Quinone methide
Topic: Chemistry
 From Handwiki - Reading time: 6 min
From Handwiki - Reading time: 6 min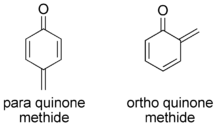
| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
| 1922177 | |
| ChEBI |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C7H6O | |
| Molar mass | 106.124 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
A quinone methide is a type of conjugated organic compound that contain a cyclohexadiene with a carbonyl and an exocyclic methylidene or extended alkene unit. It is analogous to a quinone, but having one of the double bonded oxygens replaced with a carbon. The carbonyl and methylidene are usually oriented either ortho or para to each other. There are some examples of transient synthetic meta quinone methides.
Properties
Quinone methides are cross-conjugated rather than aromatic. Nucleophilic addition at the exo-cyclic double bond will result in rearomatisation, making such reactions highly favourable. As a result, quinone methides are excellent, electrophilic Michael acceptors, react quickly with nucleophiles and can be easily reduced. They are able to act as radical scavengers via a similar process, a behaviour exploited by certain polymerisation inhibitors. Quinone methides are more polar than quinones, and therefore more chemically reactive. Simple unhindered quinone methides are short lived reactive intermediates that are not stable enough to be isolated under normal circumstances, they will trimerise in the absence of nucleophiles.[1] Sterically hindered quinone methides can be sufficiently stable to be isolated, with some examples being commercially available.
Preparation
Quinone methides are often prepared by oxidation of the corresponding ortho or para cresol.
Quinone methides can be produced in aqueous solution by photochemical dehydration of o-hydroxybenzyl alcohols (i.e. salicyl alcohol).
Occurrence and applications
Quinones methides are commonly invoked in biochemistry, but are rarely observed as long-lived intermediates.
Biosynthesis of dehydroglycine
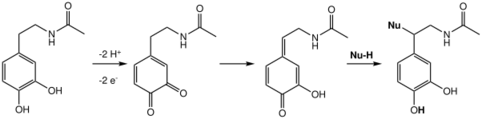
Quinone methide itself arises by the degradation of tyrosine, leading ultimately to p-cresol.[2] Various quinone methides are directly involved in the process of lignification (creation of complex lignin polymers) in plants.[3]
Quinone methides have been implicated as the ultimate cytotoxins responsible for the effects of such agents as antitumor drugs, antibiotics, and DNA alkylators.[4] Oxidation to a reactive quinone methide is the mechanistic basis of many phenolic anti-cancer drugs.
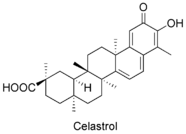
Celastrol is a triterpenoid quinone methide isolated from Tripterygium wilfordii (Thunder of God vine) and Celastrus regelii that exhibits antioxidant (15 times the potency of α-tocopherol),[6] anti-inflammatory,[7] anticancer,[8][9][10][11] and insecticidal [12] activities.
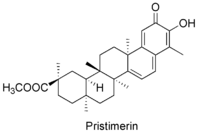
Pristimerin, the methyl ester of celasterol, is a triterpenoid quinone methide isolated from Maytenus heterophylla that displays antitumor and antiviral [13] activities. Pristimerin has also been found to have a contraceptive effect due to its inhibiting effect on the calcium channel of sperm (CatSper).[14]
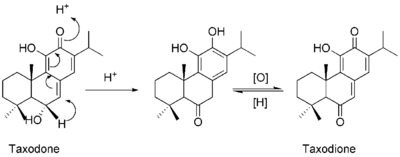
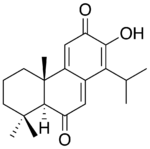
Taxodone and its oxidized rearrangement product, taxodione, are diterpenoid quinone methides found in Taxodium distichum (bald cypress), Rosmarinus officinalis (rosemary), several Salvia species and other plants, that display anticancer,[15][16][17] antibacterial,[18][19][20] antioxidant,[21] antifungal,[22] insecticide,[23] and antifeedant [24] activities.
Maytenoquinone, an isomer of taxodione, is a biologically active quinone methide found in Maytenus dispermus.[25]
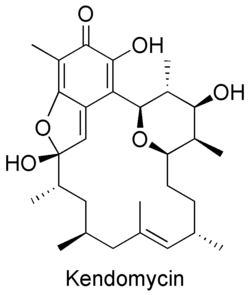
Kendomycin is an antitumor antibacterial quinone methide macrolide first isolated from the bacterium Streptomyces violaceoruber.[26] It has potent activity as an endothelin receptor antagonist and anti-osteoporosis agent.[27]
Elansolid A3 is a quinone methide from the bacterium Chitinophaga sancti that displays antibiotic activity.[28] Antibacterial quinone methides, 20-epi-isoiguesterinol, 6-oxoisoiguesterin, isoiguesterin and isoiguesterinol were found in Salacia madagascariensis.[29] Quinone methides tingenone and netzahualcoyonol were isolated from Salacia petenensis.[30] Nortriterpenoid quinone methide amazoquinone and (7S, 8S)-7-hydroxy-7,8-dihydro-tingenone were isolated from Maytenus amazonica.[31] An antimicrobial quinone methide, 15 alpha-hydroxypristimerin, was isolated from a South American medicinal plant, Maytenus scutioides.[32]
Quinone dimethides
A quinone dimethide (or "xylylene") is a compound with the formula C6H4(=CH2)2. Thus they are related to quinone monomethides (the topic of this article) by replacing the keto group with methylidene. A well studied example is tetracyanoquinodimethane.
