Coronary artery bifurcation
 From Wikidoc - Reading time: 20 min
From Wikidoc - Reading time: 20 min
|
Coronary Angiography | |
|
General Principles | |
|---|---|
|
Anatomy & Projection Angles | |
|
Normal Anatomy | |
|
Anatomic Variants | |
|
Projection Angles | |
|
Epicardial Flow & Myocardial Perfusion | |
|
Epicardial Flow | |
|
Myocardial Perfusion | |
|
Lesion Complexity | |
|
ACC/AHA Lesion-Specific Classification of the Primary Target Stenosis | |
|
Lesion Morphology | |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-In-Chief: Lisa Battaglia, M.D., Xin Yang, M.D., Arzu Kalayci, M.D.
Overview
[edit | edit source]Bifurcation lesions occur when the atherosclerotic plaque involves the origin of two separate arteries. A bifurcation is defined as a division of a main, parent branch into two daughter branches of at least 2.0 mm. Bifurcation lesions in coronary artery disease (CAD) are common, encompassing 15-18% of lesions treated with percutaneous coronary intervention (PCI). The optimal treatment for these lesions is subject to intense debate; current practice includes a variety of approaches. There is no data to suggest that stenting of a side branch improves outcomes over conventional balloon dilation of the side branch origin. In fact, sidebranch stenting may be associated with a higher risk of stent thrombosis. The consensus view at this time is that if there is adequate flow in the side branch and no evidence of ischemia at the end of the procedure, then further dilations of the side branch are not warranted.
Pathophysiology
[edit | edit source]Intravascular ultrasound indicates that the majority of plaque burden resides in the hips of the lesion and not at the flow divider or carina of the lesion. The hips of the bifurcation are areas of low shear stress where plaque accumulates.
Classification
[edit | edit source]Bifurcation lesions are classified according to the angulation of the bifurcation and plaque burden. This determines the ease of access to the side branch, plaque shift, and hence the preferred treatment strategy.
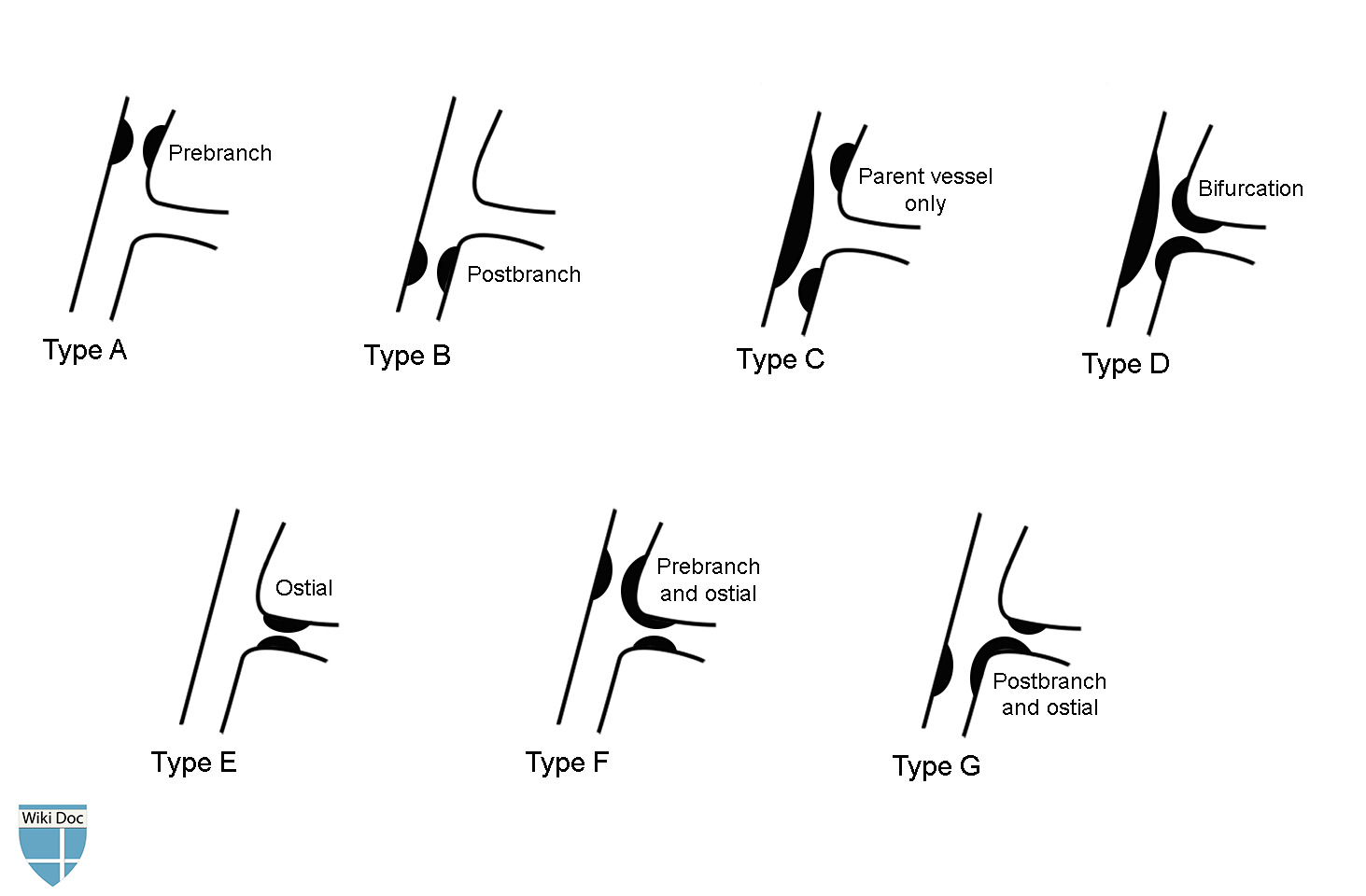
Several classifications have been proposed over the years to help better define the anatomy in bifurcation lesions. The original schemes published by Sanborn,[1] Safian,[2] Lefevre,[3] and Duke[4] are similar in their approach of using numbers or letters to represent various lesion types. However the absence of intuitive correlations between the various lesion type and associated classifications makes it difficult to remember and apply these classifications in routine clinical practice.
Shown below is an image depicting the modified Duke and ICPS classification systems for coronary artery bifurcation.
Image is adapted from definition and classification of bifurcation lesions and treatments.[5]
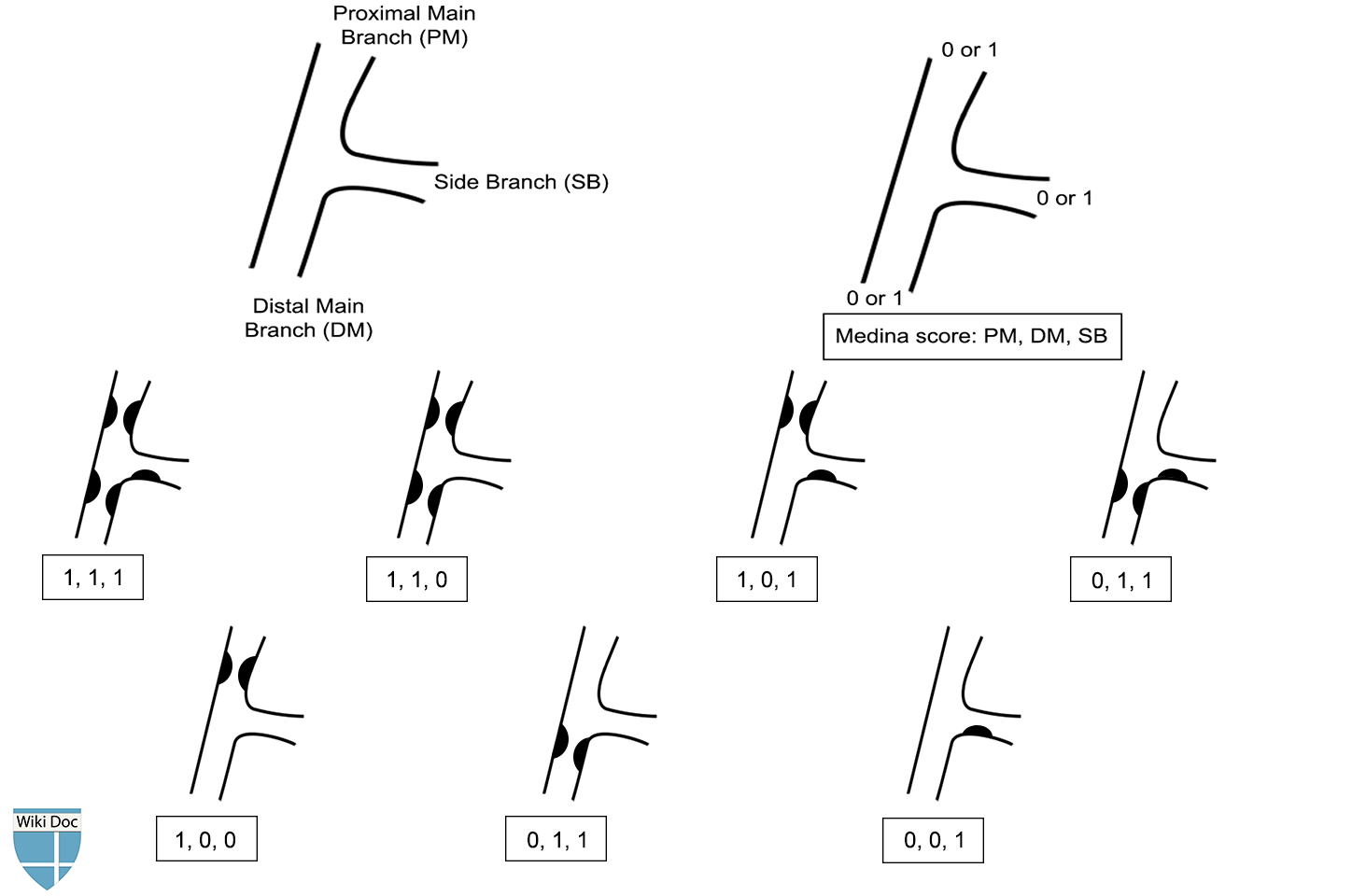
Medina et al.[6] subsequently published their classification which assigns a suffix of 1 (diseased) or 0 (not diseased) to the proximal main branch, side branch and distal main branch, respectively. According to Medina classification, a diseased artery is defined as having a stenosis of ≥ 50%. This classification allows for easy remembrance and it has since become the most commonly used scheme for defining anatomy of bifurcation lesions. However, Medina classification does not take into account the side branch size nor the side branch angle.
Shown below is an image depicting Medina classification for coronary artery bifurcation.
