Fingolimod
 From Mdwiki - Reading time: 14 min
From Mdwiki - Reading time: 14 min
 | |
 | |
| Names | |
|---|---|
| Trade names | Gilenya |
| |
| Clinical data | |
| Drug class | Immunosuppressants |
| Main uses | Multiple sclerosis (MS)[1] |
| Side effects | Headache, liver problems, diarrhea, nausea, cough, pain[1] |
| Pregnancy category |
|
| Routes of use | By mouth (capsules) |
| Typical dose | 500 ug OD[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a611006 |
| Legal | |
| License data |
|
| Legal status | |
| Chemical and physical data | |
| Formula | C19H33NO2 |
| Molar mass | 307.478 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Fingolimod, sold under the brand name Gilenya, is a medication, mostly used to treat multiple sclerosis (MS).[1] It reduces relapses in relapsing-remitting MS by approximately one-half over a two-year period.[3] It may also be used for chronic inflammatory demyelinating polyneuropathy.[1] It is taken by mouth.[2]
Common side effects include headache, liver problems, diarrhea, nausea, cough, and pain.[1] Other side effects may include AV block, infection, allergic reactions, skin cancer, lung problems, progressive multifocal leukoencephalopathy, and posterior reversible encephalopathy syndrome.[1] Use in pregnancy may harm the baby.[1] It is a sphingosine-1-phosphate receptor modulator, which prevents T cells from travelling from lymph nodes to the brain and spinal cord.[4]
Fingolimod was approved for medical use in the United States in 2010 and Europe in 2011.[1][4] In the United Kingdom 4 weeks of treatment costs the NHS about £1,470 as of 2021.[2] This amount in the United States costs about 8,300 USD.[5] A generic version was approved in 2021 in the USA.[6]
Medical uses[edit | edit source]
Fingolimod is used in the treatment of the relapsing form of multiple sclerosis.[7] Its effect in those with primary progressive MS is not clear.[7] It may also be used in chronic inflammatory demyelinating polyneuropathy.[7]
Dosage[edit | edit source]
It is generally taken at a dose of 500 micrograms per day.[2]
Side effects[edit | edit source]
The most common side effects is head colds, headache,[8] increased gamma-glutamyl transfer (≤15%), diarrhea (13%), nausea (13%), abdominal pain (11%)[8] and fatigue. A few cases of skin cancer have been reported, which has also been reported in patients taking natalizumab (Tysabri), an approved MS drug.[9] Fingolimod has also been associated with potentially fatal infections, bradycardia and, in 2009, a case of hemorrhaging focal encephalitis, an inflammation of the brain with bleeding.[10] Two people died: one due to brain herpes infection, and a second one due to herpes zoster. It is unclear whether the drug was responsible for the events.[11] At least three cases of progressive multifocal leukoencephalopathy had also occurred as of 2015.[12]
Fingolimod has also been known to cause macular edema, resulting in decreased vision.[13][14] Therefore, frequent surveillance eye examinations are required while taking this medication.
