Chlamydia infection pathophysiology
 From Wikidoc - Reading time: 4 min
From Wikidoc - Reading time: 4 min
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: Maliha Shakil, M.D. [2], Aysha Anwar, M.B.B.S[3]
|
Chlamydia infection Microchapters |
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Chlamydia infection pathophysiology On the Web |
|
American Roentgen Ray Society Images of Chlamydia infection pathophysiology |
|
Risk calculators and risk factors for Chlamydia infection pathophysiology |
Overview
[edit | edit source]Chlamydia infection may be transmitted during vaginal, anal, or oral sex, and can be passed from an infected mother to her baby during childbirth. Chlamydiae are obligate intracellular bacterial pathogens which have the ability to establish long-term associations with host cells. These pathogens have evolved a distinct biphasic life cycle wherein they alternate between two functionally and morphologically distinct forms: elementary body (EB) and reticulate body (RB).[1][2][3][4][5][6][7][8]
Pathophysiology
[edit | edit source]Chlamydia infection can be transmitted during vaginal, anal, or oral sex, and can be passed from an infected mother to her baby during childbirth.
Chlamydial Life Cycle
[edit | edit source]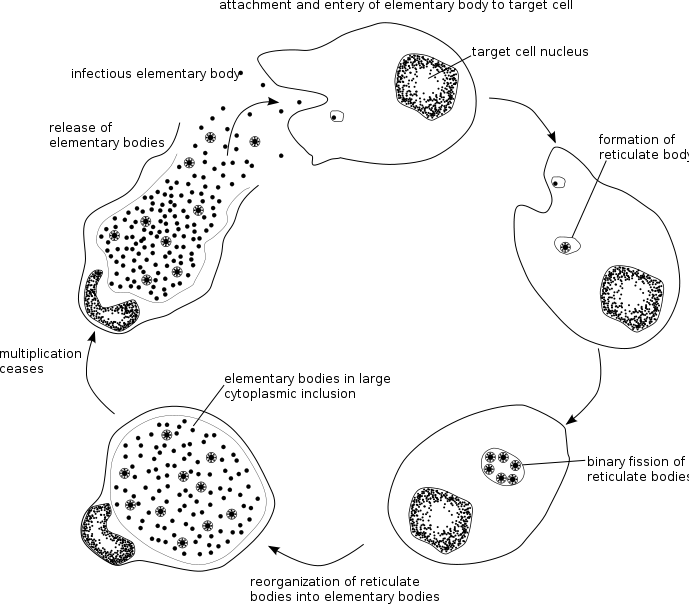
Chlamydiae are obligate intracellular bacterial pathogens, which means they are unable to replicate outside of a host cell. However, to disseminate effectively, these pathogens have evolved a distinct biphasic life cycle wherein they alternate between two functionally and morphologically distinct forms.[9][10]
- The elementary body (EB) is infectious, but metabolically inert (much like a spore), and can survive for limited amounts of time in the extracellular milieu. Once the EB attaches to a susceptible host cell, it mediates its own internalization through pathogen-specified mechanisms (via type III secretion system) that allow for the recruitment of actin with subsequent engulfment of the bacterium.
- The internalized EB, within a membrane-bound compartment, immediately begins differentiation into the reticulate body (RB). RBs are metabolically active but non-infectious; in many regards, they resemble normal replicating bacteria. The intracellular bacteria rapidly modifies its membrane-bound compartment into the so-called chlamydial inclusion so as to prevent phagosome-lysosome fusion. The inclusion is thought to have no interactions with the endocytic pathway and apparently inserts itself into the exocytic pathway as it retains the ability to intercept sphingomyelin-containing vesicles.
- The mechanism by which the host cell protein is trafficked to the inclusion through the exocytic pathway is not fully understood. As the RBs replicate, the inclusion grows as well to accommodate the increasing numbers of organisms. Through unknown mechanisms, RBs begin a differentiation program back to the infectious EBs, which are released from the host cell to initiate a new round of infection. Because of their obligate intracellular nature, Chlamydiae have no tractable genetic system, unlike E. coli, which makes Chlamydiae and related organisms difficult to investigate.
