow on the skin and mucous membranes, and saprophytes, which grow mainly in the soil and in decaying matter. The blood and tissue fluids contain nutrients sufficient to sustain the growth of many bacteria. The body has defence mechanisms that enable it to resist microbial invasion of its tissues and give it a natural immunity or innate resistance against many microorganisms.
Pathogenic bacteria are specially adapted and endowed with mechanisms for overcoming the normal body defences, and can invade parts of the body, such as the blood, where bacteria are not normally found. Some pathogens invade only the surface epithelium, skin or mucous membrane, but many travel more deeply, spreading through the tissues and disseminating by the lymphatic and blood streams. In some rare cases a pathogenic microbe can infect an entirely healthy person, but infection usually occurs only if the body's defence mechanisms are damaged by some local trauma or an underlying debilitating disease, such as wounding, intoxication, chilling, fatigue, and malnutrition. In many cases, it is important to differentiate infection and colonization, which is when the bacteria are causing little or no harm.
Caused by Mycobacterium tuberculosis bacteria, one of the diseases with the highest disease burden is tuberculosis, which killed 1.4 million people in 2019, mostly in sub-Saharan Africa.[4] Pathogenic bacteria contribute to other globally important diseases, such as pneumonia, which can be caused by bacteria such as Staphylococcus, Streptococcus and Pseudomonas, and foodborne illnesses, which can be caused by bacteria such as Shigella, Campylobacter, and Salmonella. Pathogenic bacteria also cause infections such as tetanus, typhoid fever, diphtheria, syphilis, and leprosy.
Pathogenic bacteria are also the cause of high infant mortality rates in developing countries.[5] A GBD study estimated the global death rates from (33) bacterial pathogens, finding such infections contributed to one in 8 deaths (or ~7.7 million deaths), which could make it the second largest cause of death globally in 2019.[6][3]
Most pathogenic bacteria can be grown in cultures and identified by Gram stain and other methods. Bacteria grown in this way are often tested to find which antibiotics will be an effective treatment for the infection. For hitherto unknown pathogens, Koch's postulates are the standard to establish a causative relationship between a microbe and a disease.
Diseases
Each species has specific effect and causes symptoms in people who are infected. Some people who are infected with a pathogenic bacteria do not have symptoms. Immunocompromised individuals are more susceptible to pathogenic bacteria.[7]
Pathogenic susceptibility
Some pathogenic bacteria cause disease under certain conditions, such as entry through the skin via a cut, through sexual activity or through compromised immune function.[citation needed]
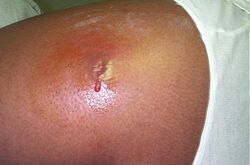
Some species of Streptococcus and Staphylococcus are part of the normal skin microbiota and typically reside on healthy skin or in the nasopharyngeal region. Yet these species can potentially initiate skin infections. Streptococcal infections include sepsis, pneumonia, and meningitis.[8] These infections can become serious creating a systemic inflammatory response resulting in massive vasodilation, shock, and death.[9]
Other bacteria are opportunistic pathogens and cause disease mainly in people with immunosuppression or cystic fibrosis. Examples of these opportunistic pathogens include Pseudomonas aeruginosa, Burkholderia cenocepacia, and Mycobacterium avium.[10][11]
Intracellular
Obligate intracellular parasites (e.g. Chlamydophila, Ehrlichia, Rickettsia) have the ability to only grow and replicate inside other cells. Even these intracellular infections may be asymptomatic, requiring an incubation period. An example of this is Rickettsia which causes typhus. Another causes Rocky Mountain spotted fever.[citation needed]
Chlamydia are intracellular parasites. These pathogens can cause pneumonia or urinary tract infection and may be involved in coronary heart disease.[12]
Other groups of intracellular bacterial pathogens include Salmonella, Neisseria, Brucella, Mycobacterium, Nocardia, Listeria, Francisella, Legionella, and Yersinia pestis. These can exist intracellularly, but can exist outside host cells.[citation needed]
Infections in specific tissue
Bacterial pathogens often cause infection in specific areas of the body. Others are generalists.
- Bacterial vaginosis is a condition of the vaginal microbiota in which an excessive growth of Gardnerella vaginalis and other mostly anaerobic bacteria displace the beneficial Lactobacilli species that maintain healthy vaginal microbial populations.[13]
- Bacterial meningitis is a bacterial inflammation of the meninges, which are the protective membranes covering the brain and spinal cord.
- Bacterial pneumonia is a bacterial infection of the lungs.
- Urinary tract infection is predominantly caused by bacteria. Symptoms include the strong and frequent sensation or urge to urinate, pain during urination, and urine that is cloudy.[14] The most frequent cause is Escherichia coli. Urine is typically sterile but contains a variety of salts, and waste products Bacteria can ascend into the bladder or kidney and causing cystitis and nephritis.[15][16]
- Bacterial gastroenteritis is caused by enteric, pathogenic bacteria. These pathogenic species are usually distinct from the usually harmless bacteria of the normal gut flora. But a different strain of the same species may be pathogenic. The distinction is sometimes difficult as in the case of Escherichia.
- Bacterial skin infections include:
- Impetigo is a highly contagious bacterial skin infection commonly seen in children.[17] It is caused by Staphylococcus aureus, and Streptococcus pyogenes.[18]
- Erysipelas is an acute streptococcus bacterial infection[19] of the deeper skin layers that spreads via with lymphatic system.
- Cellulitis is a diffuse inflammation[20] of connective tissue with severe inflammation of dermal and subcutaneous layers of the skin. Cellulitis can be caused by normal skin flora or by contagious contact, and usually occurs through open skin, cuts, blisters, cracks in the skin, insect bites, animal bites, burns, surgical wounds, intravenous drug injection, or sites of intravenous catheter insertion. In most cases it is the skin on the face or lower legs that is affected, though cellulitis can occur in other tissues.
