Ferrate
Topic: Chemistry
 From Handwiki - Reading time: 2 min
From Handwiki - Reading time: 2 minFerrate loosely refers to a material that can be viewed as containing anionic iron complexes. The term ferrate derives from la ferrum 'iron'.
Examples include tetrachloroferrate ([FeCl4]2−), oxyanions (e.g. FeO2−4), and the organoferrates, including highly reduced tetracarbonylferrate ([Fe(CO)4]2−) salts.[1] Although rarely studied, ferrate(V) [FeO4]3− and ferrate(IV) [FeO4]4− oxyanions of iron also exist. These too are called ferrates.[2]
- Ferrates
-
Disodium salt of tetracarbonylferrate
-
Structure of ferrate(VI), [FeO4]2−
-
1-Butyl-3-methylimidazolium salt of [FeCl4]−
-
Potassium tris(oxalato)ferrate
Ferrate(VI) is a strong oxidizer,[3] used in water purification[4] and battery technology.[5][6]
References
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Graham Hill; John Holman (2000). Chemistry in context (5th ed.). Nelson Thornes. p. 202. ISBN 0-17-448276-0.
- ↑ Sharma, Virender K. (2002-03-01). "Potassium ferrate(VI): an environmentally friendly oxidant". Advances in Environmental Research 6 (2): 143–156. doi:10.1016/S1093-0191(01)00119-8. ISSN 1093-0191. Bibcode: 2002AdvER...6..143S. https://www.sciencedirect.com/science/article/pii/S1093019101001198.
- ↑ Licht, Stuart; Yu, Xingwen (2005-10-01). "Electrochemical Alkaline Fe(VI) Water Purification and Remediation" (in en). Environmental Science & Technology 39 (20): 8071–8076. doi:10.1021/es051084k. ISSN 0013-936X. PMID 16295877. Bibcode: 2005EnST...39.8071L. https://pubs.acs.org/doi/10.1021/es051084k.
- ↑ Licht, Stuart; Wang, Baohui; Ghosh, Susanta (1999-08-13). "Energetic Iron(VI) Chemistry: The Super-Iron Battery" (in en). Science 285 (5430): 1039–1042. doi:10.1126/science.285.5430.1039. ISSN 0036-8075. PMID 10446044. https://www.science.org/doi/10.1126/science.285.5430.1039.
- ↑ Licht, Stuart; Naschitz, Vera; Halperin, Leonid; Halperin, Nadezhda; Lin, Lin; Chen, Jianjun; Ghosh, Susanta; Liu, Bing (2001-10-15). "Analysis of ferrate(VI) compounds and super-iron Fe(VI) battery cathodes: FTIR, ICP, titrimetric, XRD, UV/VIS, and electrochemical characterization". Journal of Power Sources 101 (2): 167–176. doi:10.1016/S0378-7753(01)00786-8. ISSN 0378-7753. Bibcode: 2001JPS...101..167L. https://www.sciencedirect.com/science/article/pii/S0378775301007868.
 |
This article is licensed under CC BY-SA 3.0.
Original source: https://handwiki.org/wiki/Ferrate
Status: article is cached
Original source: https://handwiki.org/wiki/Ferrate
Status: article is cached
 KSF
KSF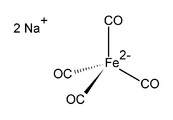
![Structure of ferrate(VI), [FeO4]2−](https://handwiki.org/wiki/images/thumb/6/65/Ferrate_ion.svg/141px-Ferrate_ion.svg.png)
![1-Butyl-3-methylimidazolium salt of [FeCl4]−](https://handwiki.org/wiki/images/thumb/4/45/1-Butyl-3-methylimidazolium_tetrachloroferrate.svg/180px-1-Butyl-3-methylimidazolium_tetrachloroferrate.svg.png)

