Bicyclobutane
Topic: Chemistry
 From Handwiki - Reading time: 3 min
From Handwiki - Reading time: 3 min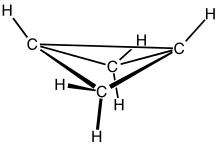
| |
| Names | |
|---|---|
| Preferred IUPAC name
Bicyclo[1.1.0]butane | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C4H6 | |
| Molar mass | 54.092 g·mol−1 |
| Appearance | colorless gas |
| Boiling point | 8.3 ± 0.2 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Bicyclobutane is an organic compound with the formula C4H6. It is a bicyclic molecule consisting of two cis-fused cyclopropane rings, and is a colorless and easily condensed gas.[1] Bicyclobutane is noted for being one of the most strained compounds that is isolatable on a large scale — its strain energy is estimated at 63.9 kcal mol−1. It is a nonplanar molecule, with a dihedral angle between the two cyclopropane rings of 123°.[2]
The first reported bicyclobutane was the carboxyethyl derivative, C4H5CO2Et, which was prepared by dehydrohalogenation the corresponding bromocyclobutanecarboxylate ester with sodium hydride.[2] The parent hydrocarbon was prepared from 1-bromo-3-chlorocyclobutane by conversion of the bromocyclobutanecarboxylate ester,[1] followed by intramolecular Wurtz coupling using molten sodium.[3] The intermediate 1-bromo-3-chlorocyclobutane can also be prepared via a modified Hunsdiecker reaction from 3-chlorocyclobutanecarboxylic acid using mercuric oxide and bromine:[4]
A synthetic approach to bicyclobutane derivatives involves ring closure of a suitably substituted 2-bromo-1-(chloromethyl)cyclopropane with magnesium in THF.[5]
Stereochemical evidence indicates that bicyclobutane undergoes thermolysis to form 1,3-butadiene with an activation energy of 41 kcal mol−1 via a concerted pericyclic mechanism (cycloreversion, [σ2s+σ2a]).[6]
Biological synthesis
Multiple research groups have reported success in synthesizing bicyclobutane-containing molecules using enzymes in living cells. One group reported the transformation of linolenic acid into a bicyclobutane fatty acid via a protein produced by a strain of the cyanobacterium Anabaena sp. (strain PCC 7120).[7] The other group reported a directed evolution approach, whereby engineered heme protein was expressed in E. coli and successfully optimized for increased rate and yield of biosynthesis of a substituted bicyclobutane derivative.[8]
See also
- Propalene (Bicyclobutadiene)
- Bicyclopentane
- 1.1.1-Propellane
References
- ↑ 1.0 1.1 Wiberg, K. B.; Lampman, G. M.; Ciula, R. P.; Connor, D. S.; Schertler, P.; Lavanish, J. (1965). "Bicyclo[1.1.0]butane". Tetrahedron 21 (10): 2749–2769. doi:10.1016/S0040-4020(01)98361-9.
- ↑ 2.0 2.1 Wiberg, K. B. (1968). "Small Ring Bicyclo[n.m.0alkanes"]. in Hart, H.; Karabatsos, G. J.. Advances in Alicyclic Chemistry. 2. Academic Press. pp. 185–254. ISBN 9781483224213. https://books.google.com/books?id=9h0SBQAAQBAJ&pg=PA191.
- ↑ Lampman, Gary M.; Aumiller, James C. (1971). "Bicyclo[1.1.0butane"]. Org. Synth. 51: 55. doi:10.15227/orgsyn.051.0055. http://www.orgsyn.org/demo.aspx?prep=CV6P0133.; Coll. Vol., 6, pp. 133
- ↑ Lampman, Gary M.; Aumiller, James C. (1971). "Mercury(II) oxide-modified Hunsdiecker reaction: 1-Bromo-3-chlorocyclobutane". Org. Synth. 51: 106. doi:10.15227/orgsyn.051.0106. http://www.orgsyn.org/demo.aspx?prep=cv6p0179.; Coll. Vol., 6, pp. 179
- ↑ D'yachenko, A. I.; Abramova, N. M.; Zotova, S. V.; Nesmeyanova, O. A.; Bragin, O. V. (1985). "New synthesis of bicyclo[1.1.0]butane hydrocarbons". Bulletin of the Academy of Sciences of the USSR 34 (9): 1885–1889. doi:10.1007/BF00953929.
- ↑ Woodward, Robert B.; Hoffmann, Roald (1969). "The Conservation of Orbital Symmetry". Angewandte Chemie International Edition 8 (11): 781–853. doi:10.1002/anie.196907811.
- ↑ Schneider, Claus; Niisuke, Katrin; Boeglin, William E.; Voehler, Markus; Stec, Donald F.; Porter, Ned A.; Brash, Alan R. (2007-11-27). "Enzymatic synthesis of a bicyclobutane fatty acid by a hemoprotein lipoxygenase fusion protein from the cyanobacterium Anabaena PCC 7120". Proceedings of the National Academy of Sciences of the United States of America 104 (48): 18941–18945. doi:10.1073/pnas.0707148104. ISSN 1091-6490. PMID 18025466. Bibcode: 2007PNAS..10418941S.
- ↑ Chen, Kai; Huang, Xiongyi; Kan, S. B. Jennifer; Zhang, Ruijie K.; Arnold, Frances H. (6 April 2018). "Enzymatic construction of highly strained carbocycles". Science 360 (6384): 71–75. doi:10.1126/science.aar4239. ISSN 1095-9203. PMID 29622650. Bibcode: 2018Sci...360...71C.
Original source: https://handwiki.org/wiki/Bicyclobutane
Status: article is cached
 KSF
KSF