References
- ↑ Cavitt, S. B.; R., H. Sarrafizadeh; Gardner, P. D. (April 1962). "The Structure of o-Quinone Methide Trimer". The Journal of Organic Chemistry 27 (4): 1211–1216. doi:10.1021/jo01051a021.
- ↑ Stich, T. A.; Myers, W. K.; Britt, R. D., "Paramagnetic intermediates generated by radical S-adenosylmethionine (SAM) enzymes", Acc. Chem. Res. 2014, 47, 2235-2243.
- ↑ Quinone Methides in Lignification
- ↑ "Quinone methide derivatives: important intermediates to DNA alkylating and DNA cross-linking actions". Curr Med Chem 12 (24): 2893–2913. 2005. doi:10.2174/092986705774454724. PMID 16305478.
- ↑ Andersen, Svend Olav (2010). "Insect Cuticular Sclerotization: A Review". Insect Biochemistry and Molecular Biology 40 (3): 166–178. doi:10.1016/j.ibmb.2009.10.007. PMID 19932179.
- ↑ "Celastrol, a potent antioxidant and anti-inflammatory drug, as a possible treatment for Alzheimer's disease.". Prog Neuropsychopharmacol Biol Psychiatry 25 (7): 1341–1357. 2001. doi:10.1016/S0278-5846(01)00192-0. PMID 11513350.
- ↑ "Suppression of inflammatory responses by celastrol, a quinone methide triterpenoid isolated from Celastrus regelii". Eur J Clin Invest 39 (9): 819–827. 2009. doi:10.1111/j.1365-2362.2009.02186.x. PMID 19549173.
- ↑ "Enhancement of radiation sensitivity in lung cancer cells by celastrol is mediated by inhibition of Hsp90.". Int J Mol Med 27 (3): 441–446. 2011. doi:10.3892/ijmm.2011.601. PMID 21249311.
- ↑ Tiedemann (2009). "Identification of a potent natural triterpenoid inhibitor of proteosome chymotrypsin-like activity and NF-kappaB with antimyeloma activity in vitro and in vivo.". Blood 113 (17): 4027–37. doi:10.1182/blood-2008-09-179796. PMID 19096011.
- ↑ "Celastrol acts as a potent antimetastatic agent targeting beta1 integrin and inhibiting cell-extracellular matrix adhesion, in part via the p38 mitogen-activated protein kinase pathway". J Pharmacol Exp Ther 334 (2): 489–499. 2010. doi:10.1124/jpet.110.165654. PMID 20472666.
- ↑ Byun (2009). "Reactive oxygen species-dependent activation of Bax and Poly(ADP)-ribose) polymerase-1 is required for mitochondrial cell death induced by triterpenoid Pristimerin in human cervical cancer cells.". Mol. Pharmacol. 76 (4): 734–44. doi:10.1124/mol.109.056259. PMID 19574249.
- ↑ "Insecticidal activity of Maytenus species (Celastraceae) nortriterpene quinone methides against codling moth, Cydia pomonella (L.) (Lepidoptera: tortricidae).". Journal of Agricultural and Food Chemistry 48 (1): 88–92. 2000. doi:10.1021/jf990008w. PMID 10637057.
- ↑ "Anticytomegalovirus activity of pristimerin, a triterpenoid quinone methide isolated from Maytenus heterophylla (Eckl. & Zeyh.).". Antivir Chem Chemother 18 (3): 133–139. 2007. doi:10.1177/095632020701800303. PMID 17626597.
- ↑ Nadja Mannowetza; Melissa R. Millera; Polina V. Lishko (2017). "Regulation of the sperm calcium channel CatSper by endogenous steroids and plant triterpenoids". Proceedings of the National Academy of Sciences of the United States of America 114 (22): 5743–5748. doi:10.1073/pnas.1700367114. PMID 28507119. Bibcode: 2017PNAS..114.5743M.
- ↑ Kupchan, S. M.; Karim, A; Marcks, C. (1968). "Tumor inhibitors. XXXIV. Taxodione and taxodone, two novel diterpenoid quinone methide tumor inhibitors from Taxodium distichum". J Am Chem Soc 90 (21): 5923–4. doi:10.1021/ja01023a061. PMID 5679178.
- ↑ "Taxodione, a DNA-binding compound from Taxodium distichum L. (Rich.).". Z Naturforsch C 63 (5–6): 355–360. 2008. doi:10.1515/znc-2008-5-608. PMID 18669020.