Image is adapted from classification and treatment of coronary artery bifurcation lesions: putting the Medina classification to the test.[7]
Diagnosis
[edit | edit source]Angiography
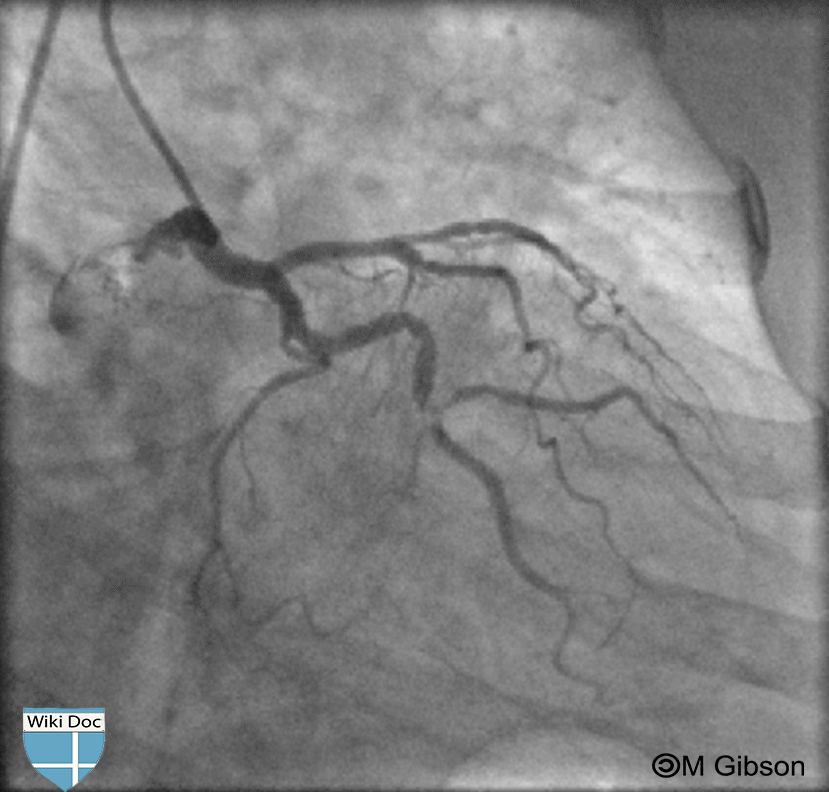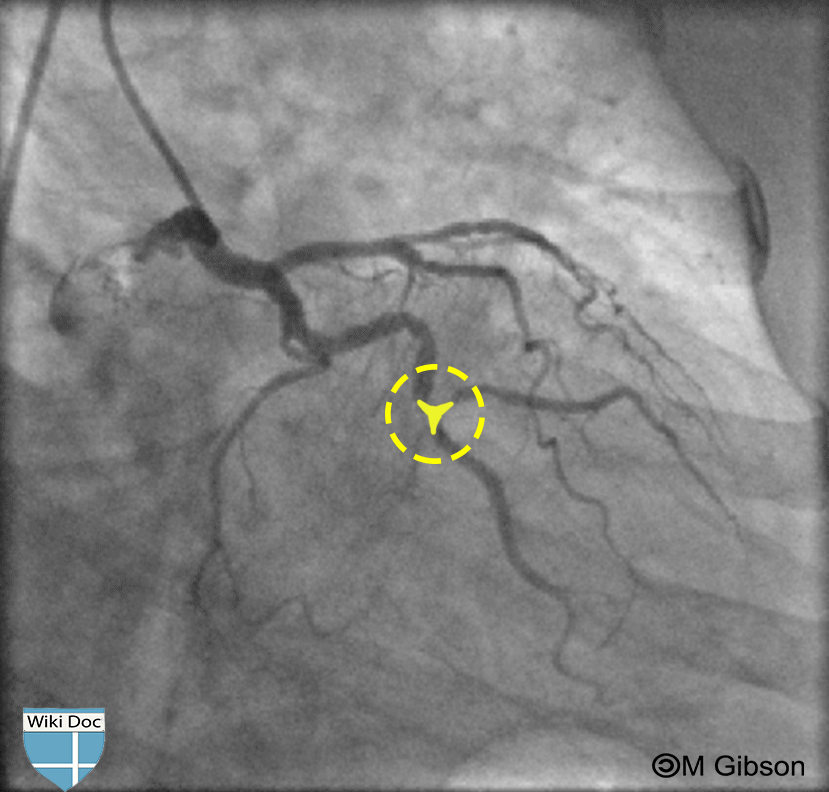
[edit | edit source]Coronary artery bifurcation is detected on angiography. Shown below are an animated and a static angiogrpahy images depicting bifurcation of a left coronary artery lesion. Encircled in yellow in the image on the right is the bifurcated lesion. Note that the lesion at the bifurcation has a "Mercedes" like shape which is outlined in yellow in the picture on the right.
PCI in Coronary Artery Bifurcation
[edit | edit source]Goals of PCI
[edit | edit source]The goal of PCI in the bifurcation lesion is to:
- Maximize flow in both the parent vessel while maintaining flow in the side branch
- Prevent side branch occlusion or compromise
- Maximize long-term patency of both parent vessel and side branch
Risks of Side Branch Involvement in Main Vessel Interventions
[edit | edit source]Early studies demonstrated that the occlusion of a side branch (with or without baseline disease in the side branch) during coronary angioplasty led to a higher rate of peri-procedural myocardial infarction (MI).
In the stent era, the use of angioplasty in a compromised side branch can lead to stent deformation in the main branch. Studies have also demonstrated that patients undergoing PCI of a main vessel were at an increased risk for a MI within 30 days of their procedure if the stent compromised a side branch.
If there is a lesion at the origin of a side branch, the consensus view is that this should be dilated before treating or stenting the main vessel.
Guiding Catheter Selection in the Management of Bifurcation Lesions
[edit | edit source]Frequently large guide catheters (7F and above) are used to accommodate the multiple wires and the multiple simultaneous devices that are used in the treatment of a bifurcation lesion. Larger guiding catheters accommodate the use of multiple balloons for the kissing technique, and allow for the use of both balloons and stents as needed. It should be noted that 6F guiding catheters can be used for kissing balloon inflations.
Guidewire Selection And Technique in the Management of Bifurcation Lesions
[edit | edit source]Oftentimes operators will use two different wires so that it is clear which wire is which on the fluoroscopy screen. In general the more difficult lesion is crossed first. If the wire cannot be passed, then a hydrophilic wire can be used and if necessary a balloon can be inflated at low pressure to deflect the wire into the side branch.
Some operators advocate placement and retention of the wire in the side branch and stenting over this wire. These operators have hypothesized that this improves the geometry of the side branch and makes it more accessible to wiring through the stent strut for a kissing balloon inflation at the end of the case. This technique does. The risk of wire entrapment and wire fracture and should be used with extreme caution. This is not a technique utilized by the editor C. Michael Gibson M.S., M.D..
Balloon on a Wire Techniques
[edit | edit source]If balloon-on-wire devices are used (Ace balloon, Svelte balloon), the guidewire position may be lost if upsizing or exchange becomes necessary.
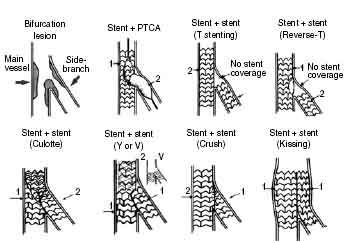
Treatment Choices
[edit | edit source]PTCA
[edit | edit source]- Double guidewire followed by sequential PTCA
- Kissing balloon dilation (i.e. simultaneous balloon inflations)
Debulking
[edit | edit source]Stenting
[edit | edit source]Single Stent Technique
[edit | edit source]The single stent technique (also known as “provisional stenting”) is most commonly adopted. It involves stenting the main branch and then rescuing the side branch with either balloon angioplasty or stenting if necessary.
T-stent Technique
[edit | edit source]This involves stenting the side branch first to cover the ostium and then stenting the main branch. This approach can be difficult if the angle of the side branch is not 90 degrees and thus not allowing perfect alignment of the two stents. In this situation, there will either be a side branch stent strut protruding into the main vessel or incomplete coverage of the side branch ostium.
The modified T-stent approach (also known as “blocking balloon technique”) positions the side branch stent followed by a balloon placed in the main branch at the bifurcation. The main branch balloon is inflated first at low pressure to provide support against which the side branch stent can be aligned more accurately to cover the ostium without protruding. The side branch stent is then deployed followed by main branch stent deployment.
In the TAP (T and small protrusion) technique, the main branch is stented first, and then the side branch is reaccessed and redilated. The side branch stent is then placed with a small amount of strut protruding into the main branch. A main branch blocking balloon is then inflated simultaneously with the side branch stent.
Culotte/Trousers Technique
[edit | edit source]If necessary, both the main branch and side branch are first wired with pre-dilation to allow for better access. The parent vessel is then stented before accessing the side branch through the main branch stent struts and a second stent is deployed in the side branch. This second stent extends back into the main branch stent. The main branch is then reaccessed through the side branch stent strut and both stents are kissing ballooned to complete the procedure, which allows flaring of the small protrusion.
Kissing Stent Technique
[edit | edit source]This involves simultaneous stent deployment in parent and side branch vessels in parallel. This technique is useful in lesions that are difficult to cross, as wires do not need to be removed and lesions are not re-crossed. However, overdilation of the bifurcation carina may occur.
V-stenting and Y stenting
[edit | edit source]These are used when the stenosis is limited to the ostium of the side branch and the main vessel distal to the side branch. In V-stenting, stents are deployed simultaneously without either stent extending proximally enough to cover the other. Y stenting is when an additional stent is placed proximally in the main branch.
Crush Technique
[edit | edit source]Standard Crush Stenting
[edit | edit source]Two stents are deployed with the side branch stent prior to main branch stent. The standard crush technique involves positioning both stents simultaneously. Next, the side branch stent is deployed and both the stent-delivery system and side branch guidewire are withdrawn. The main branch stent is then deployed – crushing the portion of the side branch stent in the parent vessel into the vessel wall. In such a case, the main branch proximal to the bifurcation lesion is scaffolded with a triple layer of stent. The side branch may then be re-wired and kissing balloon inflations employed in both vessels. This technique ensures stent coverage in the origin of the side branch and protects functional side branches during main branch stenting, however re-wiring and re-inflating balloons makes the procedure more technically challenging and time-consuming.
Inverse Crush Stenting
[edit | edit source]The inverse crush approach is similar to the standard crush approach, however, there are a few differences. The side branch stent is positioned more proximally than the parent vessel stent, the parent vessel stent is deployed first, and then it is crushed by the side branch stent when it is deployed. The main branch is then re-wired and kissing balloon inflations are employed. Hoye and colleagues found that the crush technique was effective with low rates of target lesions revascularization and major adverse cardiac events (MACE), except in left main disease. They also noted that the final kissing balloon inflation significantly decreased side branch restenosis rates. Furthermore, a study using registry data in Canada found that patients treated with the crush technique had decreased rates of MACE and angina compared to those patients with just main branch treatment.
Technical Considerations
[edit | edit source]The operator needs to consider the following when formulating a strategy:
- How important is the side branch?
- Is there a significant lesion at the origin of the side branch?
- Will the angle of the side branch make directing a wire into it difficult and thereby complicate a side branch "rescue" procedure?
- Is there a significant plaque volume at the bifurcation?
- Is a two stent strategy necessary?
Advantages and Disadvantages of Each Choice
[edit | edit source]Single Stent Technique
[edit | edit source]Studies have shown there appears to be no overall benefit in adopting two stents strategies up front in comparison to the single stent strategy. Studies from the bare metal stent era demonstrated consistently that the single stent technique is superior to the two stents technique with less major adverse cardiac events (MACE). Studies from the drug eluting stent era continued to demonstrate higher overall restenosis rates in the two stents technique. Furthermore, two stents strategies appear to be associated with a higher risk for possible stent thrombosis in some of the studies performed. The NORDIC study[8] is a randomized control trial comparing the two strategies, and there was no significant difference in clinical outcomes including death, MI, TVR (target vessel revascularization) or composite MACE. The stent thrombosis rate was low in both groups (0.5% in the main branch only vs 0% in the main and side branches together) as were main branch restenosis rates (4.6% in the main branch only vs 5.1% in the main and side branches together). The side branch restenosis rates were acceptable (19.2% in the main branch only vs 11.5% in the main and side branches together). In view of the above results, the current concensus is to treat the majority of bifurcation lesions with the single stent technique with a view of converting to provisional side branch stenting in cases of unsatisfactory angiographic result.
