Immune checkpoint
 From Wikidoc - Reading time: 4 min
From Wikidoc - Reading time: 4 min
Cancers considered "hot tumors", such as melanoma, contain antitumor T cells and may respond to checkpoint inhibitor antibodies[1].
Immune checkpoint inhibitors
[edit | edit source]Monoclonal antibodies are a type of cancer immunotherapy that have been developed to target immune checkpoints. These checkpoints down-regulate programmed cell death 1 or apoptosis. Cancer immunotherapy to block these checkpoints may encourage apoptosis of cancer cells.
Drug toxicities include[2][3][4]:
- Gastrointestinal
- Dermatologic
- Endocrine
- Exacerbation of pre-existing autoimmune disease[5].
PD-1 inhibitors
[edit | edit source]These antibodies can activate the immune system to attack cancers. These antibodies target the Programmed Cell Death 1 Receptor (PD-1 Receptor or CD279). Programmed Cell Death Type I is also known as apoptosis. The PD-1 Receptor is "an inhibitory T-lymphocyte receptor that has specificity for CD274 antigen and Programmed Cell Death 1 Ligand 2 Protein."[6][7]
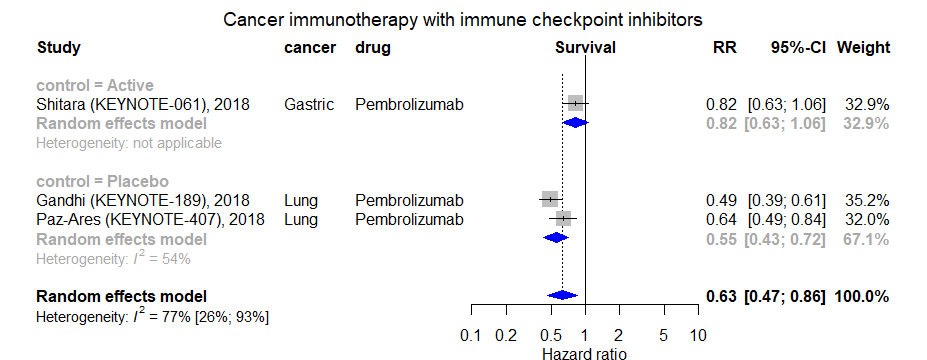
Pembrolizumab is a 'humanized monoclonal IgG4 kappa isotype antibody against PD-1'.[8] Pembrolizumab was approved by the FDA for “patients with unresectable or metastatic, microsatellite-instability–high (MSI-H) or mismatch-repair–deficient (dMMR) solid tumors, regardless of tumor site or histology”. [9] Pembrolizumab creates more objective tumor response among tumors in which at least 50% of its cells express PD-L1[8].
Nivolumab may case drug toxicity in about 40% of patients - rash and diarrhea are the most common effects[10].
Examples:
- Pembrolizumab (Keytruda)
- Nivolumab (Opdivo)
PD-L1 inhibitors
[edit | edit source]These antibodies can activate the immune system to attack cancers. These antibodies target PD-L1, or cluster of differentiation 274 (CD274) which is the transmembrane protein ligand to PD-1.
- Atezolizumab (Tecentriq)
- Avelumab (Bavencio)
- Durvalumab (Imfinzi)
CTLA-4 blockade
[edit | edit source]The CTLA-4 Antigen is "an inhibitory T cell receptor that is closely related to CD28 antigen. It has specificity for CD80 antigen and CD86 antigen and acts as a negative regulator of peripheral T cell function. CTLA-4 antigen is believed to play role in inducing peripheral tolerance."[11]
Ipilimumab may cause autoimmune pituitary disease[12] and exacerbate autoimmune disease in recipients with pre-existing autoimmune disease[13]. The U.S. FDA has issued guidance on managing side effects.[14][15]
Examples:
- Ipilimumab (Yervoy)
Raf protein kinase inhibitors
[edit | edit source]Raf inhibitors are "a family of closely-related serine-threonine kinases that were originally identified as the cellular homologs of the retrovirus-derived V-RAF kinases. They are MAP kinase kinase kinases that play important roles in signal transduction."[16]
Examples:
Vaccines
[edit | edit source]Vaccines may provide immunotherapy of cancers[1].