The European Medicines Agency (EMA) has advised doctors to increase their level of monitoring of people after the first dose of the medicine. This includes electrocardiogram (ECG) monitoring before treatment and then continuously for the first six hours after the first dose, and measurement of blood pressure and heart rate every hour.[15]
In the United States, fingolimod must be dispensed with a medication guide that contains important information about its uses and risks.[16] Serious risks include slowing of the heart rate, especially after the first dose.[16] Fingolimod may increase the risk of serious infections.[16] Patients should be monitored for infection during treatment and for two months after discontinuation of treatment.[16] A rare brain infection that usually leads to death or severe disability, called progressive multifocal leukoencephalopathy (PML) has been reported in patients being treated with the drug.[16] PML cases usually occur in patients with weakened immune systems.[16] Fingolimod can cause vision problems.[16] It may increase the risk for swelling and narrowing of the blood vessels in the brain (posterior reversible encephalopathy syndrome).[16] Other serious risks include respiratory problems, liver injury, increased blood pressure and skin cancer.[16] Fingolimod may cause harm to a developing fetus; health care professionals should advise women of child-bearing age of the potential risk to the fetus and to use effective contraception.[16]
The FDA has issued a safety alert warning that when fingolimod is stopped, multiple sclerosis (MS) can become much worse than before fingolimod was started or while it was being taken, and can result in permanent disability.[17]
Pregnancy[edit | edit source]
The European Medicines Agency (EMA) stated that the multiple sclerosis medicine fingolimod must not be used in pregnant women and in women able to have children who are not using effective contraception.[18]
Mechanism of action[edit | edit source]
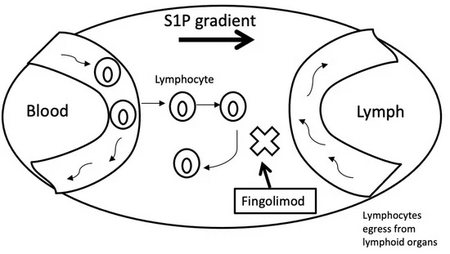
It is derived from myriocin (ISP-1), a metabolite of the fungus Isaria sinclairii. It is a structural analogue of sphingosine and is phosphorylated by sphingosine kinases in the cell (most importantly sphingosine kinase 2).[20][21][22] The molecular biology of phospho-fingolimod is thought to lie in its activity at one of the five sphingosine-1-phosphate receptors, S1PR1.[23] Phospho-fingolimod causes the internalization of S1P receptors, which sequesters lymphocytes in lymph nodes, preventing them from moving to the central nervous system and causing a relapse of multiple sclerosis.
The unphosphorylated moiety of fingolimod, which is the predominant form of the drug in the body, is also an active molecule. Unphosphorylated fingolimod impairs the ability of cytotoxic CD8 T cells to kill their target cells by a different mechanism, which involves the arachidonic acid pathway, which is unrelated to sphigosine phosphate receptors.[24] This has implications both for increasing susceptibility to viral infections as well as enhancing therapeutic efficacy in multiple sclerosis.[24]
Additionally, fingolimod shifts macrophages to an anti-inflammatory M2 phenotype. It modulates their proliferation, morphology, and cytokine release via inhibition of the transient receptor potential cation channel, subfamily M, member 7. (TRPM7).[25]
Finally, fingolimod has also been found to have other molecular targets and functions. Fingolimod has been reported to be a cannabinoid receptor antagonist,[26] a cPLA2 inhibitor[27] and a ceramide synthase inhibitor.[28][29] It has also been reported to stimulate the repair process of glial cells and glial precursor cells after injury.[30]
History[edit | edit source]
First synthesized in 1992 by Yoshitomi Pharmaceuticals, fingolimod was derived from an immunosuppressive natural product, myriocin (ISP-I) through chemical modification. Myriocin was isolated from the culture broth a type of entomopathogenic fungus (Isaria sinclairii) that was an eternal youth nostrum in traditional Chinese medicine.[31] Showing positive results in both in vitro (mixed lymphocyte reaction) and in vivo screening (prolonging rat skin graft survival time), myriocin was modified through a series of steps to yield fingolimod, code named at the time FTY720.[32] A recent review highlights the synthetic methods, mode of action and potential applications of this molecule.[33] Structure activity relationship (SAR) studies on myriocin homologs and partially synthetic derivatives showed that the configuration at the carbon bearing the 3-hydroxy group or the 14-ketone, the 6-double bond, and the 4-hydroxy group were not important for its activity and simplification of the structure of ISP-I was done in an attempt to reduce toxicity and improve drug ability.[31]
Elimination of side chain functionalities and removal of chiral centers was part of the simplification process and an intermediate compound (ISP-I-28) with the carboxylic acid of myriocin transformed to a hydroxymethyl group was generated. ISP-I-28 was found to be less toxic and more effective at lengthening rat skin allograft time than ISP-1.
