Norrish reaction
Topic: Physics
 From HandWiki - Reading time: 5 min
From HandWiki - Reading time: 5 min
A Norrish reaction, named after Ronald George Wreyford Norrish, is a photochemical reaction taking place with ketones and aldehydes. Such reactions are subdivided into Norrish type I reactions and Norrish type II reactions.[1] While of limited synthetic utility these reactions are important in the photo-oxidation of polymers such as polyolefins,[2] polyesters, certain polycarbonates and polyketones.
Type I
The Norrish type I reaction is the photochemical cleavage or homolysis of aldehydes and ketones into two free radical intermediates (α-scission). The carbonyl group accepts a photon and is excited to a photochemical singlet state. Through intersystem crossing the triplet state can be obtained. On cleavage of the α-carbon bond from either state, two radical fragments are obtained.[3] The size and nature of these fragments depends upon the stability of the generated radicals; for instance, the cleavage of 2-butanone largely yields ethyl radicals in favor of less stable methyl radicals.[4]
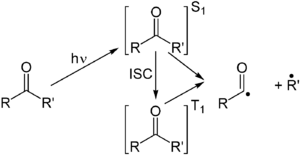
Several secondary reaction modes are open to these fragments depending on the exact molecular structure.
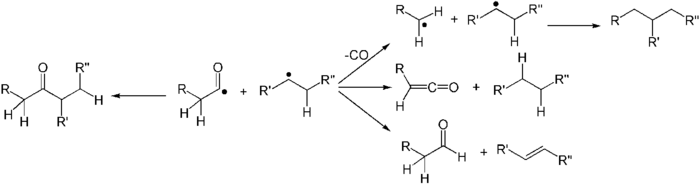
- The fragments can simply recombine to the original carbonyl compound, with racemisation at the α-carbon.
- The acyl radical can lose a molecule of carbon monoxide, forming a new carbon radical at the other α-carbon, followed by formation of a new carbon–carbon bond between the radicals.[3] The ultimate effect is simple extraction of the carbonyl unit from the carbon chain. The rate and yield of this product depends upon the bond-dissociation energy of the ketone's α substituents. Typically the more α substituted a ketone is, the more likely the reaction will yield products in this way.[5][6]
- The abstraction of an α-proton from the carbonyl fragment may form a ketene and an alkane.
- The abstraction of a β-proton from the alkyl fragment may form an aldehyde and an alkene.
The synthetic utility of this reaction type is limited, for instance it often is a side reaction in the Paternò–Büchi reaction. One organic synthesis based on this reaction is that of bicyclohexylidene.[7]
Type II
A Norrish type II reaction is the photochemical intramolecular abstraction of a γ-hydrogen (a hydrogen atom three carbon positions removed from the carbonyl group) by the excited carbonyl compound to produce a 1,4-biradical as a primary photoproduct.[8] Norrish first reported the reaction in 1937.[9]
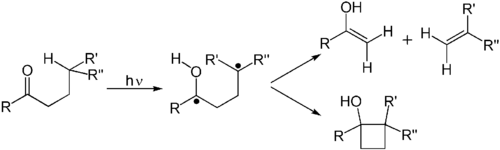
Secondary reactions that occur are fragmentation (β-scission) to form an alkene and an enol (which will rapidly tautomerise to a carbonyl), or intramolecular recombination of the two radicals to a substituted cyclobutane (the Norrish–Yang reaction).[10]
Scope
The Norrish reaction has been studied in relation to environmental chemistry with respect to the photolysis of the aldehyde heptanal, a prominent compound in Earth's atmosphere.[11] Photolysis of heptanal in conditions resembling atmospheric conditions results in the formation of 1-pentene and acetaldehyde in 62% chemical yield together with cyclic alcohols (cyclobutanols and cyclopentanols) both from a Norrish type II channel and around 10% yield of hexanal from a Norrish type I channel (the initially formed n-hexyl radical attacked by oxygen).
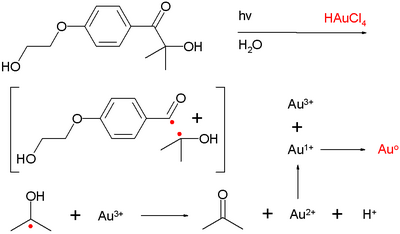
In one study[12] the photolysis of an acyloin derivative in water in presence of hydrogen tetrachloroaurate (HAuCl4) generated nanogold particles with 10 nanometer diameter. The species believed to responsible for reducing Au3+ to Au0[13] is the Norrish generated ketyl radical.
Leo Paquette's 1982 synthesis of dodecahedrane involves three separate Norrish-type reactions in its approximately 29-step sequence.