See also
[edit | edit source]References
[edit | edit source]- ↑ 1.0 1.1 Schlom J, Gulley JL. Vaccines as an Integral Component of Cancer Immunotherapy. JAMA. Published online November 08, 2018. doi:10.1001/jama.2018.9511
- ↑ Bourke JM, O'Sullivan M, Khattak MA (2016). "Management of adverse events related to new cancer immunotherapy (immune checkpoint inhibitors)". Med J Aust. 205 (9): 418–424. PMID 27809739.
- ↑ Barroso-Sousa R, Barry WT, Garrido-Castro AC, Hodi FS, Min L, Krop IE; et al. (2017). "Incidence of Endocrine Dysfunction Following the Use of Different Immune Checkpoint Inhibitor Regimens: A Systematic Review and Meta-analysis". JAMA Oncol. doi:10.1001/jamaoncol.2017.3064. PMID 28973656.
- ↑ Postow et al. http://www.nejm.org/doi/full/10.1056/NEJMra1703481 NEJM 2018
- ↑ Abdel-Wahab N, Shah M, Lopez-Olivo MA, Suarez-Almazor ME (2018). "Use of Immune Checkpoint Inhibitors in the Treatment of Patients With Cancer and Preexisting Autoimmune Disease: A Systematic Review". Ann Intern Med. doi:10.7326/M17-2073. PMID 29297009.
- ↑ Anonymous (2026), Programmed Cell Death 1 Receptor (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ Boussiotis VA (2016). "Molecular and Biochemical Aspects of the PD-1 Checkpoint Pathway". N Engl J Med. 375 (18): 1767–1778. doi:10.1056/NEJMra1514296. PMC 5575761. PMID 27806234.
- ↑ 8.0 8.1 Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP; et al. (2015). "Pembrolizumab for the treatment of non-small-cell lung cancer". N Engl J Med. 372 (21): 2018–28. doi:10.1056/NEJMoa1501824. PMID 25891174.
- ↑ Lemery S, Keegan P, Pazdur R (2017). "First FDA Approval Agnostic of Cancer Site - When a Biomarker Defines the Indication". N Engl J Med. 377 (15): 1409–1412. doi:10.1056/NEJMp1709968. PMID 29020592.
- ↑ Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF; et al. (2012). "Safety, activity, and immune correlates of anti-PD-1 antibody in cancer". N Engl J Med. 366 (26): 2443–54. doi:10.1056/NEJMoa1200690. PMC 3544539. PMID 22658127.
- ↑ Anonymous (2026), CTLA-4 Antigen (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ Corsello SM, Barnabei A, Marchetti P, De Vecchis L, Salvatori R, Torino F (2013). "Endocrine side effects induced by immune checkpoint inhibitors". J Clin Endocrinol Metab. 98 (4): 1361–75. doi:10.1210/jc.2012-4075. PMID 23471977.
- ↑ Johnson DB, Sullivan RJ, Ott PA, Carlino MS, Khushalani NI, Ye F; et al. (2016). "Ipilimumab Therapy in Patients With Advanced Melanoma and Preexisting Autoimmune Disorders". JAMA Oncol. 2 (2): 234–40. doi:10.1001/jamaoncol.2015.4368. PMID 26633184.
- ↑ https://www.fda.gov/downloads/Drugs/DrugSafety/PostMarketDrugsafetyInformationforPatientsandProviders/UCM249435.pdf
- ↑ https://dailymed.nlm.nih.gov/dailymed/search.cfm?labeltype=all&query=ipilimumab
- ↑ Template:Mesh
 KSF
KSF