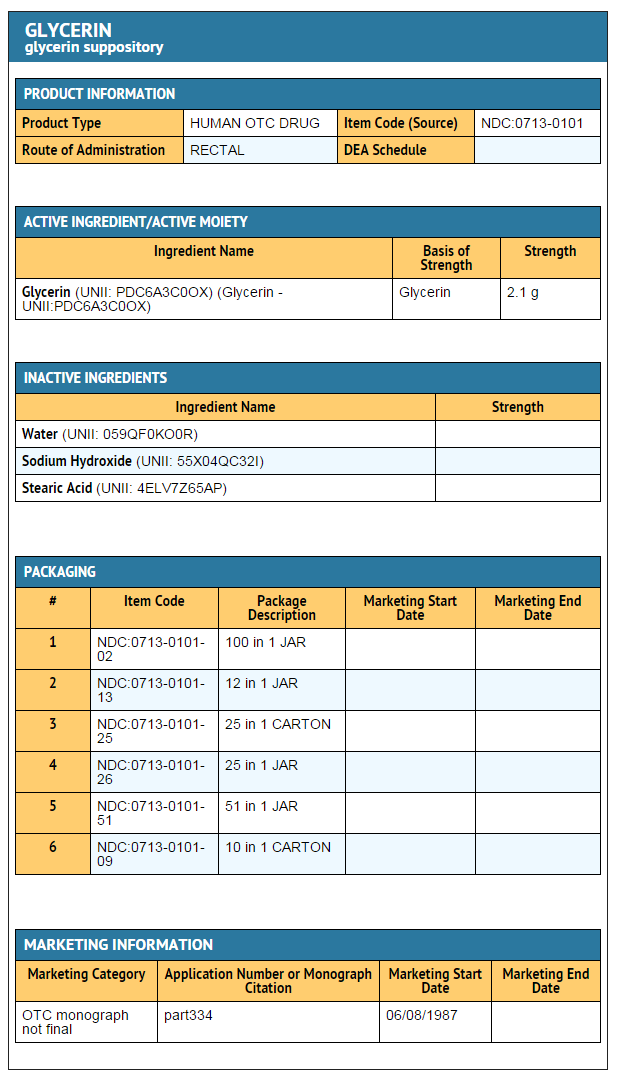
Glycerin
 From Wikidoc - Reading time: 5 min
From Wikidoc - Reading time: 5 minEditor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
NOTE: Most over the counter (OTC) are not reviewed and approved by the FDA. However, they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Overview
Glycerin is a Laxative that is FDA approved for the treatment of constipation. Common adverse reactions include rectal discomfort or a burning sensation, diarrhea.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 1/4 to 1 hour
Directions
- insert 1 suppository well up into rectum and retain for 15 minutes
- it need not melt to produce laxative action
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Glycerin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Glycerin in adult patients.
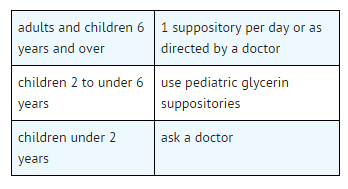
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Indications
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 1/4 to 1 hour
Directions

- insert 1 suppository well up into rectum and retain for 15 minutes
- it need not melt to produce laxative action
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Glycerin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Glycerin in pediatric patients.
Contraindications
There is limited information regarding Glycerin Contraindications in the drug label.
Warnings
- For rectal use only
- Do not use laxative products for longer than 1 week unless directed by a doctor
- Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that lasts over 2 weeks
- When using this product you may have rectal discomfort or a burning sensation
- Stop use and ask a doctor if you have rectal bleeding or fail to have a bowel movement after use of a laxative. These could be signs of a serious condition.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Glycerin in the drug label.
Postmarketing Experience
- rectal discomfort or a burning sensation, diarrhea
Drug Interactions
There is limited information regarding Glycerin Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- IF PREGNANT OR BREAST FEEDING
- ask a health professional before use.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Glycerin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Glycerin during labor and delivery.
Nursing Mothers
- IF PREGNANT OR BREAST FEEDING
- ask a health professional before use.
Pediatric Use
There is no FDA guidance on the use of Glycerin with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Glycerin with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Glycerin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Glycerin with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Glycerin in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Glycerin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Glycerin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Glycerin in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
There is limited information regarding Monitoring of Glycerin in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Glycerin in the drug label.
Overdosage
- If swallowed, get medical help or contact a Poison Control Center right away.
Pharmacology
Template:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox entryTemplate:Chembox E numberTemplate:Chembox AppearanceTemplate:Chembox OdourTemplate:Chembox DensityTemplate:Chembox MeltingPtTemplate:Chembox BoilingPtTemplate:Chembox SolubilityInWaterTemplate:Chembox VaporPressureTemplate:Chembox RefractIndexTemplate:Chembox ViscosityTemplate:Chembox NFPATemplate:Chembox FlashPtTemplate:Chembox NIOSH (set)Template:Chembox Supplement| Template:Chembox header2 | Glycerol | |
|---|---|
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | Lua error in Module:Wikidata at line 879: attempt to index field 'wikibase' (a nil value). Lua error in Module:Wikidata at line 879: attempt to index field 'wikibase' (a nil value). |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C3H8O3 | |
| Molar mass | 92.09 g·mol−1 |
| Hazards | |
| Template:Chembox header2 | Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |
Mechanism of Action
Structure
- ACTIVE INGREDIENT (IN EACH SUPPOSITORY)
- Glycerin, USP 2.1 grams
- INACTIVE INGREDIENT
- purified water, sodium hydroxide, stearic acid
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Glycerin in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Glycerin in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Glycerin in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Glycerin in the drug label.
How Supplied
There is limited information regarding Glycerin How Supplied in the drug label.
Storage
There is limited information regarding Glycerin Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Glycerin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Ingredients and Appearance
{{#ask: Label Page::Glycerin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Glycerin in the drug label.
Precautions with Alcohol
- Alcohol-Glycerin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- GLYCERIN®[4]
Look-Alike Drug Names
There is limited information regarding the look alike drug names.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ 1.0 1.1 1.2 1.3 1.4 Template:PGCH
- ↑ Template:Cite doi
- ↑ Lide, D. R., ed. (1994). CRC Handbook of Data on Organic Compounds (3rd ed.). Boca Raton, FL: CRC Press. p. 4386.
- ↑ "Glycerin suppository".
Original source: https://wikidoc.org/Glycerin
Status: article is cached
 KSF
KSF