In September 2010, fingolimod became the first oral disease-modifying drug approved by the U.S. Food and Drug Administration (FDA) to reduce relapses and delay disability progression in patients with relapsing forms of multiple sclerosis.[34][35] In April 2011 Novartis said that the drug would be available in Canadian pharmacies.[36][37] In March 2011, the European Medicines Agency approved the drug for use in the European Union.[38]
In 2015, after a challenge at the US Patent and Trademark Office by a generic competitor, the patent office quashed Novartis's patent claims stating they were obvious. Novartis appealed and the federal circuit upheld the patent office decision in April 2017, leaving a high likelihood of generics coming to market by 2019.[39]
In 2016, a systematic review concluded that treatment of people relapsing-remitting MS is effective in reducing the probability of acute inflammatory relapses, with potentially little or no effect on disability progression, compared to placebo.[40] The risk/benefit profile compared to other disease-modifiying therapies being unclear due to a lack of direct comparisons.[40]
In December 2019, generic fingolimod was approved in the United States for the treatment of relapsing forms of multiple sclerosis (MS) in adults.[16] The FDA granted approvals of generic fingolimod applications to HEC Pharm Co. Limited, Biocon Limited and Sun Pharmaceutical Industries Limited.[16]
On July 19, 2019, fingolimod received fast approval for use in China.[41]
On January 9, 2020, a panel of judges at the Court of Appeal for the Federal Circuit called into question the validity of the last remaining orange book patent protecting Gilenya.[42]
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 "Fingolimod Monograph for Professionals". Drugs.com. Archived from the original on 16 June 2018. Retrieved 10 December 2021.
- ↑ 2.0 2.1 2.2 2.3 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 901. ISBN 978-0857114105.
- ↑ Sanford M (August 2014). "Fingolimod: a review of its use in relapsing-remitting multiple sclerosis". Drugs. 74 (12): 1411–33. doi:10.1007/s40265-014-0264-y. PMID 25063048. S2CID 42807019.
- ↑ 4.0 4.1 "Gilenya". Archived from the original on 29 September 2021. Retrieved 10 December 2021.
- ↑ "Gilenya Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 17 May 2020. Retrieved 10 December 2021.
- ↑ Research, Center for Drug Evaluation and (10 February 2022). "2021 First Generic Drug Approvals". FDA. Archived from the original on 21 June 2022. Retrieved 22 October 2022.
- ↑ 7.0 7.1 7.2 "Fingolimod Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 16 June 2018. Retrieved 9 August 2015.
- ↑ 8.0 8.1 "UpToDate". www.uptodate.com. Archived from the original on 29 September 2021. Retrieved 24 June 2019.
- ↑ "Good News for Oral MS Drug Fingolimod". Webmd.com. 16 April 2008. Archived from the original on 13 July 2018. Retrieved 30 September 2013.
- ↑ Leypoldt F, Münchau A, Moeller F, Bester M, Gerloff C, Heesen C (March 2009). "Hemorrhaging focal encephalitis under fingolimod (FTY720) treatment: a case report". Neurology. 72 (11): 1022–4. doi:10.1212/01.wnl.0000344567.51394.e3. PMID 19289744. S2CID 8523513.
- ↑ "MS-UK | Multiple Sclerosis Information, Helpline, support, MS news and research". Archived from the original on 19 March 2015. Retrieved 26 August 2021.
- ↑ Brooks, Megan (18 August 2015). "Third Case of PML With Fingolimod (Gilenya) in MS". Medscape. Archived from the original on 8 October 2017. Retrieved 20 August 2015.
- ↑ Jain N, Bhatti MT (February 2012). "Fingolimod-associated macular edema: incidence, detection, and management". Neurology. 78 (9): 672–80. doi:10.1212/WNL.0b013e318248deea. PMID 22371414. S2CID 11742356.
- ↑ Jain N, Bhatti MT (April 2012). "Macular Edema Associated With Fingolimod" (PDF). EyeNet. 78 (9): 672–80. doi:10.1212/WNL.0b013e318248deea. PMID 22371414. S2CID 11742356. Archived from the original (PDF) on 16 June 2016. Retrieved 20 August 2015.
- ↑ "European Medicines Agency - News and Events - European Medicines Agency starts review of Gilenya (fingolimod)". Ema.europa.eu. 20 January 2012. Archived from the original on 20 June 2018. Retrieved 30 September 2013.