DCA
[edit | edit source]Older studies have suggested less revascularization and restenosis rates and higher success rates with DCA compared to PTCA, but DCA has also been associated with more ischemic complications. However, this has become less an issue in the current stent era, especially with the adoption of drug eluting stents.
T Stenting
[edit | edit source]The stent in the side branch may not adequately cover the side branch ostium. Alternatively, it may protrude into the parent vessel with T stenting, especially if the side branch is not at 90 degrees to the main branch (i.e. the lesion is angulated).
Kissing Stents
[edit | edit source]In difficult-to-cross lesions, kissing stents may be preferred. The wires need not be removed and the side branch and parent lesions need not be recrossed. A disadvantage is the potential for overdilation of bifurcation carina.
Crush Technique
[edit | edit source]This approach ensures stent coverage in the origin of the side branch, but past studies suggest a possible increased stent thrombosis risk, a higher target lesion restenosis rate, and more major adverse cardiac events in the setting of left main stenting and among patients with multivessel disease.
Technical Considerations in Selecting a Strategy
[edit | edit source]- Decision whether to stent compromised side branch based on size of vessel, amount of jeopardized myocardium, and probability of repeat revascularization
- Side branch protection with a guidewire should be considered for any vessel >2.0 mm; in addition, if >50% stenosis of side branch is present, risk of side branch occlusion is >25% after dilation of parent vessel, and difficulty wiring branch vessel is anticipated
- If side branch is <1.5 mm or small amount of viable myocardium is supplied, side branch protection not usually necessary
- Kissing balloon inflations prevent plaque from prolapsing from one vessel to the other.
- Rotational atherectomy and PTCA may be performed for late ostial side branch stenosis, but there is increased risk of burr entrapment if stent struts have not been dilated with previous PTCA
- Sequential balloon inflation as opposed to kissing balloon dilation may lead to shifting plaque and side branch occlusion, but the same balloon may be used to dilate both vessels; parent vessel may remain underexpanded if balloon is sized according to caliber of vessel distal to bifurcation
- Final kissing balloon inflations prevent plaque from prolapsing from one vessel to the other and have been shown to result in better clinical outcomes, including lower restenosis rates, so generally this technique is recommended, especially when angioplasty or rotational atherectomy is used.
- T stenting and crush stenting are well suited for branches that originate at 90-degree angles from parent vessel, because coverage of branch ostium is improved in these cases
- T stenting and Y stenting are technically demanding; stent and retrieve technique may be preferred, depending on the operator's experience
- Isolated side branch ostial stent does not require protection of parent vessel
Detailed Discussion of Specific Techniques
[edit | edit source]The Crush Technique
[edit | edit source]Benefits
[edit | edit source]- Ensures complete coverage of the side branch ostium
- Immediate patency of parent and side branch
- Excellent immediate angiographic results
- Restenosis is usually very focal[8]
Detriments
[edit | edit source]- 3 Layers of stent in the main artery
- Without final kissing balloon inflation high restenosis rate
- May be difficult to re-cross side branch
- Minimal 7F guide system required
Technical Considerations
[edit | edit source]- Requires minimum of a 7F Guiding system
- Final Kissing balloon inflation required due to high restenosis rate
Technique
[edit | edit source]- Using a minimum 7F guide system, both the parent and side branch are wired. Predilation is recommended to ensure optimal balloon expansion and ease stent delivery. It is important to identify which wire is placed in which branch. We recommend either using 2 separate colored guidewires, keeping a marker on 1 wire, or covering a wire with a sterile towel while not in use.
- Any disease distal to the side branch should optimally be repaired before the final crush step.
- The 1st stent is advanced into the side branch. This may require further predilation. Generally, the stent is advanced beyond the delivery point.
 |
- The 2nd stent is advanced into the parent vessel. This is also placed beyond the intended deployment site.
- The side branch stent is pulled back into the parent vessel about 5 mm. This ensures adequate coverage of the ostium.
- The parent stent is pulled back crossing the side branch and the proximal end is placed proximal to the proximal end of the side branch stent.
 |
- The side branch stent is deployed.
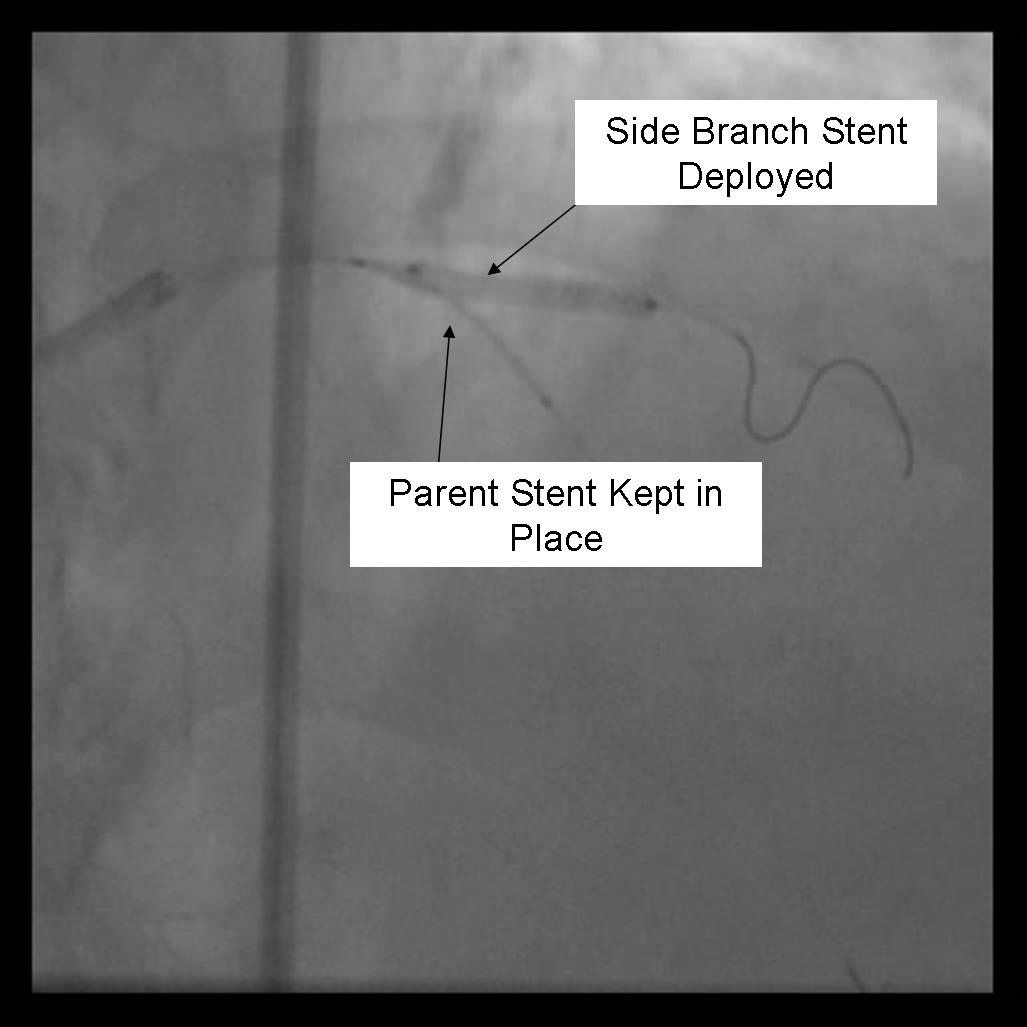 |
- Angiography should now be performed to ensure adequate side branch coverage and stent deployment. Distal edge dissections or disease should be addressed before parent stent deployment. Once complete, the side branch wire is removed.
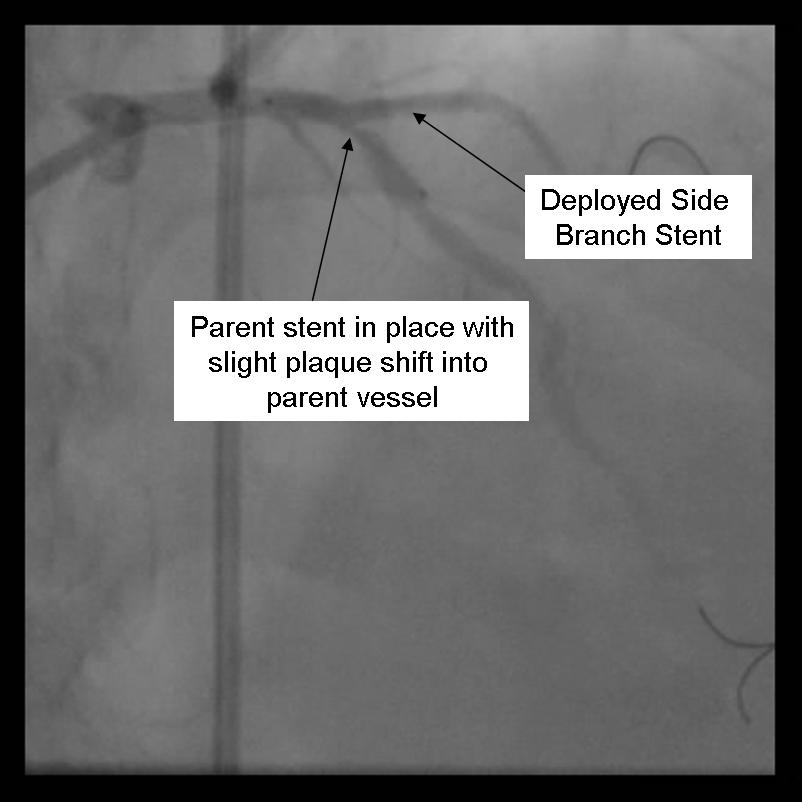 |
- The parent stent is deployed.
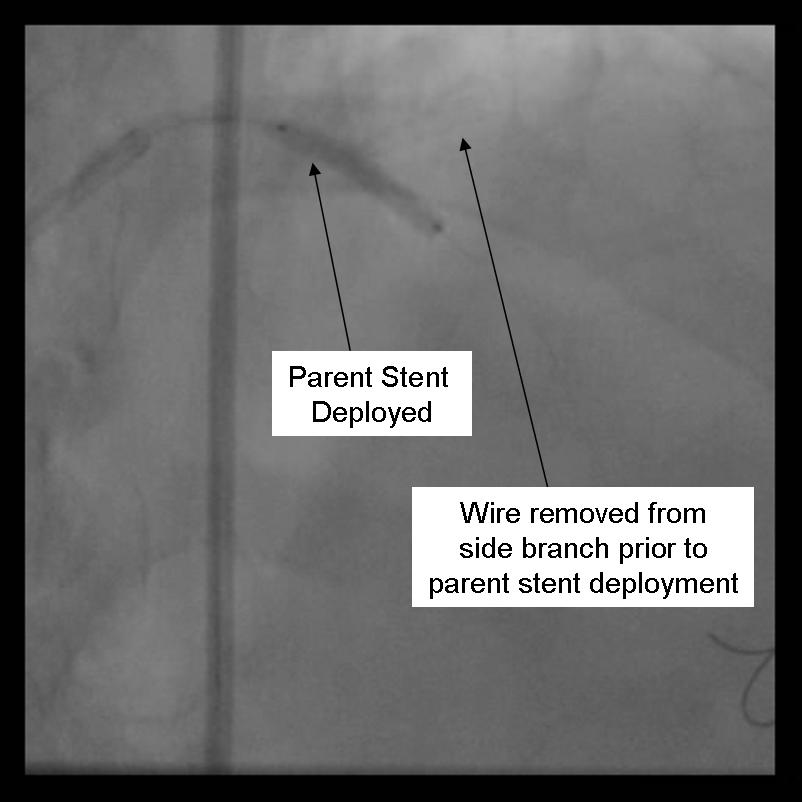 |
 |
- The side-branch is rewired. It is sometimes necessary to change wires to either a hydrophilic wire eg. Whisper, or Pilot 50 or 150, or ASAHI Fielder, one of increasing stiffness e.g. ASAHI Miracle Bros 3, or both e.g. Shinobi or Confienza.