Persistence
[edit | edit source]Chlamydiae have the ability to establish long-term associations with host cells. When an infected host cell is starved for various nutrients such as amino acids (e.g. tryptophan),[3] iron, or vitamins, this has a negative consequence for Chlamydiae since the organism depends upon the host cell for these nutrients. The starved chlamydiae enter a persistent growth state wherein they stop cell division and become morphologically aberrant by increasing in size.[4] Persistent organisms remain viable as they are capable of returning to a normal growth state once conditions in the host cell improve. There is much debate as to whether persistence has in-vivo relevance. Many believe that persistent chlamydiae are the cause of chronic chlamydial diseases. Some antibiotics, such as β-lactams, can also induce a persistent-like growth state, which can contribute to the chronicity of chlamydial diseases.
Diseases caused by Chlamydia Trachomatis
[edit | edit source]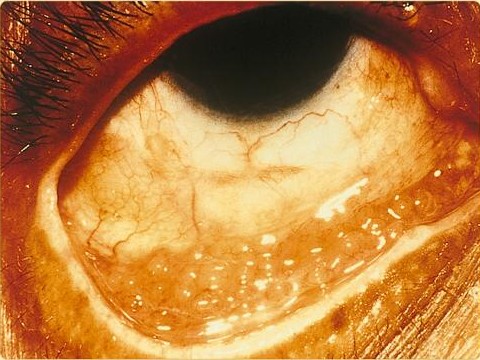
Chlamydia trachomatis can cause the following conditions:[5][6][7][8]
References
[edit | edit source]- ↑ Beatty, Wandy L., Richard P. Morrison, and Gerald I. Byrne. "Persistent chlamydiae: from cell culture to a paradigm for chlamydial pathogenesis." Microbiological reviews 58.4 (1994): 686-699.
- ↑ Baron, Samuel. Medical microbiology. Galveston, Tex: University of Texas Medical Branch at Galveston, 1996. Print.
- ↑ 3.0 3.1 Leonhardt RM, Lee SJ, Kavathas PB, Cresswell P (2007). "Severe Tryptophan Starvation Blocks Onset of Conventional Persistence and Reduces Reactivation of Chlamydia trachomatis". Infect. Immun. 75 (11): 5105–17. doi:10.1128/IAI.00668-07. PMID 17724071.
- ↑ 4.0 4.1 Mpiga P, Ravaoarinoro M (2006). "Chlamydia trachomatis persistence: an update". Microbiol. Res. 161 (1): 9–19. doi:10.1016/j.micres.2005.04.004. PMID 16338585.
- ↑ 5.0 5.1 Paroli E, Franco E (1990). "[Oculogenital infections caused by Chlamydia trachomatis]". Recenti Prog Med. 81 (7–8): 539–48. PMID 2247702.
- ↑ 6.0 6.1 Holstege G, van Ham JJ, Tan J (1986). "Afferent projections to the orbicularis oculi motoneuronal cell group. An autoradiographical tracing study in the cat". Brain Res. 374 (2): 306–20. PMID 3719340.
- ↑ 7.0 7.1 Feltham N, Fahey D, Knight E (1987). "A growth inhibitory protein secreted by human diploid fibroblasts. Partial purification and characterization". J Biol Chem. 262 (5): 2176–9. PMID 3818592.
- ↑ 8.0 8.1 Peipert JF (2003). "Clinical practice. Genital chlamydial infections". N Engl J Med. 349 (25): 2424–30. doi:10.1056/NEJMcp030542. PMID 14681509.
- ↑ Beatty, Wandy L., Richard P. Morrison, and Gerald I. Byrne. "Persistent chlamydiae: from cell culture to a paradigm for chlamydial pathogenesis." Microbiological reviews 58.4 (1994): 686-699.
- ↑ Baron, Samuel. Medical microbiology. Galveston, Tex: University of Texas Medical Branch at Galveston, 1996. Print.
 KSF
KSF