6-Methylsalicylic acid
Topic: Chemistry
 From Handwiki - Reading time: 1 min
From Handwiki - Reading time: 1 min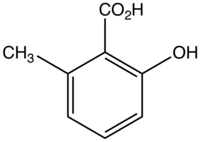
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Hydroxy-6-methylbenzoic acid | |
| Other names
2-Hydroxy-6-methylbenzoic acid
o-Cresotic acid 2,6-Cresotic acid o-Cresotinic acid 2-Hydroxy-o-toluic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| 3DMet | |
| 2208693 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C8H8O3 | |
| Molar mass | 152.149 g·mol−1 |
| Appearance | white solid |
| Melting point | 141.5–142 °C (286.7–287.6 °F; 414.6–415.1 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
6-Methylsalicylic acid is an organic compound with the formula CH3C6H3(CO2H)(OH). It is a white solid that is soluble in basic water and in polar organic solvents. At neutral pH, the acid exists as 6-methylsalicylate Its functional groups include a carboxylic acid and a phenol group. It is one of four isomers of methylsalicylic acid.
It occurs naturally, being a biosynthetic precursor to m-cresol.[1] Its decarboxylation is catalyzed by 6-methylsalicylate decarboxylase:
- 6-methylsalicylate [math]\displaystyle{ \rightleftharpoons }[/math] 3-cresol + CO2
See also
References
- ↑ Staunton, James; Weissman, Kira J. (2001). "Polyketide Biosynthesis: A Millennium Review". Natural Product Reports 18 (4): 380–416. doi:10.1039/a909079g. PMID 11548049.
 |
This article is licensed under CC BY-SA 3.0.
Original source: https://handwiki.org/wiki/6-Methylsalicylic acid
Status: article is cached
Original source: https://handwiki.org/wiki/6-Methylsalicylic acid
Status: article is cached
 KSF
KSF