- Kissing balloons are inflated at the bifurcation. It is sometimes necessary to dilate the side branch with a small balloon (1.5 mm middle marker balloon) to facilitate a larger balloon for the kissing technique.
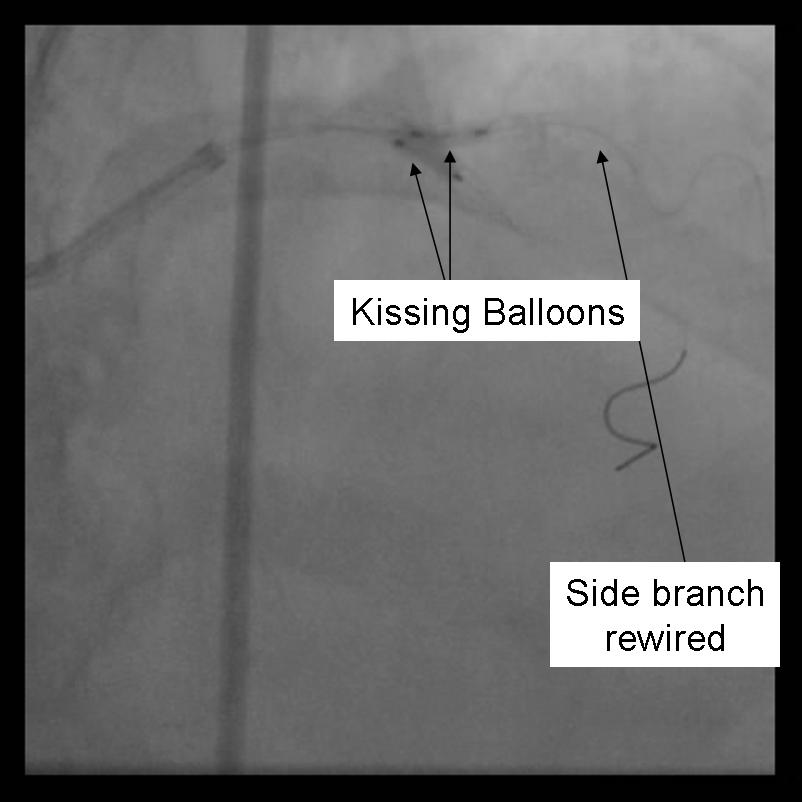 |
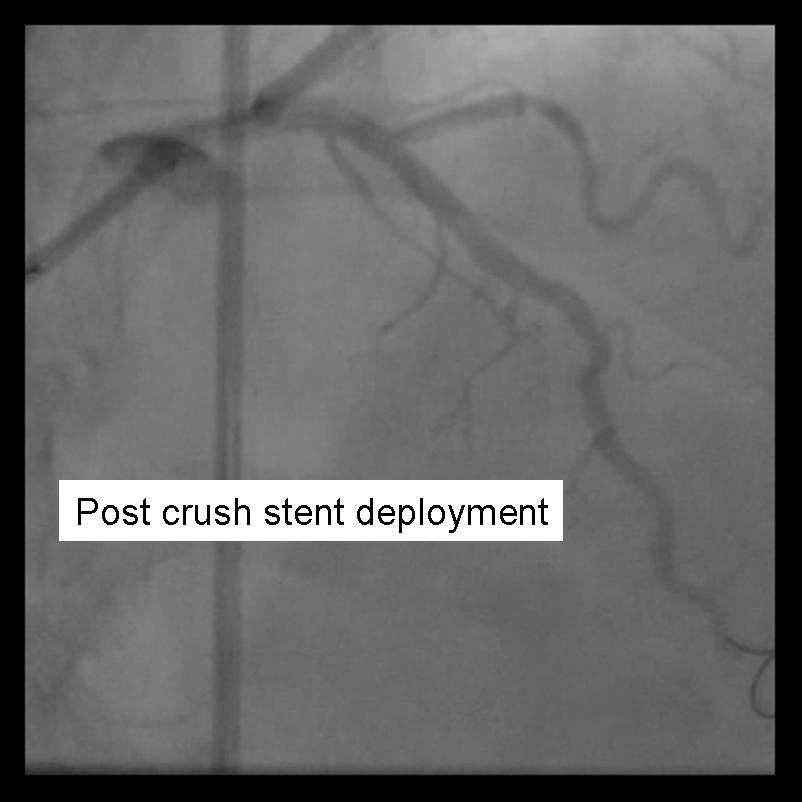
- Repeat angiography is performed. Wires are removed and final angiography completed.
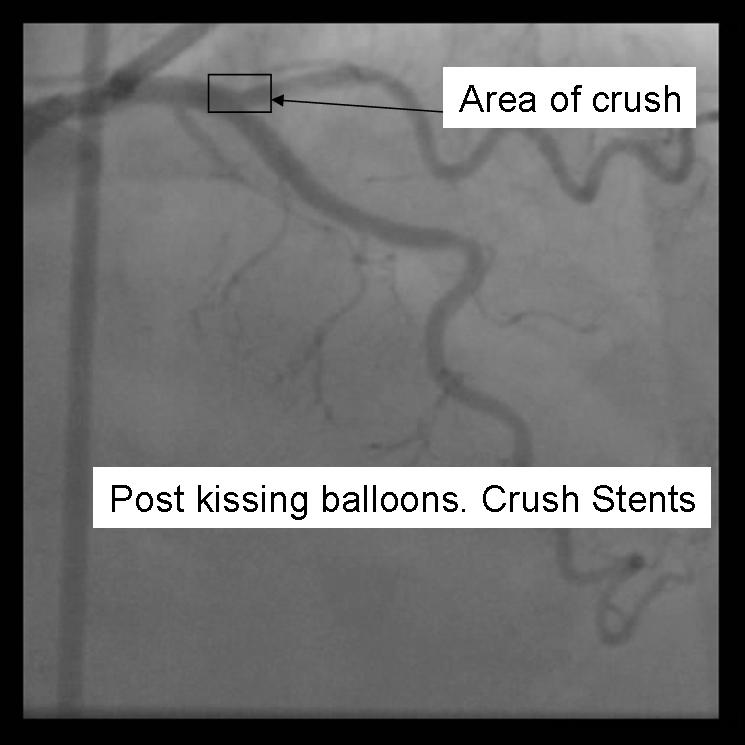 |
- Despite excellent initial angiographic results, side branch restenosis is still a significant problem with Crush stenting (Up to 25% without kissing ballon inflation). However, most of the restenosis is silent. The above example used Cypher drug eluting stents in the side branch and parent vessels. The patient returned with positive stress testing 13 months later. Repeat angiography demonstrated a short segment of significant in-stent restenosis.
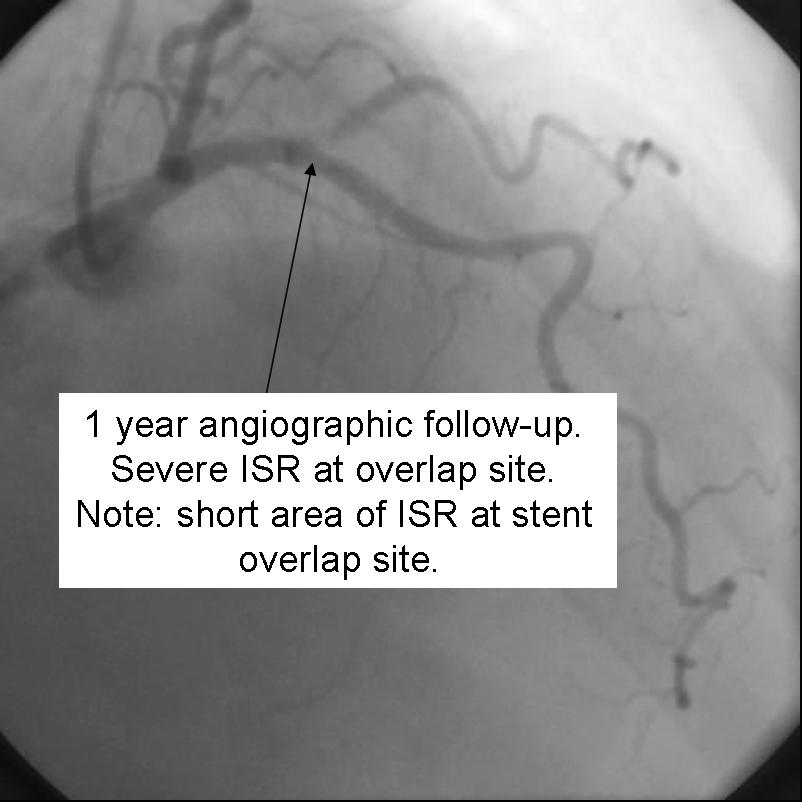 |
The Kissing Stents Technique
[edit | edit source]Benefits
[edit | edit source]- Ensures complete coverage of the side branch ostium
- Immediate patency of parent and side branch
- Access to both branches maintained through procedure
- No need for final kissing balloon
Detriments
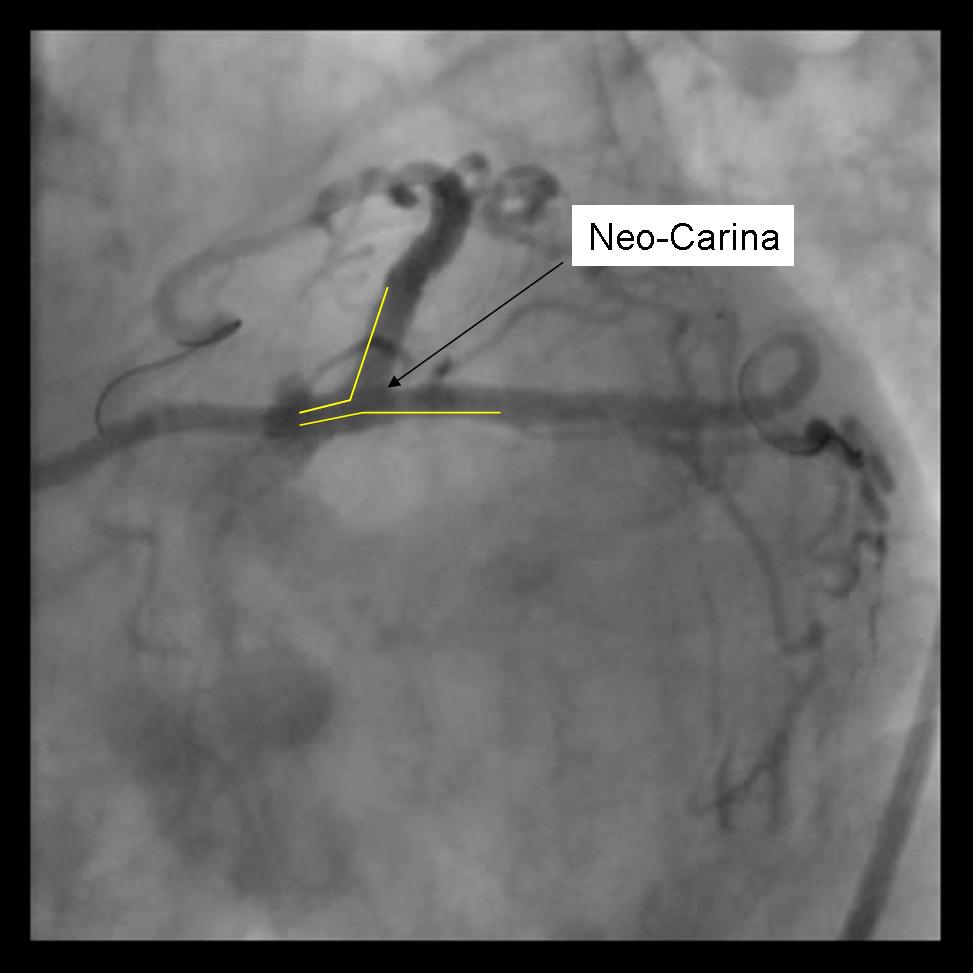
[edit | edit source]- Unendothelialized layer of stents forming neo-carina may predispose to stent thrombosis.
