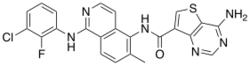
Belvarafenib
Topic: Chemistry
 From Handwiki - Reading time: 1 min
From Handwiki - Reading time: 1 minShort description: Investigational cancer drug
 | |
| Clinical data | |
|---|---|
| Other names | HM95573
GDC5573 RG6185 |
| Routes of administration | By mouth |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C23H16ClFN6OS |
| Molar mass | 478.93 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Belvarafenib (developed by Hanmi Pharmaceuticals and Genentech) is a small molecule RAF dimer (type II) inhibitor[1] which shows anti-tumor clinical activity in cancer patients with BRAFV600E- and NRAS- mutations.[2]
References
- ↑ "Melanoma with genetic alterations beyond the BRAFV600 mutation: management and new insights". Current Opinion in Oncology 34 (2): 115–122. March 2022. doi:10.1097/CCO.0000000000000817. PMID 35050937.
- ↑ "Belvarafenib, a novel pan-RAF inhibitor, in solid tumor patients harboring BRAF, KRAS, or NRAS mutations: Phase I study.". Journal of Clinical Oncology 37 (15 suppl): 3000. 31 May 2019. doi:10.1200/JCO.2019.37.15_suppl.3000.
 |
This article is licensed under CC BY-SA 3.0.
Original source: https://handwiki.org/wiki/Belvarafenib
Status: article is cached
Original source: https://handwiki.org/wiki/Belvarafenib
Status: article is cached
 KSF
KSF