Mechanisms of damage
The symptoms of disease appear as pathogenic bacteria damage host tissues or interfere with their function. The bacteria can damage host cells directly or indirectly by provoking an immune response that inadvertently damages host cells,[21] or by releasing toxins.[22]
Direct
Once pathogens attach to host cells, they can cause direct damage as the pathogens use the host cell for nutrients and produce waste products.[23] For example, Streptococcus mutans, a component of dental plaque, metabolizes dietary sugar and produces acid as a waste product. The acid decalcifies the tooth surface to cause dental caries.[24]
Toxin production
Endotoxins are the lipid portions of lipopolysaccharides that are part of the outer membrane of the cell wall of gram-negative bacteria. Endotoxins are released when the bacteria lyses, which is why after antibiotic treatment, symptoms can worsen at first as the bacteria are killed and they release their endotoxins. Exotoxins are secreted into the surrounding medium or released when the bacteria die and the cell wall breaks apart.[25]
Indirect
An excessive or inappropriate immune response triggered by an infection may damage host cells.[1]
Survival in host
Nutrients
Iron is required for humans, as well as the growth of most bacteria. To obtain free iron, some pathogens secrete proteins called siderophores, which take the iron away from iron-transport proteins by binding to the iron even more tightly. Once the iron-siderophore complex is formed, it is taken up by siderophore receptors on the bacterial surface and then that iron is brought into the bacterium.[25]
Bacterial pathogens also require access to carbon and energy sources for growth. To avoid competition with host cells for glucose which is the main energy source used by human cells, many pathogens including the respiratory pathogen Haemophilus influenzae specialise in using other carbon sources such as lactate that are abundant in the human body [26]
Identification

Typically identification is done by growing the organism in a wide range of cultures which can take up to 48 hours. The growth is then visually or genomically identified. The cultured organism is then subjected to various assays to observe reactions to help further identify species and strain.[27]
Treatment
Bacterial infections may be treated with antibiotics, which are classified as bacteriocidal if they kill bacteria or bacteriostatic if they just prevent bacterial growth. There are many types of antibiotics and each class inhibits a process that is different in the pathogen from that found in the host. For example, the antibiotics chloramphenicol and tetracyclin inhibit the bacterial ribosome but not the structurally different eukaryotic ribosome, so they exhibit selective toxicity.[28] Antibiotics are used both in treating human disease and in intensive farming to promote animal growth. Both uses may be contributing to the rapid development of antibiotic resistance in bacterial populations.[29] Phage therapy, using bacteriophages can also be used to treat certain bacterial infections.[30]
Prevention
Infections can be prevented by antiseptic measures such as sterilizing the skin prior to piercing it with the needle of a syringe and by proper care of indwelling catheters. Surgical and dental instruments are also sterilized to prevent infection by bacteria. Disinfectants such as bleach are used to kill bacteria or other pathogens on surfaces to prevent contamination and further reduce the risk of infection. Bacteria in food are killed by cooking to temperatures above 73 °C (163 °F).[citation needed]
List of genera and microscopy features
Many genera contain pathogenic bacterial species. They often possess characteristics that help to classify and organize them into groups. The following is a partial listing.
| Genus | Species | Gram staining | Shape | Oxygen requirement | Intra/Extracellular |
|---|---|---|---|---|---|
| Bacillus[31] | Positive | Rods | Facultative anaerobic | Extracellular | |
| Bartonella[31] | Negative | Rods | Aerobic | Facultative intracellular | |
| Bordetella[31] | Negative | Small coccobacilli | Aerobic | Extracellular | |
| Borrelia[31] | Negative, stains poorly | Spirochete | Anaerobic | Extracellular | |
| Brucella[31] | Negative | Coccobacilli | Aerobic | Intracellular | |
| Campylobacter[31] | Negative | Spiral rods[34] coccoid in older cultures[34] |
Microaerophilic[34] | Extracellular | |
| Chlamydia and Chlamydophila[31] |
|
(not Gram-stained) | Small, round, ovoid | Facultative or strictly aerobic | Obligate intracellular |
| Clostridium[31] |
|
Positive | Large, blunt-ended rods | Obligate anaerobic | Extracellular |
| Corynebacterium[31] | Positive (unevenly) | Rods | Mostly facultative anaerobic | Extracellular | |
| Enterococcus[33][37] | Positive | Cocci | Facultative Anaerobic | Extracellular | |
| Escherichia[5][33][38] | Negative | Rods | Facultative anaerobic | Extracellular or Intracellular | |
| Francisella[31] | Negative | Coccobacillus | Strictly aerobic | Facultative intracellular | |
| Haemophilus | Negative | Coccobacilli to long and slender filaments | Facultative anaerobic 5 - 10% CO2 | Extracellular | |
| Helicobacter |
|
Negative | Spiral rod | Microaerophile | Extracellular |
| Legionella[31] | Negative, stains poorly | Cocobacilli | Aerobic | Facultative intracellular | |
| Leptospira[33][41] | Negative, stains poorly | Spirochete | Strictly aerobic | Extracellular | |
| Listeria[31] | Positive, darkly | Slender, short rods | Facultative Anaerobic | Facultative intracellular | |
| Mycobacterium[31] |
|
(none) | Long, slender rods | Aerobic | Intracellular |
| Mycoplasma[31] | (none) | Indistinct 'fried egg' appearance, no cell wall | Mostly facultative anaerobic; M. pneumoniae strictly aerobic | Extracellular | |
| Neisseria[33][42] | Negative | Kidney bean-shaped | Aerobic | Gonococcus: facultative intracellular N. meningitidis: extracellular | |
| Pseudomonas[33][43] | Negative | Rods | Obligate aerobic | Extracellular | |
| Rickettsia[31] | Negative, stains poorly | Small, rod-like coccobacillary | Aerobic | Obligate intracellular | |
| Salmonella[31] |
|
Negative | Rods | Facultative anaerobica | Facultative intracellular |
| Shigella[33][44] | Negative | Rods | Facultative anaerobic | Extracellular | |
| Staphylococcus[5] | Positive, darkly | Round cocci | Facultative anaerobic | Extracellular, facultative intracellular | |
| Streptococcus[31] | Positive | Ovoid to spherical | Facultative anaerobic | Extracellular | |
| Treponema[31] | Negative, stains poorly | Spirochete | Aerobic | Extracellular | |
| Ureaplasma[5] | Stains poorly[45] | Indistinct, 'fried egg' appearance, no cell wall | Anaerobic | Extracellular | |
| Vibrio[33][46] | Negative | Spiral with single polar flagellum | Facultative anaerobic | Extracellular | |
| Yersinia[33][47] | Negative, bipolarly | Small rods | Facultative anaerobe | Intracellular |
List of species and clinical characteristics
-
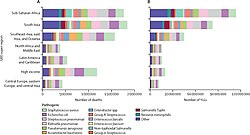
Overall age-standardised mortality rate per 100 000 population for 33 pathogens investigated, 2019[3]
-
Global number of deaths (A) and YLLs (B), by pathogen and infectious syndrome, 2019[3]
-
Global number of deaths, by pathogen, age, and sex groups, 2019[3]
This is description of the more common genera and species presented with their clinical characteristics and treatments.