- 8F Guide system required
- May be difficult to re-access side branch or parent vessel
Technical Considerations
[edit | edit source]- Requires minimum of a 8F Guiding system
- Optimal angle of bifurcation less than 90 degrees.
Technique
[edit | edit source]- Using a minimum 8F guide system, both the parent and side branch are wired. Predilation is recommended to ensure optimal balloon expansion and ease stent delivery. It is important to identify which wire is placed in which branch. Either using 2 separate colored guidewires, keeping a marker on 1 wire, or covering a wire with a sterile towel while not in use are recommended.
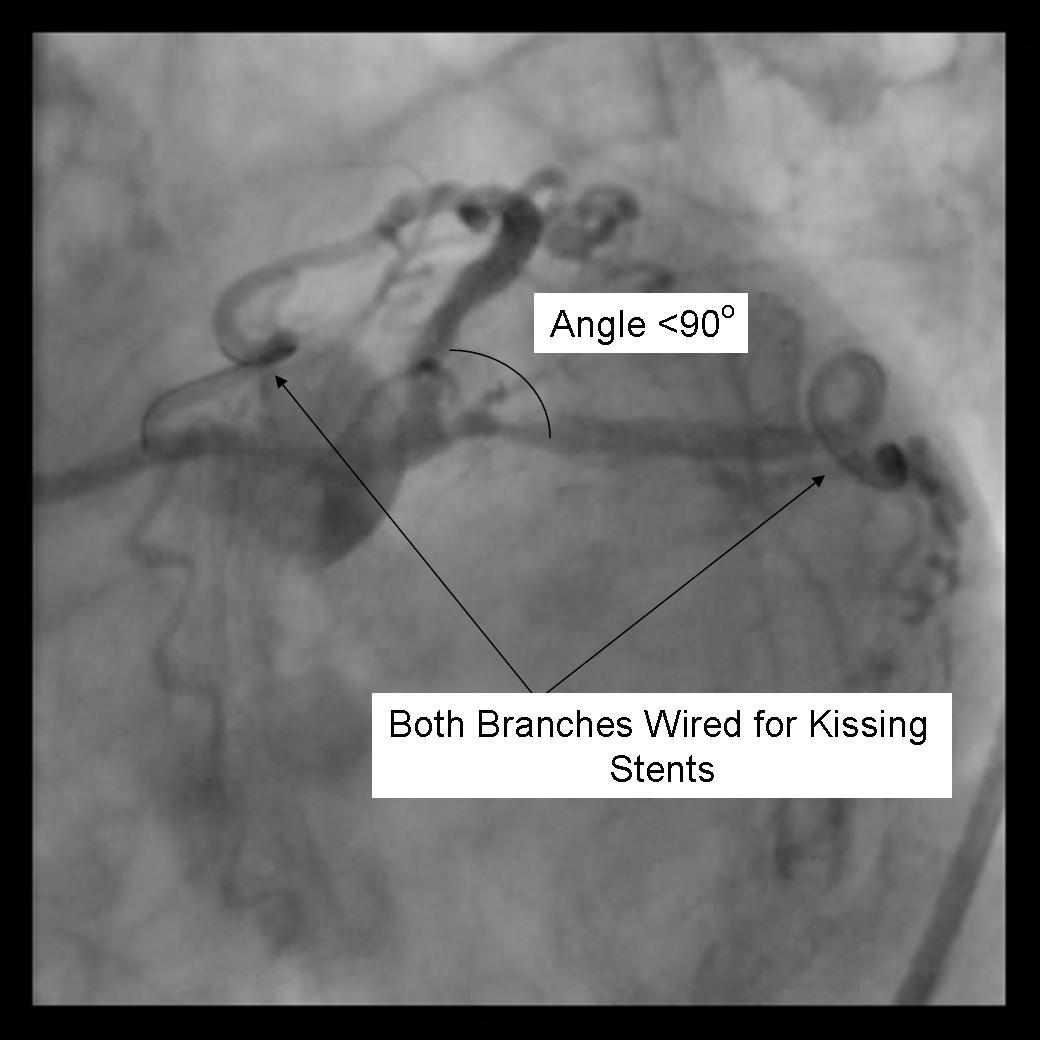 |
- Any disease distal to the side branch or parent vessel should optimally be repaired before the kissing stents are deployed.
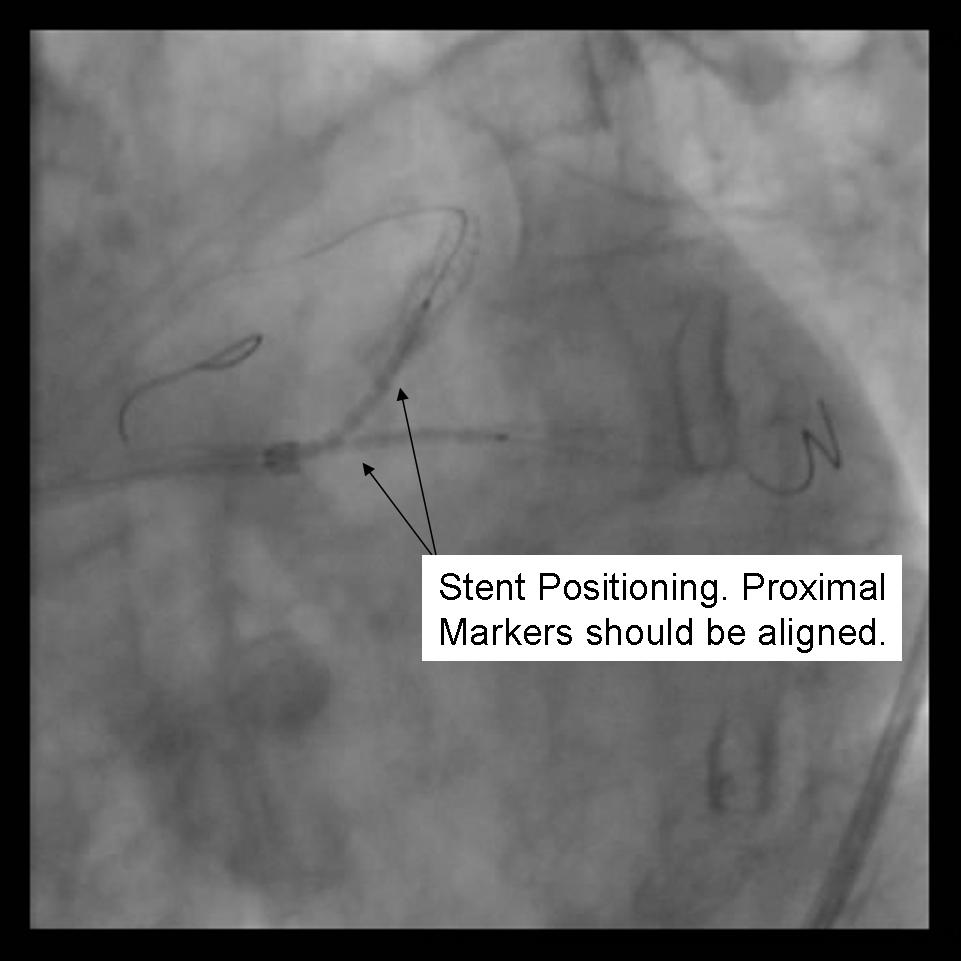
- Both stents are advanced into the side branch and parent vessel. This may require further predilation. Generally, the stents are advanced beyond the delivery point. Operator preference differs with respect to amount of overlap. Some operators form a long segment of overlap (>5mm), while others try to minimize overlap. It is absolutely essential that both vessels are covered during the balloon inflation. Optimal placement is ensured by advancing both stents distal to the intended deployment site then pulling them back into position. The proximal marker segments should be aligned, with positional confirmation in at least 2 separate views.
