Nesiritide
 From Mdwiki - Reading time: 8 min
From Mdwiki - Reading time: 8 min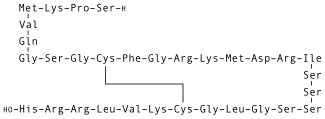 | |
| Clinical data | |
|---|---|
| Drug class | Vasodilatation[1] |
| Main uses | Heart failure[1] |
| Side effects | Low blood pressure, headache, nausea, back pain[1] |
| Routes of use | IV only |
| External links | |
| AHFS/Drugs.com | Monograph |
| Chemical and physical data | |
| Formula | C143H244N50O42S4 |
| Molar mass | 3464.07 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Nesiritide, sold under the brand name Natrecor among others, is a medication developed to treat acute decompensated congestive heart failure.[1] Evidence; however, has failed to show benefit.[2][1] It is given into a vein.[1]
Common side effects include low blood pressure, headache, nausea, and back pain.[1] Other side effects may include allergic reactions, kidney problems, and death.[1] It is a recombinant form of B-type natriuretic peptide, which results in vasodilatation.[1]
Nesiritide was approved for medical use in the United States in 2001.[1] In the United States it costs about 1,100 USD per 1.5 mg vial.[3] Commercial sale; however, has been discontinued as of 2021.[4]
Medical uses[edit | edit source]
Dosage[edit | edit source]
Nesiritide is only administered intravenously, usually by bolus, followed by IV infusion. For most adults and the elderly, a normal dosage is 2 mg/kg followed by a continuous IV infusion of 0.01 mg/kg/min. This may be increased every three hours for a maximum of 0.03 mg/kg/min.
Side effects[edit | edit source]
Common side effects include:
More rare side effects include:
Controversy[edit | edit source]
In 2005, after several academic papers published by Jonathan Sackner-Bernstein[6][7][8] on the efficacy and side effects of nesiritide, Johnson & Johnson met with the FDA and altered its stated plans for the drug and agreed to revise its labeling.[9][10]
Heart doctors at the Cleveland Clinic then voted unanimously not to permit the prescription of the drug.[10] Johnson and Johnson convened a panel of experts whose advice included the recommendation to conduct the large-scale clinical trial that was subsequently published in 2011.[11] Following this, the United States Department of Justice announced an inquiry into the marketing of the drug[12] that led to a fine against the Scios unit of J&J.[13]
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 "Nesiritide Monograph for Professionals". Drugs.com. Archived from the original on 22 June 2020. Retrieved 12 November 2021.
- ↑ O'Connor CM, Starling RC, Hernandez AF, Armstrong PW, Dickstein K, Hasselblad V, et al. (July 2011). "Effect of nesiritide in patients with acute decompensated heart failure" (PDF). The New England Journal of Medicine. 365 (1): 32–43. doi:10.1056/NEJMoa1100171. hdl:11379/60663. PMID 21732835. Archived (PDF) from the original on 2021-02-11. Retrieved 2021-09-18.
- ↑ "Natrecor Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 12 November 2021.
- ↑ "Drugs@FDA: FDA-Approved Drugs". www.accessdata.fda.gov. Archived from the original on 21 March 2021. Retrieved 12 November 2021.
- ↑ "Natrecor Adverse Reactions - Epocrates Online". online.epocrates.com. Archived from the original on 2020-03-08. Retrieved 2021-09-18.
- ↑ Sackner-Bernstein JD, Skopicki HA, Aaronson KD (March 2005). "Risk of worsening renal function with nesiritide in patients with acutely decompensated heart failure". Circulation. 111 (12): 1487–91. doi:10.1161/01.CIR.0000159340.93220.E4. PMID 15781736.
- ↑ Sackner-Bernstein JD, Kowalski M, Fox M, Aaronson K (April 2005). "Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials". JAMA. 293 (15): 1900–5. doi:10.1001/jama.293.15.1900. PMID 15840865.
- ↑ Aaronson KD, Sackner-Bernstein J (September 2006). "Risk of death associated with nesiritide in patients with acutely decompensated heart failure". JAMA. 296 (12): 1465–6. doi:10.1001/jama.296.12.1465. PMID 17003394.
- ↑ Saul S (17 May 2005). "The Marketing and Success of Natrecor". Archived from the original on 4 September 2021. Retrieved 18 September 2021 – via NYTimes.com.
- ↑ 10.0 10.1 Saul S (4 May 2005). "Heart Clinic May End or Curtail Use of a Drug". Archived from the original on 4 June 2019. Retrieved 18 September 2021 – via NYTimes.com.
- ↑ Herper M. "Bitter Pill For J&J On Heart Drug". Forbes.
{{cite web}}: Check|url=value (help)CS1 maint: url-status (link) - ↑ Saul S (21 July 2005). "U.S. Looking at Marketing by Johnson & Johnson". Archived from the original on 23 March 2021. Retrieved 18 September 2021 – via NYTimes.com.
- ↑ "Johnson & Johnson Subsidiary Scios Pleads Guilty to Misbranding Heart Failure Drug Natrecor". www.justice.gov. 5 October 2011. Archived from the original on 12 June 2021. Retrieved 18 September 2021.
External links[edit | edit source]
| Identifiers: |
|
|---|
- Noviasky JA, Kelberman M, Whalen KM, Guharoy R, Darko W (August 2003). "Science or fiction: use of nesiritide as a first-line agent?". Pharmacotherapy. 23 (8): 1081–3. doi:10.1592/phco.23.8.1081.32882. PMC 3746126. PMID 12921256.
Original source: https://mdwiki.org/wiki/Nesiritide
Status: article is cached
 KSF
KSF