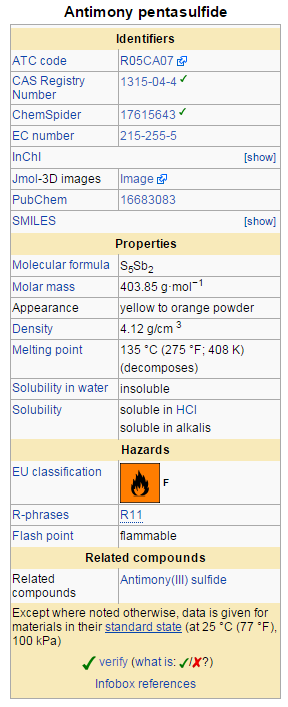
Antimony pentasulfide
 From Wikidoc - Reading time: 3 min
From Wikidoc - Reading time: 3 min
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
[edit | edit source]Antimony pentasulfide is an inorganic compound of antimony and sulfur, also known as antimony red. It is a nonstoichiometric compound with a variable composition. Its exact structure is unknown.[1] Commercial samples are usually are contaminated with sulfur, which may be removed by washing with carbon disulfide in a Soxhlet extractor.
Production
[edit | edit source]Antimony pentasulfide can be produced by the reaction of antimony with sulfur at a temperature between 250-400 °C in an inert atmosphere.
Uses
[edit | edit source]It may be used as a red pigment and is one possible precursor to Schlippe's Salt, Na3SbS4, which can be prepared according to the equation:
- 3 Na2S + Sb2S5 + 9 H2O → 2 Na3SbS4·9H2O
Physical chemistry
[edit | edit source]Like many sulfides, this compound liberates hydrogen sulfide upon treatment with strong acids like hydrochloric acid.[2]
- 6 HCl + Sb2S5 → 2 SbCl3 + 3 H2S + 2 S
Analysis by Mössbauer spectroscopy indicates that this compound is a derivative antimony(III),[3] explaining the production of antimony(III) chloride, rather than antimony(V) chloride, upon acidification. It is therefore not analogous to the phosphorus(V) compound phosphorus pentasulfide.
References
[edit | edit source]- ↑ Arnold F. Holleman, Nils Wiberg: Lehrbuch der Anorganischen Chemie, 102nd edition, de Gruyter, Berlin 2007, p. 849, ISBN 978-3-11-017770-1.
- ↑ Strem MSDS
- ↑ G. G. Long, J. G. Stevens, L. H. Bowen, S. L. Ruby (1969). "The oxidation number of antimony in antimony pentasulfide". Inorganic and Nuclear Chemistry Letters. 5 (1): 21–25. doi:10.1016/0020-1650(69)80231-X.
Template:Antimony compounds Template:Cough and cold preparations
 KSF
KSF