| Species | Transmission | Diseases | Treatment | Prevention | ||
|---|---|---|---|---|---|---|
| Actinomyces israelii | Oral flora[48] | Actinomycosis:[48] painful abscesses and cysts MRSA in the mouth, lungs,[49][50] or gastrointestinal tract.[35] | Prolonged penicillin G and drainage[48] | |||
| Bacillus anthracis |
Contact with cattle, sheep, goats and horses[51] |
Anthrax: pulmonary, gastrointestinal and/or cutaneous symptoms.[48] |
In early infection:[52] |
Anthrax vaccine[33] | ||
| Bacteroides fragilis | Gut flora[48] | Abscesses in gastrointestinal tract, pelvic cavity and lungs[48] | metronidazole[48] | Wound care[54] | ||
| Bordetella pertussis |
Contact with respiratory droplets expelled by infected human hosts.[33] |
Macrolides[33] such as erythromycin,[33][48] before paroxysmal stage[48] |
Pertussis vaccine,[33][48] such as in DPT vaccine[33][48] | |||
| Borrelia | B. burgdorferi[33][48] |
Ixodes hard ticks |
|
Doxycycline for adults, amoxicillin for children, ceftriaxone for neurological involvement[56] |
Wearing clothing that limits skin exposure to ticks.[33] | |
| B. recurrentis[58] and others[note 1] |
Pediculus humanus corporis body louse (B. recurrentis only) and Ornithodoros soft ticks[58] | Relapsing fever | Penicillin, tetracycline, doxycycline[59] | Avoid areas where ticks are found[58] Better access to washing facilities[58] | ||
| Brucella | B. abortus |
Direct contact with infected animal[33] |
Brucellosis: mainly fever, muscular pain and night sweats |
|||
| Campylobacter jejuni |
Fecal–oral from animals (mammals and fowl)[33][48] |
|
Treat symptoms[33] |
Good hygiene[33] | ||
| Chlamydia | C. pneumoniae | Atypical pneumonia[48] | None[33] | |||
| C. trachomatis |
vaginal sex[33] |
Trachoma[33][48] |
Erythromycin[33][48] |
Erythromycin or silver nitrate in newborn's eyes[33] | ||
| Chlamydophila psittaci | Inhalation of dust with secretions or feces from birds (e.g. parrots) | Psittacosis, mainly atypical pneumonia | - | |||
| Clostridium | C. botulinum | Spores from soil,[33][48] persevere in canned food, smoked fish and honey[48] |
Botulism: Mainly muscle weakness and paralysis[48] |
Antitoxin[33][48] |
Proper food preservation techniques | |
| C. difficile |
Gut flora,[33][48] overgrowing when other flora is depleted[33] |
Discontinuing responsible antibiotic[33][48] |
Fecal bacteriotherapy | |||
| C. perfringens |
Spores in soil[33][48] |
Anaerobic cellulitis[33][48] |
Gas gangrene:
Debridement or amputation[33][48] |
Appropriate food handling[33] | ||
| C. tetani |
Tetanus immune globulin[33][48]
Sedatives[33] |
Tetanus vaccine (such as in the DPT vaccine)[33] | ||||
| Corynebacterium diphtheriae |
respiratory droplets |
Diphtheria: Fever, sore throat and neck swelling, potentially narrowing airways.[61] |
Horse serum antitoxin |
|||
| Ehrlichia | E. canis[48] |
Dog tick[48] | Ehrlichiosis:[48] headache, muscle aches, and fatigue |
| ||
| Enterococcus | E. faecalis |
Part of gut flora,[48] opportunistic or entering through GI tract or urinary system wounds[33] |
Bacterial endocarditis,[48] biliary tract infections,[48] urinary tract infections[48] |
Ampicillin (combined with aminoglycoside in endocarditis)[48] Vancomycin[33] |
No vaccine Hand washing and other nosocomial prevention | |
| Escherichia | E. coli (generally) | UTI:[33]
(resistance-tests are required first)
Meningitis:[33]
Diarrhea:[33]
|
(no vaccine or preventive drug)[33] | |||
| Enterotoxigenic E. coli (ETEC) |
|
|||||
| Enteropathogenic E. coli |
|
| ||||
| Enteroinvasive E.coli (EIEC) |
| |||||
| Enterohemorrhagic (EHEC), including E. coli O157:H7 |
|
|||||
| Francisella tularensis | Tularemia: Fever, ulceration at entry site and/or lymphadenopathy.[63] Can cause severe pneumonia.[63] | |||||
| Haemophilus influenzae |
|
Meningitis:[33]
(resistance-tests are required first)
|
| |||
| Helicobacter pylori |
|
|
(No vaccine or preventive drug)[33] | |||
| Klebsiella pneumoniae |
|
|
|
|||
| Legionella pneumophila |
|
|
(no vaccine or preventive drug)[33]
Heating water[33] | |||
| Leptospira species |
|
|
|
Vaccine not widely used[33]
Prevention of exposure[33]
| ||
| Listeria monocytogenes |
|
(no vaccine)[33]
| ||||
| Mycobacterium | M. leprae |
|
|
Tuberculoid form:
Lepromatous form: |
| |
| M. tuberculosis |
|
|
(difficult, see Tuberculosis treatment for more details)[33] Standard "short" course:[33]
|
| ||
| Mycoplasma pneumoniae | ||||||