- ↑ Ayhan Ulubelen, Gülaçti Topçu, Hee-Byung Chai and John M. Pezzuto (1999). "Cytotoxic Activity of Diterpenoids Isolated from Salvia hypargeia". Pharmaceutical Biology 37 (2): 148–151. doi:10.1076/phbi.37.2.148.6082.
- ↑ Vivek K. Bajpai; Sun Chul Kan (2010). "Antibacterial abietane-type diterpenoid, taxodone from Metasequoia glyptostroboides Miki ex Hu". Journal of Biosciences 35 (4): 533–538. doi:10.1007/s12038-010-0061-z. PMID 21289435.
- ↑ Vivek K. Bajpai; Minkyun Na; Sun Chul Kang (2010). "The role of bioactive substances in controlling foodborne pathogens derived from Metasequoia glyptostroboides Miki ex Hu". Food and Chemical Toxicology 48 (7): 1945–1949. doi:10.1016/j.fct.2010.04.041. PMID 20435080.
- ↑ "Syntheses and antibacterial activities of diterpene catechol derivatives with abietane, totarane and podocarpane skeletons against methicillin-resistant Staphylococcus aureus and Propionibacterium acnes". Chem Pharm Bull 58 (6): 818–824. 2010. doi:10.1248/cpb.58.818. PMID 20522992.
- ↑ Ufuk Kolak; Ahmed Kabouche; Mehmet Öztürk; Zahia Kabouche; Gülaçtl Topçu; Ayhan Ulubelen (2009). "Antioxidant diterpenoids from the roots of Salvia barrelieri". Phytochemical Analysis 20 (4): 320–327. doi:10.1002/pca.1130. PMID 19402189. Bibcode: 2009PChAn..20..320K.
- ↑ Norihisa Kusumoto; Tatsuya Ashitani; Tetsuya Murayama; Koichi Ogiyama; Koetsu Takahashi (2010). "Antifungal Abietane-Type Diterpenes from the Cones of Taxodium distichum Rich". Journal of Chemical Ecology 36 (12): 1381–1386. doi:10.1007/s10886-010-9875-2. PMID 21072573.
- ↑ Norihisa Kusumoto; Tatsuya Ashitani; Yuichi Hayasaka; Tetsuya Murayama; Koichi Ogiyama; Koetsu Takahashi (2009). "Antitermitic Activities of Abietane-type Diterpenes from Taxodium distichum Cones". Journal of Chemical Ecology 35 (6): 635–642. doi:10.1007/s10886-009-9646-0. PMID 19475449.
- ↑ M. C. Ballesta-Acosta1, M. J. Pascual-Villalobos and B. Rodríguez (2008). "Short communication. The antifeedant activity of natural plant products towards the larvae of Spodoptera littoralis". Spanish Journal of Agricultural Research 6 (1): 85–91. doi:10.5424/sjar/2008061-304.
- ↑ J. D. Martín (1973). "New diterpenoids extractives of Maytenus dispermus". Tetrahedron 29 (17): 2553–2559. doi:10.1016/0040-4020(73)80172-3.
- ↑ H B Bode; A Zeeck (2000). "Structure and biosynthesis of kendomycin, a carbocyclic ansa-compound from Streptomyces". J Chem Soc Perkin Trans 1 323 (3): 323–328. doi:10.1039/a908387a.
- ↑ Burke Research Group University of Wisconsin
- ↑ "Elansolid A3, a Unique p-Quinone Methide Antibiotic from Chitinophaga sancti.". Chem. Eur. J. 17 (28): 7739–44. 2011. doi:10.1002/chem.201100457. PMID 21626585.
- ↑ "Bisnortriterpenes from Salacia madagascariensis.". J Nat Prod 68 (2): 251–254. 2005. doi:10.1021/np0497088. PMID 15730255.
- ↑ "Isolation and frontier molecular orbital investigation of bioactive quinone-methide triterpenoids from the bark of Salacia petenensis.". Planta Med 67 (1): 65–69. 2001. doi:10.1055/s-2001-10879. PMID 11270725.
- ↑ "New phenolic and quinone-methide triterpenes from Maytenus amazonica.". J Nat Prod 62 (3): 434–436. 1999. doi:10.1021/np980412+. PMID 10096852.
- ↑ "A new bioactive norquinone-methide triterpene from Maytenus scutioides.". Planta Med 64 (8): 767–771. 1998. doi:10.1055/s-2006-957581. PMID 10075545.
External links
- Formation and Stability of Simple Quinone Methides
- Quinone methide intermediates in organic Photochemistry
- Reactive intermediates. Some chemistry of quinone methides
- "o-Quinone methides: intermediates underdeveloped and underutilized in organic synthesis". Tetrahedron 58 (27): 5367–5405. 2002. doi:10.1016/S0040-4020(02)00496-9.
 |
Original source: https://handwiki.org/wiki/Quinone methide
Status: article is cached
 KSF
KSF