Silver bromate
Topic: Chemistry
 From Handwiki - Reading time: 2 min
From Handwiki - Reading time: 2 min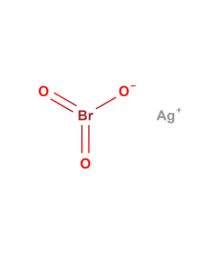
| |
| Names | |
|---|---|
| Systematic IUPAC name
Silver(I) bromate | |
| Other names
Argentous bromate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| AgBrO3 | |
| Molar mass | 235.770 g/mol |
| Appearance | white powder photosensitive |
| Density | 5.206 g/cm3 |
| Melting point | 309 °C (588 °F; 582 K) |
| 0.167 g/100 mL | |
Solubility product (Ksp)
|
5.38×10−5[1] |
| Solubility in ammonium hydroxide | soluble |
| Hazards | |
| Safety data sheet | MSDS |
| GHS pictograms |  
|
| GHS Signal word | DANGER |
| H272, H315, H319, H335 | |
| P210, P220, P261, P264, P271, P280, P302+352, P304+340+312Script error: No such module "Preview warning".Category:GHS errors, P305+351+338, P332+313, P337+313, P362+364Script error: No such module "Preview warning".Category:GHS errors, P370+378, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Silver bromate (AgBrO3), is a toxic, light and heat-sensitive, white powder.[2]
Uses
Silver bromate can be used as an oxidant for the transformation of tetrahydropyranyl ethers to carbonyl compounds.[3]
References
- ↑ John Rumble (June 18, 2018) (in English). CRC Handbook of Chemistry and Physics (99 ed.). CRC Press. pp. 5–189. ISBN 978-1138561632.
- ↑ Perry, Dale L. (2011). Handbook of inorganic compounds (2nd ed.). Boca Raton: Taylor & Francis. pp. 365. ISBN 978-1-4398-1461-1. OCLC 587104373.
- ↑ Mohammadpoor-Baltork, Iraj; Nourozi, Ali Reza (1999). "Efficient and Selective Oxidative Deprotection of Tetrahydropyranyl Ethers, Ethylene Acetals and Ketals with Silver and Sodium Bromates in the Presence of Aluminum Chloride". Synthesis 1999 (3): 487–490. doi:10.1055/s-1999-3410.
External links
 |
This article is licensed under CC BY-SA 3.0.
Original source: https://handwiki.org/wiki/Silver bromate
Status: article is cached
Original source: https://handwiki.org/wiki/Silver bromate
Status: article is cached
 KSF
KSF