| Neisseria | N. gonorrhoeae |
|
Uncomplicated gonorrhea:[33]
Ophthalmia neonatorum: |
(No vaccine)[33]
| ||
| N. meningitidis |
|
|||||
| Pseudomonas aeruginosa | Opportunistic;[48] Infects damaged tissues or people with immunodeficiency.[33] | Pseudomonas infection:[33] |
|
(no vaccine)[33]
| ||
| Nocardia asteroides | In soil[48] | Nocardiosis:[48] Pneumonia, endocarditis, keratitis, neurological or lymphocutaneous infection | TMP/SMX[48] | |||
| Rickettsia rickettsii | (no preventive drug or approved vaccine)[33]
| |||||
| Salmonella | S typhi |
|
|
|
||
| Other Salmonella species
|
|
(No vaccine or preventive drug)[33] | ||||
| Shigella | S. sonnei[33] |
|
|
|||
| Staphylococcus | aureus | Coagulase-positive staphylococcal infections:
|
|
(no vaccine or preventive drug)
| ||
| epidermidis | Human flora in skin,[33][48] anterior nares[33] and mucous membranes[48] |
|
None[33] | |||
| saprophyticus | Part of normal vaginal flora[33] |
|
None[33] | |||
| Streptococcus | agalactiae | Human flora in vagina,[33][48] urethral mucous membranes,[33] rectum[33]
|
|
None[33] | ||
| pneumoniae |
|
|||||
| pyogenes |
|
|
No vaccine[33]
| |||
| viridans | Oral flora,[48] penetration through abrasions |
|
Penicillin G[48] | |||
| Treponema pallidum subspecies pallidum |
|
|||||
| Vibrio cholerae |
|
|
| |||
| Yersinia pestis | Plague: |
|
||||
Genetic transformation
Of the 59 species listed in the table with their clinical characteristics, 11 species (or 19%) are known to be capable of natural genetic transformation.[81] Natural transformation is a bacterial adaptation for transferring DNA from one cell to another. This process includes the uptake of exogenous DNA from a donor cell by a recipient cell and its incorporation into the recipient cell's genome by recombination. Transformation appears to be an adaptation for repairing damage in the recipient cell's DNA. Among pathogenic bacteria, transformation capability likely serves as an adaptation that facilitates survival and infectivity.[81] The pathogenic bacteria able to carry out natural genetic transformation (of those listed in the table) are Campylobacter jejuni, Enterococcus faecalis, Haemophilus influenzae, Helicobacter pylori, Klebsiella pneumoniae, Legionella pneumophila, Neisseria gonorrhoeae, Neisseria meningitidis, Staphylococcus aureus, Streptococcus pneumoniae and Vibrio cholerae.[citation needed]
See also
- Human microbiome project
- List of antibiotics
- Pathogenic viruses
Notes
- ↑ Relapsing fever can also be caused by the following Borrelia species: B. crocidurae, B. duttonii, B. hermsii, B. hispanica, B. miyamotoi, B. persica, B. turicatae and B. venezuelensis.
- Barbour, Alan G. (2017). "Relapsing Fever". in Kasper, Dennis L.; Fauci, Anthony S.. Harrison's Infectious Diseases (3rd ed.). New York: McGraw Hill Education. pp. 678–687. ISBN 978-1-259-83597-1.
References
- ↑ 1.0 1.1 Ryan, Kenneth J.; Ray, C. George; Ahmad, Nafees; Drew, W. Lawrence; Lagunoff, Michael; Pottinger, Paul; Reller, L. Barth; Sterling, Charles R. (2014). "Pathogenesis of Bacterial Infections". Sherris Medical Microbiology (6th ed.). New York: McGraw Hill Education. pp. 391–406. ISBN 978-0-07-181826-1.
- ↑ McFall-Ngai, Margaret (2007-01-11). "Adaptive Immunity: Care for the community" (in en). Nature 445 (7124): 153. doi:10.1038/445153a. ISSN 0028-0836. PMID 17215830. Bibcode: 2007Natur.445..153M.
- ↑ 3.0 3.1 3.2 3.3 3.4 Ikuta, Kevin S.Expression error: Unrecognized word "et". (21 November 2022). "Global mortality associated with 33 bacterial pathogens in 2019: a systematic analysis for the Global Burden of Disease Study 2019" (in English). The Lancet 400 (10369): 2221–2248. doi:10.1016/S0140-6736(22)02185-7. ISSN 0140-6736. PMID 36423648.
- ↑ "Tuberculosis (TB)" (in en). https://www.who.int/en/news-room/fact-sheets/detail/tuberculosis.
- ↑ 5.0 5.1 5.2 5.3 Santosham, Mathuram; Chan, Grace J.; Lee, Anne CC; Baqui, Abdullah H.; Tan, Jingwen; Black, Robert E. (2013). "Risk of Early-Onset Neonatal Infection with Maternal Infection or Colonization: A Global Systematic Review and Meta-Analysis". PLOS Medicine 10 (8): e1001502. doi:10.1371/journal.pmed.1001502. ISSN 1549-1676. PMID 23976885.
- ↑ Hou, Chia-Yi (23 November 2022). "Bacterial infections linked to 1 in 8 deaths in 2019". The Hill. https://thehill.com/changing-america/well-being/prevention-cures/3748033-bacterial-infections-linked-to-1-in-8-deaths-in-2019/.
