Flusilazole
Topic: Chemistry
 From Handwiki - Reading time: 2 min
From Handwiki - Reading time: 2 min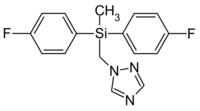
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1-{[Bis(4-fluorophenyl)(methyl)silyl]methyl}-1H-1,2,4-triazole | |
| Other names
DPX-H6573;
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C16H15F2N3Si | |
| Molar mass | 315.392 g/mol |
| 41.9 mg/L (20°C) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Flusilazole (DPX-H6573) is an organosilicon fungicide invented by DuPont, which is used to control fungal infections on a variety of fruit and vegetable crops.[1][2][3] It is moderately toxic to animals and has been shown to produce birth defects in high doses.[4][5]
References
- ↑ Moberg, W. K.; Basarab, G. S.; Cuomo, J.; Liang, P. H. (1987). "Biologically Active Organosilicon Compounds". Synthesis and Chemistry of Agrochemicals. ACS Symposium Series. 355. pp. 288–301. doi:10.1021/bk-1987-0355.ch026. ISBN 9780841214347.
- ↑ "Baseline toxicity of several pesticides to Hyaliodes vitripennis (Say) (Hemiptera: Miridae)". Pest Management Science 57 (11): 1007–10. November 2001. doi:10.1002/ps.374. PMID 11721516.
- ↑ "Effects of fungicides on in vitro spore germination and mycelial growth of the phytopathogens Leptosphaeria maculans and L. biglobosa (phoma stem canker of oilseed rape)". Pest Management Science 66 (4): 396–405. April 2010. doi:10.1002/ps.1890. PMID 20013877.
- ↑ "Developmental toxic effects of antifungal flusilazole administered by gavage to mice". Birth Defects Research Part B: Developmental and Reproductive Toxicology 80 (1): 12–7. February 2007. doi:10.1002/bdrb.20098. PMID 17187383.
- ↑ "Relative embryotoxicity of two classes of chemicals in a modified zebrafish embryotoxicity test and comparison with their in vivo potencies". Toxicology in Vitro 25 (3): 745–753. January 2011. doi:10.1016/j.tiv.2011.01.005. PMID 21238576.
External links
- Flusilazole in the Pesticide Properties DataBase (PPDB)
 |
This article is licensed under CC BY-SA 3.0.
Original source: https://handwiki.org/wiki/Flusilazole
Status: article is cached
Original source: https://handwiki.org/wiki/Flusilazole
Status: article is cached
 KSF
KSF