Acridone
Topic: Chemistry
 From Handwiki - Reading time: 2 min
From Handwiki - Reading time: 2 min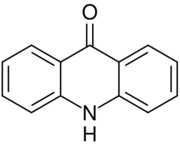
| |
| Names | |
|---|---|
| Preferred IUPAC name
Acridin-9(10H)-one | |
| Other names
9-Acridanone
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C13H9NO | |
| Molar mass | 195.221 g·mol−1 |
| Appearance | yellow powder |
| Melting point | 250 °C (482 °F; 523 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Acridone is an organic compound based on the acridine skeleton, with a carbonyl group at the 9 position.
Synthesis and structure
The molecule is planar. Optical spectra reveal that the keto tautomer predominates in the gas-phase and in ethanol solution.[1]
Acridone can be synthesized by heating fenamic acid.[2]
History
One of the first who were able to prove the compound's existence was Karl Drechsler, Student of G. Goldschmiedt, at the k.u.k. Universität Wien (Vienna, Austria) in 1914.[3]
Derivatives
Acridone constitutes the scaffold of some synthetic compounds with diverse pharmacological activities. 3-Chloro-6-(2-diethylamino-ethoxy)-10-(2-diethylamino-ethyl)-acridone has shown promise as an antimalarial drug.[4][5]
See also
References
- ↑ Beak, Peter; Fry, Fred S.; Lee, Jaekeun; Steele, Frank (1976). "Equilibration studies. Protomeric equilibria of 2- and 4-hydroxypyridines, 2- and 4-hydroxypyrimidines, 2- and 4-mercaptopyridines, and structurally related compounds in the gas phase". Journal of the American Chemical Society 98: 171–179. doi:10.1021/ja00417a027.
- ↑ C. F. H. Allen; G. H. W. McKee (1939). "Acridone". Organic Syntheses 19: 6. doi:10.15227/orgsyn.019.0006.
- ↑ Austrian National Library, Reports of the monthly meetings of the Academy of Sciences
- ↑ HISASHI FUJIOKA; YUKIHIRO NISHIYAMA; HIROSHI FURUKAWA; NOBUO KUMADA (1989). "In Vitro and In Vivo Activities of Atalaphillinine and Related Acridone Alkaloids against Rodent Malaria". Antimicrobial Agents and Chemotherapy 33 (1): 6–9. doi:10.1128/aac.33.1.6. PMID 2653215.
- ↑ Kelly, Jane X.; Smilkstein, Martin J.; Brun, Reto; Wittlin, Sergio; Cooper, Roland A.; Lane, Kristin D.; Janowsky, Aaron; Johnson, Robert A. et al. (2009). "Discovery of dual function acridones as a new antimalarial chemotype". Nature 459 (7244): 270–273. doi:10.1038/nature07937. PMID 19357645. Bibcode: 2009Natur.459..270K.
 |
This article is licensed under CC BY-SA 3.0.
Original source: https://handwiki.org/wiki/Acridone
Status: article is cached
Original source: https://handwiki.org/wiki/Acridone
Status: article is cached
 KSF
KSF