- ↑ 16.00 16.01 16.02 16.03 16.04 16.05 16.06 16.07 16.08 16.09 16.10 16.11 "FDA approves first generics of Gilenya". U.S. Food and Drug Administration (FDA) (Press release). 5 December 2019. Archived from the original on 5 December 2019. Retrieved 5 December 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "FDA warns about severe worsening of multiple sclerosis after stopping the medicine Gilenya (fingolimod)". U.S. Food and Drug Administration (FDA). 20 November 2018. Archived from the original on 6 December 2019. Retrieved 5 December 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Updated restrictions for Gilenya: multiple sclerosis medicine not to be used in pregnancy". European Medicines Agency (EMA) (Press release). 26 July 2019. Archived from the original on 13 July 2020. Retrieved 12 July 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ Zarzuelo Romero, María José; Pérez Ramírez, Cristina; Carrasco Campos, María Isabel; Sánchez Martín, Almudena; Calleja Hernández, Miguel Ángel; Ramírez Tortosa, María Carmen; Jiménez Morales, Alberto (May 2021). "Therapeutic Value of Single Nucleotide Polymorphisms on the Efficacy of New Therapies in Patients with Multiple Sclerosis". Journal of Personalized Medicine. 11 (5): 335. doi:10.3390/jpm11050335. ISSN 2075-4426.
- ↑ Paugh SW, Payne SG, Barbour SE, Milstien S, Spiegel S (November 2003). "The immunosuppressant FTY720 is phosphorylated by sphingosine kinase type 2". FEBS Letters. 554 (1–2): 189–93. doi:10.1016/S0014-5793(03)01168-2. PMID 14596938. S2CID 41465940.
- ↑ Billich A, Bornancin F, Dévay P, Mechtcheriakova D, Urtz N, Baumruker T (November 2003). "Phosphorylation of the immunomodulatory drug FTY720 by sphingosine kinases". The Journal of Biological Chemistry. 278 (48): 47408–15. doi:10.1074/jbc.M307687200. PMID 13129923. Free full text Archived 13 June 2008 at the Wayback Machine
- ↑ Sanchez T, Estrada-Hernandez T, Paik JH, Wu MT, Venkataraman K, Brinkmann V, Claffey K, Hla T (November 2003). "Phosphorylation and action of the immunomodulator FTY720 inhibits vascular endothelial cell growth factor-induced vascular permeability". The Journal of Biological Chemistry. 278 (47): 47281–90. doi:10.1074/jbc.M306896200. PMID 12954648.
- ↑ Hla T, Lee MJ, Ancellin N, Paik JH, Kluk MJ (November 2001). "Lysophospholipids--receptor revelations". Science. 294 (5548): 1875–8. Bibcode:2001Sci...294.1875H. doi:10.1126/science.1065323. PMID 11729304. S2CID 46727063.
- ↑ 24.0 24.1 Ntranos A, Hall O, Robinson DP, Grishkan IV, Schott JT, Tosi DM, Klein SL, Calabresi PA, Gocke AR (May 2014). "FTY720 impairs CD8 T-cell function independently of the sphingosine-1-phosphate pathway". Journal of Neuroimmunology. 270 (1–2): 13–21. doi:10.1016/j.jneuroim.2014.03.007. PMID 24680062. S2CID 206276944. Archived from the original on 29 September 2021. Retrieved 26 August 2021.
- ↑ Schilling T, Miralles F, Eder C (November 2014). "TRPM7 regulates proliferation and polarisation of macrophages". Journal of Cell Science. 127 (Pt 21): 4561–6. doi:10.1242/jcs.151068. PMC 4215710. PMID 25205764.
- ↑ Paugh SW, Cassidy MP, He H, Milstien S, Sim-Selley LJ, Spiegel S, Selley DE (July 2006). "Sphingosine and its analog, the immunosuppressant 2-amino-2-(2-[4-octylphenyl]ethyl)-1,3-propanediol, interact with the CB1 cannabinoid receptor". Molecular Pharmacology. 70 (1): 41–50. doi:10.1124/mol.105.020552. PMID 16571654. S2CID 11131541.
- ↑ Payne SG, Oskeritzian CA, Griffiths R, Subramanian P, Barbour SE, Chalfant CE, Milstien S, Spiegel S (February 2007). "The immunosuppressant drug FTY720 inhibits cytosolic phospholipase A2 independently of sphingosine-1-phosphate receptors". Blood. 109 (3): 1077–85. doi:10.1182/blood-2006-03-011437. PMC 1785128. PMID 17008548.