An example of a synthetically useful Norrish type II reaction can be found early in the total synthesis of the biologically active cardenolide ouabagenin by Phil Baran and coworkers.[14] The optimized conditions minimize side reactions, such as the competing Norrish type I pathway, and furnish the desired intermediate in good yield on a multi-gram scale.
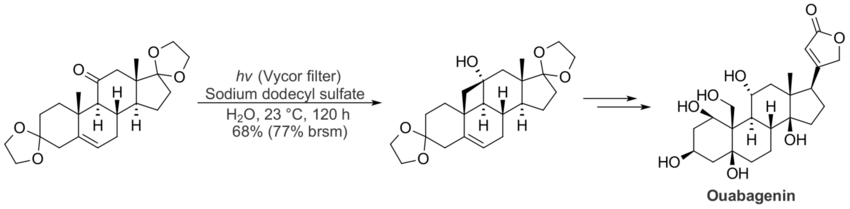
See also
- Photo-Fries rearrangement - a related reaction of aromatic carbonyls
- McLafferty rearrangement - similar to a Type II Norrish reaction. Caused by electron impact ionization rather than light
- Carbon monoxide-releasing molecules
References
- ↑ Named Organic Reactions, 2nd Edition, Thomas Laue and Andreas Plagens, John Wiley & Sons: Chichester, England, New York, 2005. 320 pp. ISBN 0-470-01041-X
- ↑ Grause, Guido; Chien, Mei-Fang; Inoue, Chihiro (November 2020). "Changes during the weathering of polyolefins". Polymer Degradation and Stability 181: 109364. doi:10.1016/j.polymdegradstab.2020.109364.
- ↑ 3.0 3.1 IUPAC Gold Book - Norrish Type I photoreaction. IUPAC. 24 February 2014. doi:10.1351/goldbook.N04219. http://goldbook.iupac.org/N04219.html. Retrieved 31 March 2014.
- ↑ Blacet, F. E.; N. Pitts Jr., James (1950). "Methyl Ethyl Ketone Photochemical Processes". Journal of the American Chemical Society 72 (6): 2810–2811. doi:10.1021/ja01162a544.
- ↑ Yang, Nien-Chu; D. Feit, Eugene; Hui, Man Him; Turro, Nicholas J.; Dalton, Christopher (1970). "Photochemistry of di-tert-butyl ketone and structural effects on the rate and efficiency of intersystem crossing of aliphatic ketones". Journal of the American Chemical Society 92 (23): 6974–6976. doi:10.1021/ja00726a046.
- ↑ Abuin, E.B.; Encina, M.V.; Lissi, E.A. (1972). "The photolysis of 3-pentanone". Journal of Photochemistry 1 (5): 387–396. doi:10.1016/0047-2670(72)80036-4.
- ↑ Bicyclohexylidene Nicholas J. Turro, Peter A. Leermakers, and George F. Vesley Organic Syntheses, Coll. Vol. 5, p.297 (1973); Vol. 47, p.34 (1967) Online article.
- ↑ IUPAC Gold Book - Norrish Type II photoreaction. IUPAC. 24 February 2014. doi:10.1351/goldbook.N04218. http://goldbook.iupac.org/N04218.html. Retrieved 31 March 2014.
- ↑ Norrish, R. G. W.; Bamford, C. H. (31 July 1937). "Photo-decomposition of Aldehydes and Ketones". Nature 140 (3535): 195–6. doi:10.1038/140195b0. Bibcode: 1937Natur.140..195N.
- ↑ IUPAC Gold Book - Norrish–Yang reaction. IUPAC. 24 February 2014. doi:10.1351/goldbook.NT07427. http://goldbook.iupac.org/NT07427.html. Retrieved 31 March 2014.
- ↑ Photolysis of Heptanal Suzanne E. Paulson, De-Ling Liu, Grazyna E. Orzechowska, Luis M. Campos, and K. N. Houk J. Org. Chem.; 2006; 71(17) pp 6403 - 6408; (Article) doi:10.1021/jo060596u
- ↑ Facile Photochemical Synthesis of Unprotected Aqueous Gold Nanoparticles Katherine L. McGilvray, Matthew R. Decan, Dashan Wang, and Juan C. Scaiano J. Am. Chem. Soc.; 2006; 128(50) pp 15980 - 15981; (Communication) doi:10.1021/ja066522h
- ↑ Technically Au3+ is reduced to Au2+ which then forms Au+ and Au3+ by disproportionation followed by final reduction of Au1+ to Auo
- ↑ Renata, H.; Zhou, Q.; Baran, P. S. (3 January 2013). "Strategic Redox Relay Enables A Scalable Synthesis of Ouabagenin, A Bioactive Cardenolide". Science 339 (6115): 59–63. doi:10.1126/science.1230631. PMID 23288535. Bibcode: 2013Sci...339...59R.
 |
 KSF
KSF