 |
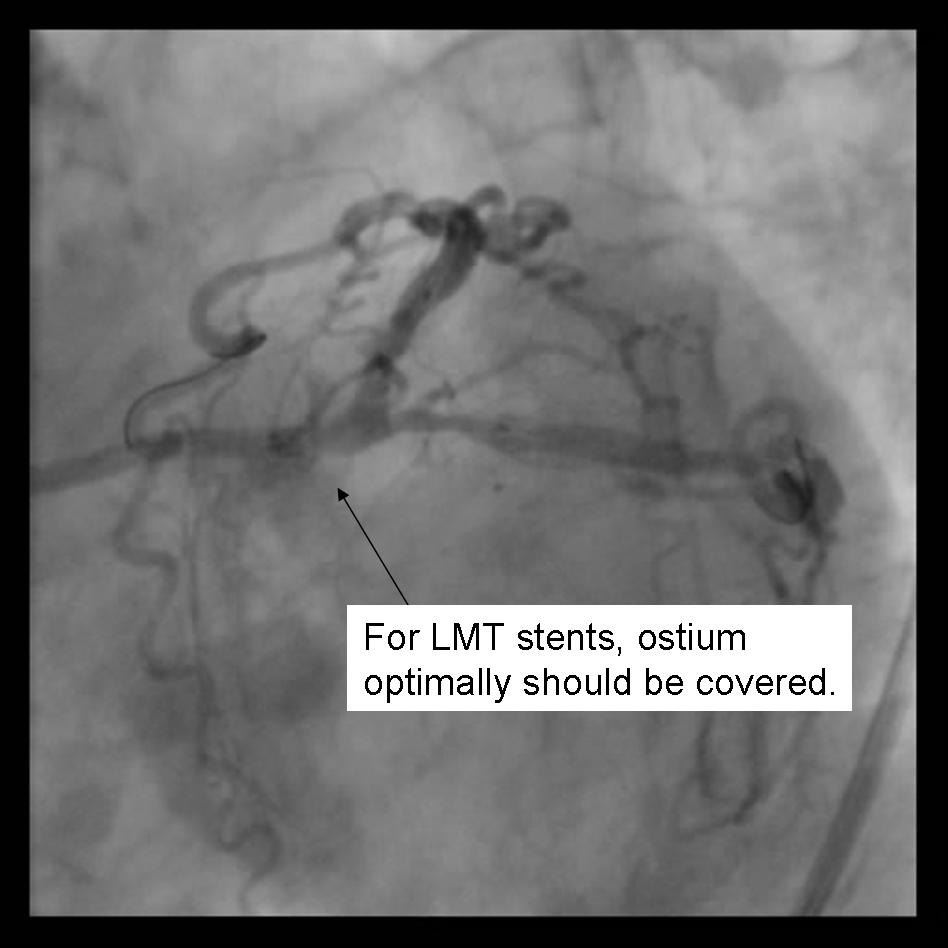 |
- Using 2 Insufflators, both stents are inflated to low pressure. Then alternating with side branch and parent branch inflated to optimal deployment size. There is some risk of over sizing the main branch with the overlapping stent segments. In the case where proximal disease may interfere with the 2 overlapping stents, a proximal stent may be deployed prior to inflating the 2 distal stents. (Pants-and-trousers technique)
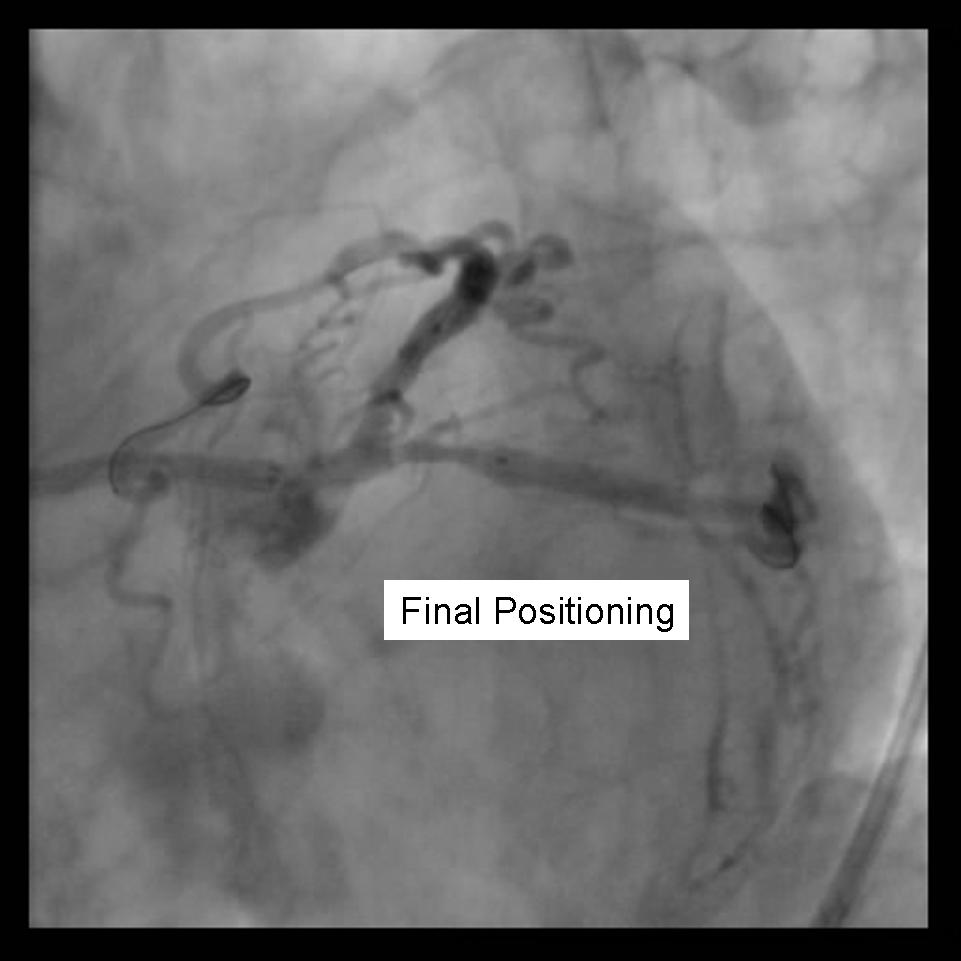 |
- Angiography should now be performed to ensure adequate side branch coverage and stent deployment. Distal edge dissections or disease should be addressed before wire removal from the vessels.
 |
- Variations of this method may be used to stent trifurcation disease. While an 8F catheter can be used depending on the stent system, optimally a 9 F system should be used in this situation.
The T-Stent Technique
[edit | edit source]Benefits
[edit | edit source]- May be used for provisional stenting of side branch if angiographic result is poor.
- Can Use 6F guide system
- Restenosis is usually very focal
Detriments
[edit | edit source]- Very difficult, if not impossible to completely cover side branch ostium
- Requires final kissing balloon inflation
- Modified T-stent technique requires 7F guide system
Technical Considerations
[edit | edit source]- May use a 6F Guiding system
- Final kissing balloon inflation required due to high restenosis rate
- Ideal angle of bifurcation is at or near 90 degrees.
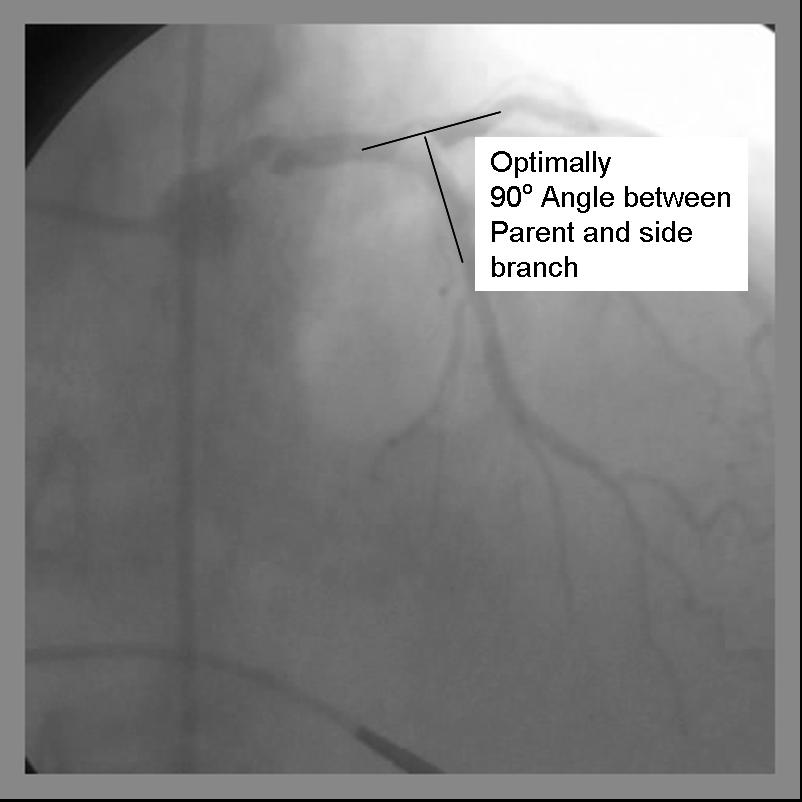 |
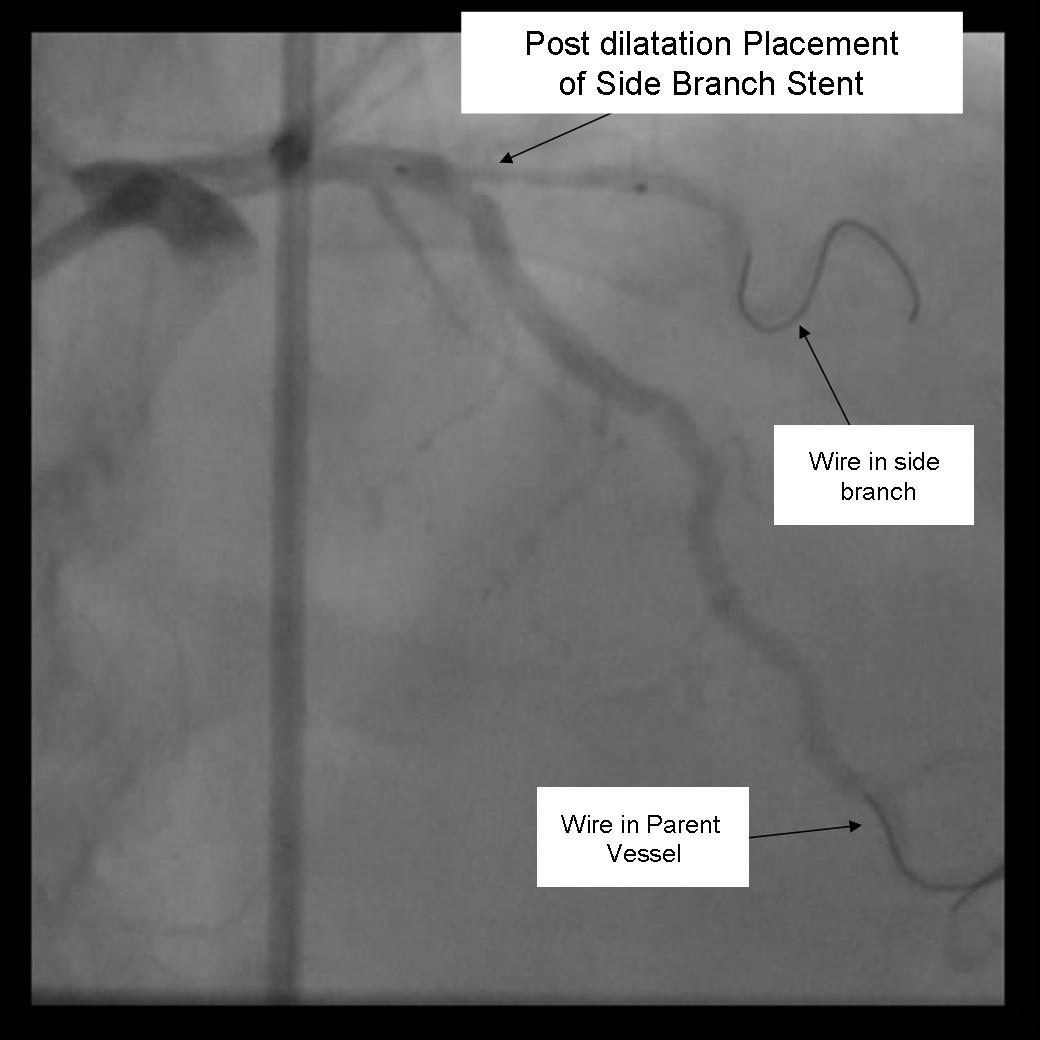
Technique (Modified T-Stenting)
[edit | edit source]- A 6F guide system may be used with standard T stenting in which the side branch stent is deployed, followed by delivery and deployment of the parent stent. The modified technique, which is preferred by this operator, requires a minimum of 7F guide system.
- Both the parent and side branch are wired. Predilation is recommended to ensure optimal balloon expansion and ease stent delivery. It is important to identify which wire is placed in which branch. We recommend either using 2 separate colored guidewires, keeping a marker on 1 wire, or covering a wire with a sterile towel while not in use.