- ↑ Berdyshev EV, Gorshkova I, Skobeleva A, Bittman R, Lu X, Dudek SM, Mirzapoiazova T, Garcia JG, Natarajan V (February 2009). "FTY720 inhibits ceramide synthases and up-regulates dihydrosphingosine 1-phosphate formation in human lung endothelial cells". The Journal of Biological Chemistry. 284 (9): 5467–77. doi:10.1074/jbc.M805186200. PMC 2645812. PMID 19119142.
- ↑ Lahiri S, Park H, Laviad EL, Lu X, Bittman R, Futerman AH (June 2009). "Ceramide synthesis is modulated by the sphingosine analog FTY720 via a mixture of uncompetitive and noncompetitive inhibition in an Acyl-CoA chain length-dependent manner". The Journal of Biological Chemistry. 284 (24): 16090–8. doi:10.1074/jbc.M807438200. PMC 2713526. PMID 19357080.
- ↑ Horga A, Montalban X (May 2008). "FTY720 (fingolimod) for relapsing multiple sclerosis". Expert Review of Neurotherapeutics. 8 (5): 699–714. doi:10.1586/14737175.8.5.699. PMID 18457527. S2CID 28071687.
- ↑ 31.0 31.1 Adachi K, Chiba K (September 2007). "FTY720 story. Its discovery and the following accelerated development of sphingosine 1-phosphate receptor agonists as immunomodulators based on reverse pharmacology". Perspectives in Medicinal Chemistry. 1: 11–23. doi:10.1177/1177391X0700100002. PMC 2754916. PMID 19812733.
- ↑ Fujita T, Yoneta M, Hirose R, Sasaki S, Inoue K, Kiuchi M, Hirase S, Adachi K, Arita M, Chiba K (1995). "Simple compounds, 2-alkyl-2-amino-1,3-propanediols have potent immunosuppressive activity". Bioorg. Med. Chem. Lett. 5 (8): 847–52. doi:10.1016/0960-894X(95)00126-E.
- ↑ Balasubramaniam S, Sankaran GS, Badle SS (2018). "Perspective on FTY720, an Immunosuppressant". Synthesis. 50 (5): 968–83. doi:10.1055/s-0036-1591877.
- ↑ "FDA approves first oral drug to reduce MS relapses". U.S. Food and Drug Administration (FDA). 22 September 2010. Archived from the original on 14 February 2017. Retrieved 5 December 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Drug Approval Package: Brand Name (Generic Name) NDA #". U.S. Food and Drug Administration (FDA). 5 December 2019. Archived from the original on 5 December 2019. Retrieved 5 December 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ First oral MS treatment approved for Canada https://vancouversun.com/health/First+oral+treatment+approved+Canada+says+drug+company/4420028/story.html Archived 27 April 2011 at the Wayback Machine
- ↑ "Novartis new MS treatment receives Notice of Compliance in Canada". NewsMedical.net. 10 March 2011. Archived from the original on 16 June 2018. Retrieved 26 August 2021.
- ↑ "EMA approval information about Gilenya". Archived from the original on 14 April 2018. Retrieved 14 March 2022.
- ↑ Sagonowsky, Eric (13 April 2017). "Novartis' Gilenya patent loss sets MS market up for battle with early generics". FiercePharma. Archived from the original on 25 October 2020. Retrieved 26 August 2021.
- ↑ 40.0 40.1 La Mantia, Loredana; Tramacere, Irene; Firwana, Belal; Pacchetti, Ilaria; Palumbo, Roberto; Filippini, Graziella (19 April 2016). "Fingolimod for relapsing-remitting multiple sclerosis". The Cochrane Database of Systematic Reviews. 4: CD009371. doi:10.1002/14651858.CD009371.pub2. ISSN 1469-493X. PMID 27091121.
- ↑ "Novartis key multiple sclerosis product Gilenya approved in China". Novartis (Press release). Archived from the original on 16 January 2021. Retrieved 26 August 2021.
- ↑ "CAFC Oral Argument Recording". Archived from the original on 29 September 2021. Retrieved 26 August 2021.
External links[edit | edit source]
| External sites: | |
|---|---|
| Identifiers: |
|
- "Fingolimod hydrochloride". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 29 September 2021. Retrieved 26 August 2021.
Original source: https://mdwiki.org/wiki/Fingolimod
Status: article is cached
 KSF
KSF