- ↑ "Diagnosis of severe respiratory infections in immunocompromised patients". Intensive Care Medicine 46 (2): 298–314. February 2020. doi:10.1007/s00134-019-05906-5. PMID 32034433.
- ↑ "Streptococcal Infections - Infectious Diseases" (in en). https://www.msdmanuals.com/professional/infectious-diseases/gram-positive-cocci/streptococcal-infections.
- ↑ Fish DN (February 2002). "Optimal antimicrobial therapy for sepsis". Am J Health Syst Pharm 59 (Suppl 1): S13–9. doi:10.1093/ajhp/59.suppl_1.S13. PMID 11885408. http://www.ajhp.org/cgi/pmidlookup?view=long&pmid=11885408.
- ↑ Heise E (1982). "Diseases associated with immunosuppression". Environ Health Perspect 43: 9–19. doi:10.2307/3429162. PMID 7037390.
- ↑ Saiman L (2004). "Microbiology of early CF lung disease". Paediatr Respir Rev 5 (Suppl A): S367–9. doi:10.1016/S1526-0542(04)90065-6. PMID 14980298.
- ↑ "Chlamydia pneumoniae and atherosclerosis". Cell Microbiol 6 (2): 117–27. 2004. doi:10.1046/j.1462-5822.2003.00352.x. PMID 14706098.
- ↑ "Pathogenesis of Bacterial Vaginosis: Discussion of Current Hypotheses". The Journal of Infectious Diseases 214 (Suppl 1): S1–5. August 2016. doi:10.1093/infdis/jiw121. PMID 27449868.
- ↑ "Urinary Tract Infections". http://www.medicinenet.com/urine_infection/page4.htm.
- ↑ Roxe DM. Urinalysis. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Chapter 191. Available from: https://www.ncbi.nlm.nih.gov/books/NBK302/
- ↑ "The challenge of urinary tract infections in renal transplant recipients". Transplant Infectious Disease 20 (2): e12828. April 2018. doi:10.1111/tid.12828. PMID 29272071.
- ↑ "Impetigo". 19 October 2017. http://www.nhs.uk/conditions/Impetigo/Pages/Introduction.aspx. Page last reviewed: 17/07/2014
- ↑ Kumar, Vinay; Abbas, Abul K.; Fausto, Nelson; & Mitchell, Richard N. (2007). Robbins Basic Pathology (8th ed.). Saunders Elsevier. pp. 843 ISBN 978-1-4160-2973-1
- ↑ "erysipelas" at Dorland's Medical Dictionary
- ↑ "cellulitis" at Dorland's Medical Dictionary
- ↑ Greenwood, David; Barer, Mike; Slack, Richard; Irving, Will (2012). "Bacterial Pathogenicity". Medical Microbiology, a Guide to Microbial Infections: Pathogenesis, Immunity, Laboratory Investigation, and Control (18th ed.). Edinburgh: Churchill Livingstone. pp. 156–167. ISBN 9780702040894.
- ↑ "Bacterial toxins: Offensive, defensive, or something else altogether?". PLOS Pathogens 13 (9): e1006452. September 2017. doi:10.1371/journal.ppat.1006452. PMID 28934339.
- ↑ Tortora, Gerald J.; Funke, Berdell R.; Case, Christine L. (2016). "Microbial Mechanisms of Pathogenicity". Microbiology, an Introduction (12th ed.). Pearson Education. pp. 417–438. ISBN 978-0-321-92915-0.
- ↑ Nash, Anthony A.; Dalziel, Robert G.; Fitzgerald, J. Ross (2015). "Mechanisms of Cell and Tissue Damage". Mims' Pathogenesis of Infectious Disease (6th ed.). London: Academic Press. pp. 171–231. ISBN 978-0-12-397188-3.
- ↑ 25.0 25.1 Tortota, Gerard (2013). Microbiology an Introduction. Pearson. ISBN 978-0-321-73360-3.
- ↑ Hosmer, Jennifer; Nasreen, Marufa; Dhouib, Rabeb; Essilfie, Ama-Tawiah; Schirra, Horst Joachim; Henningham, Anna; Fantino, Emmanuelle; Sly, Peter et al. (2022-01-27). "Access to highly specialized growth substrates and production of epithelial immunomodulatory metabolites determine survival of Haemophilus influenzae in human airway epithelial cells" (in en). PLOS Pathogens 18 (1): e1010209. doi:10.1371/journal.ppat.1010209. ISSN 1553-7374. PMID 35085362.
- ↑ "Pathogen and Biological Contamination Management in Plant Tissue Culture: Phytopathogens, Vitro Pathogens, and Vitro Pests". Plant Cell Culture Protocols. Methods in Molecular Biology. 877. 2012. pp. 57–80. doi:10.1007/978-1-61779-818-4_6. ISBN 978-1-61779-817-7.
- ↑ "Ribosomal crystallography: initiation, peptide bond formation, and amino acid polymerization are hampered by antibiotics". Annu Rev Microbiol 58: 233–51. 2004. doi:10.1146/annurev.micro.58.030603.123822. PMID 15487937.
- ↑ Khachatourians GG (November 1998). "Agricultural use of antibiotics and the evolution and transfer of antibiotic-resistant bacteria". CMAJ 159 (9): 1129–36. PMID 9835883. PMC 1229782. http://www.cmaj.ca/cgi/pmidlookup?view=reprint&pmid=9835883.
- ↑ Keen, E. C. (2012). "Phage Therapy: Concept to Cure". Frontiers in Microbiology 3: 238. doi:10.3389/fmicb.2012.00238. PMID 22833738.
- ↑ 31.00 31.01 31.02 31.03 31.04 31.05 31.06 31.07 31.08 31.09 31.10 31.11 31.12 31.13 31.14 31.15 31.16 31.17 Unless else specified in boxes then ref is: Fisher, Bruce; Harvey, Richard P.; Champe, Pamela C. (2007). Lippincott's Illustrated Reviews: Microbiology (Lippincott's Illustrated Reviews Series). Hagerstown, MD: Lippincott Williams & Wilkins. pp. 332–353. ISBN 978-0-7817-8215-9.