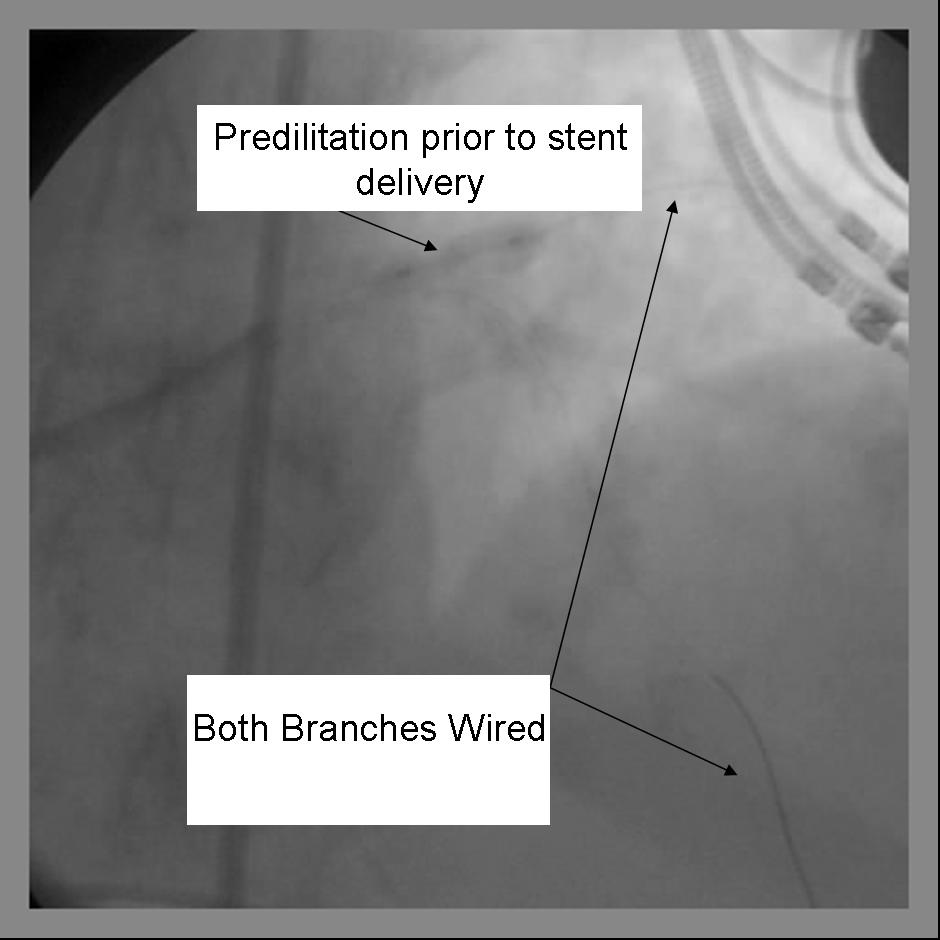 |
- Any disease in the distal side branch or parent vessel should optimally be repaired before the bifurcation stents are deployed.
- The 1st stent is advanced into the side branch. This may require further predilation. Generally, the stent is advanced beyond the delivery point.
- The 2nd stent is advanced into the parent vessel and placed across the side branch.
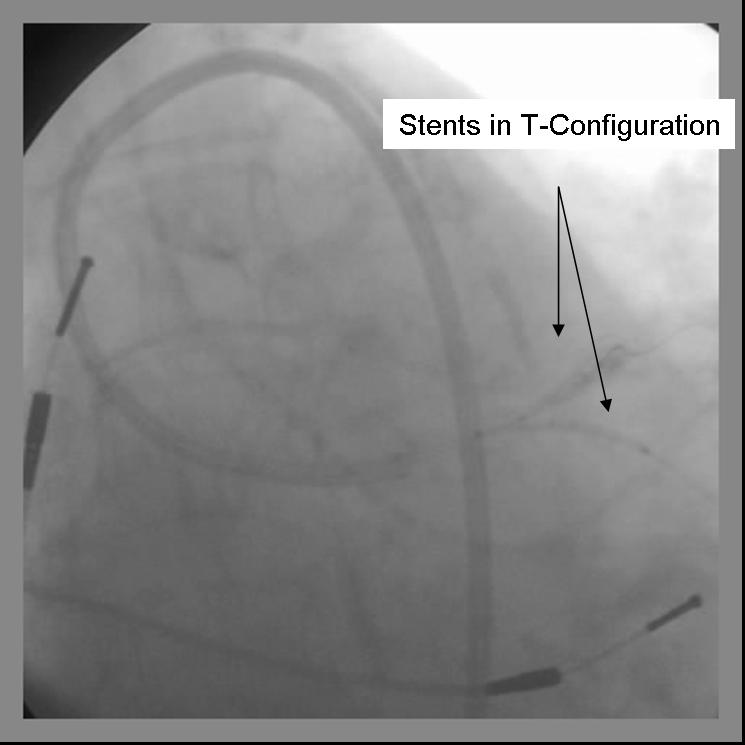 |
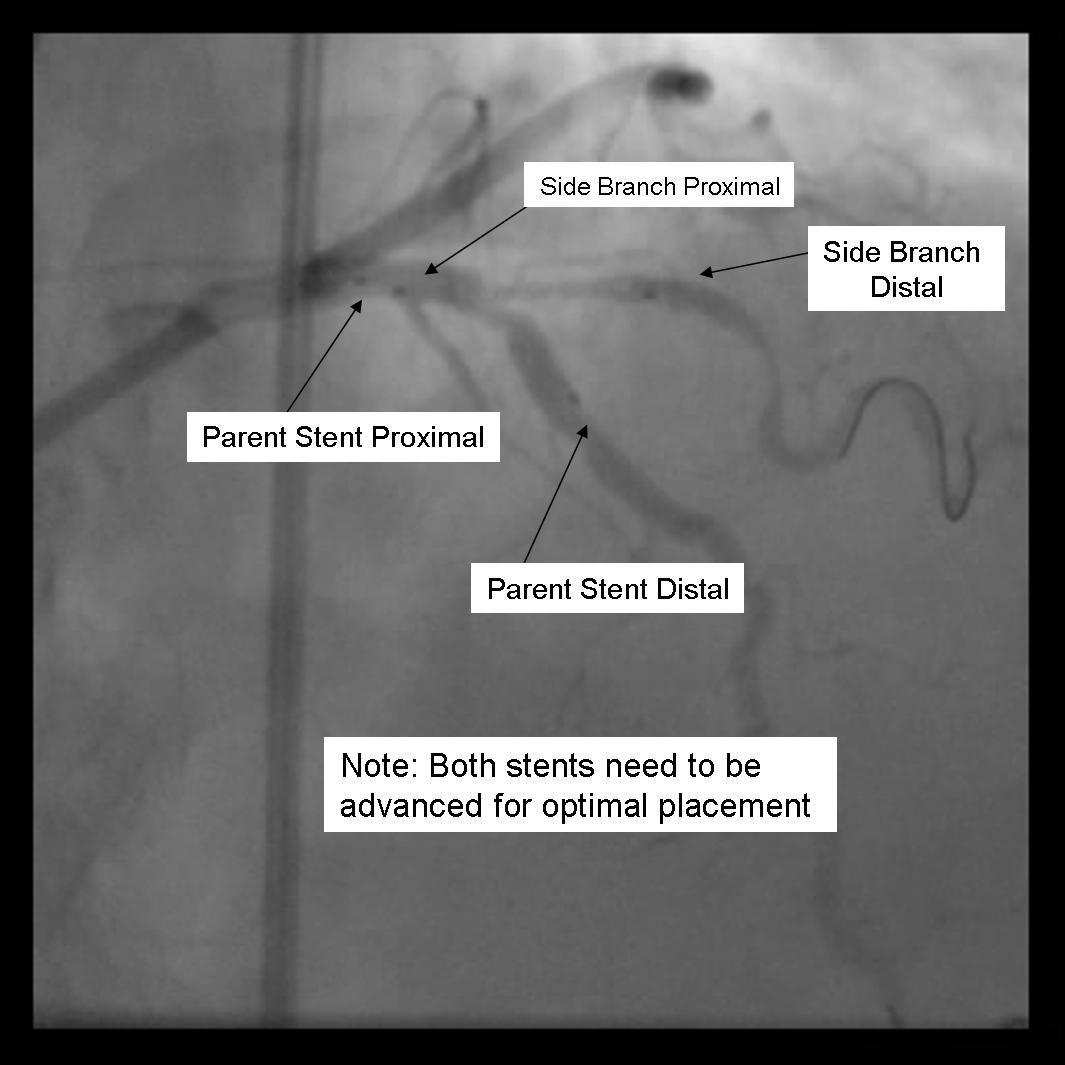
- The side branch stent is deployed. (Another option is to place a balloon in the parent branch and inflating to low atmospheres while pulling the side branch back. This may help ensure adequate coverage of the ostium.
- Side branch wire is removed once adequate angiographic result confirmed.
- The parent stent is deployed across the side branch.
 |
- Angiography should now be performed to ensure adequate side branch coverage and stent deployment. The parent stent is deployed.
- The side-branch is rewired. It is sometimes necessary to change wires to either a hydrophilic wire e.g. Whisper, or Pilot 50 or 150, or Fielder, one of increasing stiffness e.g. ASAHI Miracle Bros 3, or both e.g. Shinobi or Confienza.
- Kissing balloons are inflated at the bifurcation. It is sometimes necessary to dilate the side branch with a small balloon (1.5 mm middle marker balloon) to facilitate a larger balloon for the kiss.
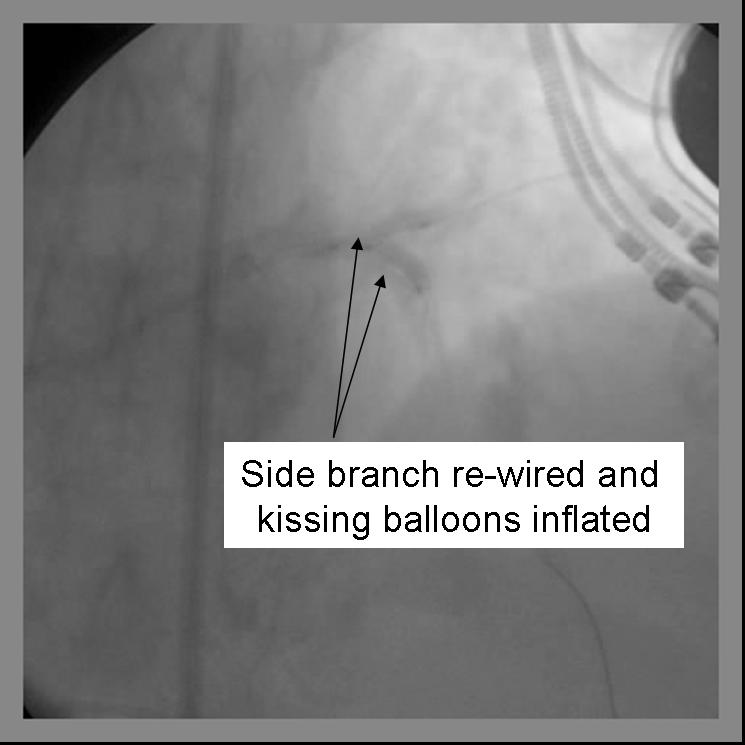 |
- Repeat angiography is performed. Wires are removed and final angiography completed.
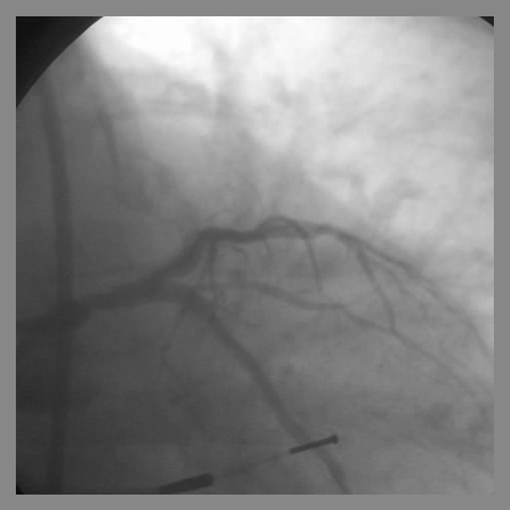 |
- Provisional T-Stenting involves the stenting the parent vessel and only the side branch if there is less than adequate side branch angiographic result. In this situation, the side branch may need to be serially dilated with up to a 2.5 mm balloon to ensure stent delivery. Final kissing balloons should be repeated.
- Sometimes the side branch stent impinges on the parent lumen making delivery of the parent stent impossible. If this happens, dilation of the parent lumen with a balloon allows for stent delivery. This may create a partial or small area of crushed stents at the bifurcation and should be followed with kissing balloons.