- ↑ "Comparison of modified Bordet-Gengou and modified Regan-Lowe media for the isolation of Bordetella pertussis and Bordetella parapertussis". J. Clin. Microbiol. 26 (12): 2661–3. 1988. doi:10.1128/JCM.26.12.2661-2663.1988. PMID 2906642.
- ↑ 33.000 33.001 33.002 33.003 33.004 33.005 33.006 33.007 33.008 33.009 33.010 33.011 33.012 33.013 33.014 33.015 33.016 33.017 33.018 33.019 33.020 33.021 33.022 33.023 33.024 33.025 33.026 33.027 33.028 33.029 33.030 33.031 33.032 33.033 33.034 33.035 33.036 33.037 33.038 33.039 33.040 33.041 33.042 33.043 33.044 33.045 33.046 33.047 33.048 33.049 33.050 33.051 33.052 33.053 33.054 33.055 33.056 33.057 33.058 33.059 33.060 33.061 33.062 33.063 33.064 33.065 33.066 33.067 33.068 33.069 33.070 33.071 33.072 33.073 33.074 33.075 33.076 33.077 33.078 33.079 33.080 33.081 33.082 33.083 33.084 33.085 33.086 33.087 33.088 33.089 33.090 33.091 33.092 33.093 33.094 33.095 33.096 33.097 33.098 33.099 33.100 33.101 33.102 33.103 33.104 33.105 33.106 33.107 33.108 33.109 33.110 33.111 33.112 33.113 33.114 33.115 33.116 33.117 33.118 33.119 33.120 33.121 33.122 33.123 33.124 33.125 33.126 33.127 33.128 33.129 33.130 33.131 33.132 33.133 33.134 33.135 33.136 33.137 33.138 33.139 33.140 33.141 33.142 33.143 33.144 33.145 33.146 33.147 33.148 33.149 33.150 33.151 33.152 33.153 33.154 33.155 33.156 33.157 33.158 33.159 33.160 33.161 33.162 33.163 33.164 33.165 33.166 33.167 33.168 33.169 33.170 33.171 33.172 33.173 33.174 33.175 33.176 33.177 33.178 33.179 33.180 33.181 33.182 33.183 33.184 33.185 33.186 33.187 33.188 33.189 33.190 33.191 33.192 33.193 33.194 33.195 33.196 33.197 33.198 33.199 33.200 33.201 33.202 33.203 33.204 33.205 33.206 33.207 33.208 33.209 33.210 33.211 33.212 33.213 33.214 33.215 33.216 33.217 33.218 33.219 33.220 33.221 33.222 33.223 33.224 33.225 33.226 33.227 33.228 33.229 33.230 33.231 33.232 33.233 33.234 33.235 33.236 33.237 33.238 33.239 33.240 33.241 33.242 33.243 33.244 33.245 33.246 33.247 33.248 33.249 33.250 33.251 33.252 33.253 33.254 33.255 33.256 33.257 33.258 33.259 33.260 33.261 33.262 33.263 33.264 33.265 33.266 33.267 33.268 33.269 33.270 33.271 33.272 33.273 33.274 33.275 33.276 33.277 33.278 33.279 33.280 33.281 33.282 33.283 33.284 33.285 33.286 33.287 33.288 33.289 33.290 33.291 33.292 33.293 33.294 33.295 33.296 33.297 33.298 33.299 33.300 33.301 33.302 33.303 Fisher, Bruce; Harvey, Richard P.; Champe, Pamela C. (2007). Lippincott's Illustrated Reviews: Microbiology (Lippincott's Illustrated Reviews Series). Hagerstown, MD: Lippincott Williams & Wilkins. pp. 332–353. ISBN 978-0-7817-8215-9.
- ↑ 34.0 34.1 34.2 "Foodborne Campylobacter: infections, metabolism, pathogenesis and reservoirs". International Journal of Environmental Research and Public Health 10 (12): 6292–304. 2013. doi:10.3390/ijerph10126292. PMID 24287853.
- ↑ 35.0 35.1 Bowden GHW (1996). Baron S. ed. Actinomycosis in: Baron's Medical Microbiology (4th ed.). Univ of Texas Medical Branch. (via NCBI Bookshelf). ISBN 978-0-9631172-1-2.
- ↑ Baron, Samuel (1996). Medical Microbiology (4th ed.). University of Texas Medical Branch at Galveston, Galveston, Texas. ISBN 978-0-9631172-1-2.
- ↑ Rollins, David M. (2000). "BSCI424 Laboratory Media". University of Maryland. http://www.life.umd.edu/classroom/bsci424/LabMaterialsMethods/BSCI424Media.htm.
- ↑ Cain, Donna (January 14, 2015). "MacConkey Agar (CCCCD Microbiology". Collin College. http://iws2.collin.edu/dcain/CCCCD%20Micro/macconkeyagar.htm.
- ↑ "Chocolate agar, a differential medium for gram-positive cocci". Journal of Clinical Microbiology 20 (4): 822–3. 1984. doi:10.1128/JCM.20.4.822-823.1984. PMID 6490866.
- ↑ "Growth of Helicobacter pylori in various liquid and plating media". Lett. Appl. Microbiol. 30 (3): 192–6. 2000. doi:10.1046/j.1472-765x.2000.00699.x. PMID 10747249.
- ↑ "Differentiation of Pathogenic and Saprophytic Leptospires I. Growth at Low Temperatures". J. Bacteriol. 94 (1): 27–31. 1967. doi:10.1128/JB.94.1.27-31.1967. PMID 6027998.
- ↑ "Thayer Martin Agar (Modified) Procedure". University of Nebraska-Medical Center, Clinical Laboratory Science Program. http://webmedia.unmc.edu/alliedhealth/CLS/CLS418%2008/Thayer%20Martin%20Agar%20Procedure%2008.pdf.