Complications
[edit | edit source]Successful dilation of the parent vessel can be achieved in >85% of bifurcation lesions, but the success rates in side branch are generally much lower. Freshly occluded side branches can be opened successfully in >75% of cases if there is no pre-existing disease, but they can be opened in <50% of cases if there is pre existing disease (>50% stenosis). Side branch occlusion occurs more commonly after rotational atherectomy if the preexisting side branch ostial stenosis was >50%. Almost 40% of side branches will occlude after debulking with DCA in parent vessel, but these usually can be rescued with PTCA. Retrograde propagation of a dissection from the side branch into the parent vessel, as well as incomplete dilation and occlusion of side branch, can occur.
If the side branch wire is left in place when deploying the stent in the main parent vessel, then there is a risk of jailing the wire. If main parent branch stent extends a significant length proximal from the side branch (therefore jailing a longer length of wire) then consideration should be given to removing the wire prior to stenting. Forceful removal of the jailed wire can result in distal embolization of the guidewire.
Angiography can confirm the prolapse of plaque into the side branch after dilation of the parent vessel or failure of dilatation of the parent vessel. It is important to note that side branch occlusion may be silent, especially in the presence of collaterals. Hemodynamic decompensation, arrhythmia, ST elevation, or chest pain may signify side branch occlusion.
Future Directions
[edit | edit source]New dedicated bifurcation stents have been developed to enhance treatment of these lesions. The AST petal DSX device[9] is a bare metal design that has “petals” in the main branch stent that allow guidewire access to the side branch and have outward-facing struts that protect the side branch ostium during main branch stenting. These petals cover the side branch ostium and allow balloon dilation of the side branch after main branch stenting. The SLK-ViewTM device[10] has a “trap door” that allows access to the side branch after main branch stenting. The AXXESS stent device[11] is a DES device with a main branch stent that includes a flared edge designed to sit in the carina of the bifurcation lesions and allow stent placement in the side branch if necessary while protecting the ostium from plaque shift and side branch closure. The BIGUARDTM[12] is a DES that allows exchange of guidewires and other devices within the main branch which may facilitate changing from a one-stent to a two-stent technique.
2011 ACCF/AHA/SCAI Guidelines for Percutaneous Coronary Intervention (DO NOT EDIT)[13]
[edit | edit source]Bifurcation Lesions (DO NOT EDIT)[13]
[edit | edit source]| Class I |
| "1. Provisional side-branch stenting should be the initial approach in patients with bifurcation lesions when the side branch is not large and has only mild or moderate focal disease at the ostium.[14][15][16][8] (Level of Evidence: A)" |
| Class IIa |
| "1. It is reasonable to use elective double stenting in patients with complex bifurcation morphology involving a large side branch where the risk of side-branch occlusion is high and the likelihood of successful side-branch reaccess is low.[17][18][19][20] (Level of Evidence: B)" |
The 16th expert consensus document of the European Bifurcation Club (2022)
[edit | edit source]The European Bifurcation Club has the following expert consensus recommendations: (DO NOT EDIT)[21]
[edit | edit source]- The Medina classification should be used in the description of bifurcation lesions.
- A provisional stenting approach is recommended for most bifurcation lesions, however for complex lesions where the side branch is large and supplies a significant coronary territory, a two-stent approach may be used.
- Proximal optimization technique should be used routinely for all bifurcation lesions.
- If a two-stent approach is used, lesion preparation should be performed in the main vessel and side branch first and final kissing ballooning and proximal optimization technique are mandatory.
- There should be a low threshold for use of intracoronary imaging (IVUS or OCT).
References
[edit | edit source]- ↑ Spokojny AM, Sanborn TM. The bifurcation lesion. In: Strategic Approaches in Coronary Intervention. Ellis SG, Holmes DR (Eds). Williams and Wilkins, MD, USA, 288 (1996)
- ↑ Safian RD. Bifurcation lesions. In: The Manual of Interventional Cardiology. Safian RD, Freed M (Eds). Physician's Press,MI, USA, 222 (2001)
- ↑ Lefèvre T, Louvard Y, Morice MC, Loubeyre C, Piéchaud JF, Dumas P (2001). "Stenting of bifurcation lesions: a rational approach". J Interv Cardiol. 14 (6): 573–85. PMID 12053378. Unknown parameter
|month=ignored (help) - ↑ Popma JJ, Leon MB, Topol EJ. Atlas of Interventional Cardiology. Philadelphia: Saunders, 1994
- ↑ Louvard Y, Medina A, Stankovic G, Definition and classification of bifurcation lesions and treatments. EuroIntervention Supplement (2010) Vol. 6 (Supplement J) J31-J35. http://www.pcronline.com/eurointervention/J_issue/J31/
- ↑ Medina A, Suárez de Lezo J, Pan M (2006). "[A new classification of coronary bifurcation lesions]". Rev Esp Cardiol (in Spanish; Castilian). 59 (2): 183. PMID 16540043. Unknown parameter
|month=ignored (help) - ↑ Zlotnick DM, Ramanath VS, Brown JR, Kaplan AV (2012). "Classification and treatment of coronary artery bifurcation lesions: putting the Medina classification to the test". Cardiovasc Revasc Med. 13 (4): 228–33. doi:10.1016/j.carrev.2012.04.002. PMID 22698367.
- ↑ 8.0 8.1 8.2 Steigen TK, Maeng M, Wiseth R; et al. (2006). "Randomized study on simple versus complex stenting of coronary artery bifurcation lesions: the Nordic bifurcation study". Circulation. 114 (18): 1955–61. doi:10.1161/CIRCULATIONAHA.106.664920. PMID 17060387. Unknown parameter
|month=ignored (help) - ↑ Ormiston J, Webster M, El-Jack S, McNab D, Plaumann SS (2007). "The AST petal dedicated bifurcation stent: first-in-human experience". Catheter Cardiovasc Interv. 70 (3): 335–40. doi:10.1002/ccd.21206. PMID 17722036. Unknown parameter
|month=ignored (help) - ↑ Ikeno F, Kim YH, Luna J; et al. (2006). "Acute and long-term outcomes of the novel side access (SLK-View) stent for bifurcation coronary lesions: a multicenter nonrandomized feasibility study". Catheter Cardiovasc Interv. 67 (2): 198–206. doi:10.1002/ccd.20556. PMID 16404749. Unknown parameter
|month=ignored (help) - ↑ Hasegawa T, Ako J, Koo BK; et al. (2009). "Analysis of left main coronary artery bifurcation lesions treated with biolimus-eluting DEVAX AXXESS plus nitinol self-expanding stent: intravascular ultrasound results of the AXXENT trial". Catheter Cardiovasc Interv. 73 (1): 34–41. doi:10.1002/ccd.21765. PMID 19089934. Unknown parameter
|month=ignored (help) - ↑ Chen SL, Zhang JJ, Ye F (2009). "Novel side branch ostial stent (BIGUARD): first-in-man study". Chin. Med. J. 122 (18): 2092–6. PMID 19781290. Unknown parameter
|month=ignored (help) - ↑ 13.0 13.1 Levine GN, Bates ER, Blankenship JC, Bailey SR, Bittl JA, Cercek B, Chambers CE, Ellis SG, Guyton RA, Hollenberg SM, Khot UN, Lange RA, Mauri L, Mehran R, Moussa ID, Mukherjee D, Nallamothu BK, Ting HH (2011). "2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention: Executive Summary A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions" (PDF). Journal of the American College of Cardiology. 58 (24): 2550–83. doi:10.1016/j.jacc.2011.08.006. PMID 22070837. Retrieved 2011-12-08. Text "PDF" ignored (help); Unknown parameter
|month=ignored (help) - ↑ Colombo A, Bramucci E, Saccà S, Violini R, Lettieri C, Zanini R, Sheiban I, Paloscia L, Grube E, Schofer J, Bolognese L, Orlandi M, Niccoli G, Latib A, Airoldi F (2009). "Randomized study of the crush technique versus provisional side-branch stenting in true coronary bifurcations: the CACTUS (Coronary Bifurcations: Application of the Crushing Technique Using Sirolimus-Eluting Stents) Study". Circulation. 119 (1): 71–8. doi:10.1161/CIRCULATIONAHA.108.808402. PMID 19103990. Retrieved 2011-12-15. Unknown parameter
|month=ignored (help) - ↑ Ferenc M, Gick M, Kienzle RP, Bestehorn HP, Werner KD, Comberg T, Kuebler P, Büttner HJ, Neumann FJ (2008). "Randomized trial on routine vs. provisional T-stenting in the treatment of de novo coronary bifurcation lesions". European Heart Journal. 29 (23): 2859–67. doi:10.1093/eurheartj/ehn455. PMC 2638653. PMID 18845665. Retrieved 2011-12-15. Unknown parameter
|month=ignored (help) - ↑ Hildick-Smith D, de Belder AJ, Cooter N, Curzen NP, Clayton TC, Oldroyd KG, Bennett L, Holmberg S, Cotton JM, Glennon PE, Thomas MR, Maccarthy PA, Baumbach A, Mulvihill NT, Henderson RA, Redwood SR, Starkey IR, Stables RH (2010). "Randomized trial of simple versus complex drug-eluting stenting for bifurcation lesions: the British Bifurcation Coronary Study: old, new, and evolving strategies". Circulation. 121 (10): 1235–43. doi:10.1161/CIRCULATIONAHA.109.888297. PMID 20194880. Retrieved 2011-12-15. Unknown parameter
|month=ignored (help) - ↑ Chen SL, Santoso T, Zhang JJ, Ye F, Xu YW, Fu Q, Kan J, Paiboon C, Zhou Y, Ding SQ, Kwan TW (2011). "A Randomized Clinical Study Comparing Double Kissing Crush With Provisional Stenting for Treatment of Coronary Bifurcation Lesions Results From the DKCRUSH-II (Double Kissing Crush versus Provisional Stenting Technique for Treatment of Coronary Bifurcation Lesions) Trial". Journal of the American College of Cardiology. 57 (8): 914–20. doi:10.1016/j.jacc.2010.10.023. PMID 21329837. Retrieved 2011-12-15. Unknown parameter
|month=ignored (help) - ↑ Moussa ID (2011). "Coronary artery bifurcation interventions: the disconnect between randomized clinical trials and patient centered decision-making". Catheterization and Cardiovascular Interventions :Official Journal of the Society for Cardiac Angiography & Interventions. 77 (4): 537–45. doi:10.1002/ccd.22865. PMID 21351230. Retrieved 2011-12-15. Unknown parameter
|month=ignored (help) - ↑ Aliabadi D, Tilli FV, Bowers TR, Benzuly KH, Safian RD, Goldstein JA, Grines CL, O'Neill WW (1997). "Incidence and angiographic predictors of side branch occlusion following high-pressure intracoronary stenting". The American Journal of Cardiology. 80 (8): 994–7. PMID 9352966. Retrieved 2011-12-15. Unknown parameter
|month=ignored (help) - ↑ Galassi AR, Tomasello SD, Capodanno D, Barrano G, Ussia GP, Tamburino C (2009). "Mini-crush versus T-provisional techniques in bifurcation lesions: clinical and angiographic long-term outcome after implantation of drug-eluting stents". JACC. Cardiovascular Interventions. 2 (3): 185–94. doi:10.1016/j.jcin.2008.12.005. PMID 19463424. Retrieved 2011-12-15. Unknown parameter
|month=ignored (help) - ↑ Albiero R, Burzotta F, Lassen JF, Lefèvre T, Banning AP, Chatzizisis YS; et al. (2022). "Treatment of coronary bifurcation lesions, part I: implanting the first stent in the provisional pathway. The 16th expert consensus document of the European Bifurcation Club". EuroIntervention. 18 (5): e362–e376. doi:10.4244/EIJ-D-22-00165. PMID 35570748 Check
|pmid=value (help).
 KSF
KSF