- ↑ Allen, Mary E. (2005). "MacConkey Agar Plates Protocols". American Society for Microbiology. http://www.microbelibrary.org/component/resource/laboratory-test/2855-macconkey-agar-plates-protocols. Created: 30 September 2005. Last update: 01 April 2013
- ↑ "Hektoen Enteric Agar". Austin Community College District. http://www.austincc.edu/microbugz/hektoen_enteric_agar.php.
- ↑ "Association of Ureaplasma urealyticum infection of the lower respiratory tract with chronic lung disease and death in very-low-birth-weight infants". Lancet 2 (8605): 240–5. 1988. doi:10.1016/s0140-6736(88)92536-6. PMID 2899235.
- ↑ Pfeffer, C.; Oliver, J.D. (2003). "A comparison of thiosulphate-citrate-bile salts-sucrose (TCBS) agar and thiosulphate-chloride-iodide (TCI) agar for the isolation of Vibrio species from estuarine environments". Letters in Applied Microbiology 36 (3): 150–151. doi:10.1046/j.1472-765X.2003.01280.x. PMID 12581373.
- ↑ "Yersinia pestis". Wadsworth Center. 2006. http://www.health.ny.gov/guidance/oph/wadsworth/yersinia_pestis.pdf.
- ↑ 48.000 48.001 48.002 48.003 48.004 48.005 48.006 48.007 48.008 48.009 48.010 48.011 48.012 48.013 48.014 48.015 48.016 48.017 48.018 48.019 48.020 48.021 48.022 48.023 48.024 48.025 48.026 48.027 48.028 48.029 48.030 48.031 48.032 48.033 48.034 48.035 48.036 48.037 48.038 48.039 48.040 48.041 48.042 48.043 48.044 48.045 48.046 48.047 48.048 48.049 48.050 48.051 48.052 48.053 48.054 48.055 48.056 48.057 48.058 48.059 48.060 48.061 48.062 48.063 48.064 48.065 48.066 48.067 48.068 48.069 48.070 48.071 48.072 48.073 48.074 48.075 48.076 48.077 48.078 48.079 48.080 48.081 48.082 48.083 48.084 48.085 48.086 48.087 48.088 48.089 48.090 48.091 48.092 48.093 48.094 48.095 48.096 48.097 48.098 48.099 48.100 48.101 48.102 48.103 48.104 48.105 48.106 48.107 48.108 48.109 48.110 48.111 48.112 48.113 48.114 48.115 48.116 48.117 48.118 48.119 48.120 48.121 48.122 48.123 48.124 48.125 48.126 48.127 48.128 48.129 48.130 48.131 48.132 48.133 48.134 48.135 48.136 48.137 48.138 48.139 48.140 48.141 48.142 48.143 48.144 48.145 48.146 48.147 48.148 48.149 48.150 48.151 48.152 48.153 48.154 48.155 48.156 48.157 48.158 48.159 48.160 48.161 48.162 48.163 48.164 48.165 48.166 48.167 48.168 48.169 48.170 48.171 48.172 48.173 48.174 48.175 48.176 48.177 48.178 48.179 48.180 48.181 48.182 48.183 48.184 48.185 48.186 48.187 48.188 48.189 48.190 48.191 48.192 48.193 48.194 48.195 48.196 48.197 48.198 48.199 48.200 48.201 48.202 48.203 48.204 48.205 48.206 48.207 48.208 48.209 48.210 48.211 48.212 48.213 48.214 "Bacteria Table". Creighton University School of Medicine. http://medschool.creighton.edu/fileadmin/user/medicine/MMI/Files/Bacteria_Table.pdf.
- ↑ Brook, I (Oct 2008). "Actinomycosis: diagnosis and management.". Southern Medical Journal 101 (10): 1019–23. doi:10.1097/SMJ.0b013e3181864c1f. PMID 18791528.
- ↑ Mabeza, GF; Macfarlane J (March 2003). "Pulmonary actinomycosis". European Respiratory Journal 21 (3): 545–551. doi:10.1183/09031936.03.00089103. PMID 12662015.
- ↑ "Anthrax in animals". Food and Agriculture Organization. 2001. http://www.fao.org/ag/magazine/0112sp.htm.
- ↑ "CDC Anthrax Q & A: Treatment". http://emergency.cdc.gov/agent/anthrax/faq/treatment.asp.
- ↑ "FDA approves raxibacumab to treat inhalational anthrax". https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm332341.htm.
- ↑ 54.0 54.1 Itzhak Brook (Jan 28, 2014). "Bacteroides Infection Follow-up". Medscape. http://emedicine.medscape.com/article/233339-followup.
- ↑ "Clinical practice. Lyme disease". The New England Journal of Medicine 370 (18): 1724–31. 2014. doi:10.1056/NEJMcp1314325. PMID 24785207.
- ↑ 56.0 56.1 "Clinical Manifestations and Treatment of Lyme Disease". Clinics in Laboratory Medicine 35 (4): 765–78. 2015. doi:10.1016/j.cll.2015.08.004. PMID 26593256.
- ↑ "Nervous System Lyme Disease". Clinics in Laboratory Medicine 35 (4): 779–95. 2015. doi:10.1016/j.cll.2015.07.002. PMID 26593257.
- ↑ 58.0 58.1 58.2 58.3 58.4 58.5 Barbour, Alan G. (2017). "Relapsing Fever". in Kasper, Dennis L.; Fauci, Anthony S.. Harrison's Infectious Diseases (3rd ed.). New York: McGraw Hill Education. pp. 678–687. ISBN 978-1-259-83597-1.
- ↑ "Relapsing Fever Borreliae: A Global Review". Clinics in Laboratory Medicine 35 (4): 847–65. 2015. doi:10.1016/j.cll.2015.07.001. PMID 26593261.
- ↑ Atkinson, William (May 2012). Tetanus Epidemiology and Prevention of Vaccine-Preventable Diseases (12 ed.). Public Health Foundation. pp. 291–300. ISBN 9780983263135. https://www.cdc.gov/vaccines/pubs/pinkbook/tetanus.html. Retrieved 12 February 2015.
- ↑ "Diphtheria vaccine". Wkly Epidemiol Rec 81 (3): 24–32. 20 January 2006. PMID 16671240. https://www.who.int/wer/2006/wer8103.pdf.
- ↑ "ESCHERICHIA COLI". Public Health Agency of Canada. 2012-04-30. http://www.phac-aspc.gc.ca/lab-bio/res/psds-ftss/escherichia-coli-eng.php.
- ↑ 63.0 63.1 "Signs & Symptoms". 13 December 2018. https://www.cdc.gov/tularemia/signssymptoms/. Page last reviewed: October 26, 2015
- ↑ Ryan, KJ; Ray, CG, eds (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.
- ↑ "Klebsiella pneumoniae in Healthcare Settings". Centers for Disease Control and Prevention. 19 February 2021. https://www.cdc.gov/HAI/organisms/klebsiella/klebsiella.html. Page last reviewed: November 24, 2010. Page last updated: August 27, 2012
- ↑ Slack, A (Jul 2010). "Leptospirosis.". Australian Family Physician 39 (7): 495–8. PMID 20628664.
- ↑ McBride, AJ; Athanazio, DA; Reis, MG; Ko, AI (Oct 2005). "Leptospirosis". Current Opinion in Infectious Diseases 18 (5): 376–86. doi:10.1097/01.qco.0000178824.05715.2c. PMID 16148523.
- ↑ 68.0 68.1 Hartskeerl, Rudy A.; Wagenaar, Jiri F.P. (2017). "Leptospirosis". in Kasper, Dennis L.; Fauci, Anthony S.. Harrison's Infectious Diseases. New York: McGraw Hill Education. pp. 672–678. ISBN 978-1-259-83597-1.
- ↑ "Leprosy Fact sheet N°101". World Health Organization. January 2014. https://www.who.int/mediacentre/factsheets/fs101/en/.
- ↑ "Tuberculosis Fact sheet N°104". October 2015. https://www.who.int/mediacentre/factsheets/fs104/en/.
- ↑ Institut Pasteur Press Office - Vaccine against shigellosis (bacillary dysentery):a promising clinical trial 15 January 2009. Retrieved on 27 February 2009
- ↑ Levinson, W. (2010). Review of Medical Microbiology and Immunology (11th ed.). pp. 94–9.
- ↑ "Syphilis - CDC Fact Sheet (Detailed)". November 2, 2015. https://www.cdc.gov/std/syphilis/stdfact-syphilis-detailed.htm.
- ↑ "Reexamining syphilis: an update on epidemiology, clinical manifestations, and management". Annals of Pharmacotherapy 42 (2): 226–36. February 2008. doi:10.1345/aph.1K086. PMID 18212261.
- ↑ "Syphilis". Lancet 389 (10078): 1550–1557. 2017. doi:10.1016/S0140-6736(16)32411-4. PMID 27993382.
- ↑ "Molecular and physiological insights into plague transmission, virulence and etiology". Microbes Infect. 8 (1): 273–84. 2006. doi:10.1016/j.micinf.2005.06.006. PMID 16182593.
- ↑ Wagle PM. (1948). "Recent advances in the treatment of bubonic plague". Indian J Med Sci 2: 489–94.
- ↑ Meyer KF. (1950). "Modern therapy of plague". JAMA 144 (12): 982–5. doi:10.1001/jama.1950.02920120006003. PMID 14774219.
- ↑ "A decade of plague epidemiology and control in the Western Usambara mountains, north-east Tanzania". Acta Tropica 50 (4): 323–9. 1992. doi:10.1016/0001-706X(92)90067-8. PMID 1356303.
- ↑ "Yersinia pestis CO92ΔyopH Is a Potent Live, Attenuated Plague Vaccine". Clin. Vaccine Immunol. 14 (9): 1235–8. September 2007. doi:10.1128/CVI.00137-07. PMID 17652523.
- ↑ 81.0 81.1 Bernstein H, Bernstein C, Michod RE (2018). Sex in microbial pathogens. Infection, Genetics and Evolution volume 57, pages 8-25. https://doi.org/10.1016/j.meegid.2017.10.024
External links
| Classification |
|---|
- Bacterial Pathogen Pronunciation by Neal R. Chamberlain, Ph.D. at A.T. Still University
- Pathogenic bacteria genomes and related information at PATRIC, a Bioinformatics Resource Center funded by NIAID
 KSF
KSF![Overall age-standardised mortality rate per 100 000 population for 33 pathogens investigated, 2019[3]](https://handwiki.org/wiki/images/thumb/b/b2/Overall_age-standardised_mortality_rate_per_100_000_population_for_33_pathogens_investigated%2C_2019.jpg/552px-Overall_age-standardised_mortality_rate_per_100_000_population_for_33_pathogens_investigated%2C_2019.jpg)
![Global number of deaths (A) and YLLs (B), by pathogen and infectious syndrome, 2019[3]](https://handwiki.org/wiki/images/thumb/5/57/Global_number_of_deaths_%28A%29_and_YLLs_%28B%29%2C_by_pathogen_and_infectious_syndrome%2C_2019.jpg/216px-Global_number_of_deaths_%28A%29_and_YLLs_%28B%29%2C_by_pathogen_and_infectious_syndrome%2C_2019.jpg)
![Global number of deaths, by pathogen, age, and sex groups, 2019[3]](https://handwiki.org/wiki/images/thumb/7/76/Global_number_of_deaths%2C_by_pathogen%2C_age%2C_and_sex_groups%2C_2019.jpg/284px-Global_number_of_deaths%2C_by_pathogen%2C_age%2C_and_sex_groups%2C_2019.jpg)
