Ketone
 From Wikidoc - Reading time: 6 min
From Wikidoc - Reading time: 6 min
|
WikiDoc Resources for Ketone |
|
Articles |
|---|
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Ketone at Clinical Trials.gov Clinical Trials on Ketone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Ketone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Ketone Risk calculators and risk factors for Ketone
|
|
Healthcare Provider Resources |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
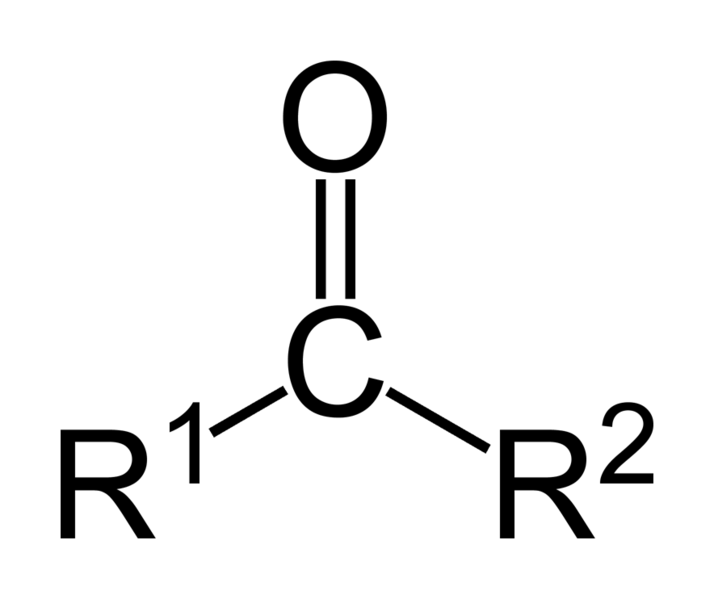
[edit | edit source]A ketone (pronounced as key tone) is either the functional group characterized by a carbonyl group (O=C) linked to two other carbon atoms or a chemical compound that contains this functional group. A ketone can be generally represented by the formula:
A carbonyl carbon bonded to two carbon atoms distinguishes ketones from carboxylic acids, aldehydes, esters, amides, and other oxygen-containing compounds. The double-bond of the carbonyl group distinguishes ketones from alcohols and ethers. The simplest ketone is acetone (systematically named 2-propanone[1]).
The carbon atom adjacent to a carbonyl group is called the α-carbon. Hydrogens attached to this carbon are called α-hydrogens. In the presence of an acid catalyst the ketone is subjected to so-called keto-enol tautomerism. The reaction with a strong base gives the corresponding enolate. A diketone is a compound containing two ketone groups.
Nomenclature
[edit | edit source]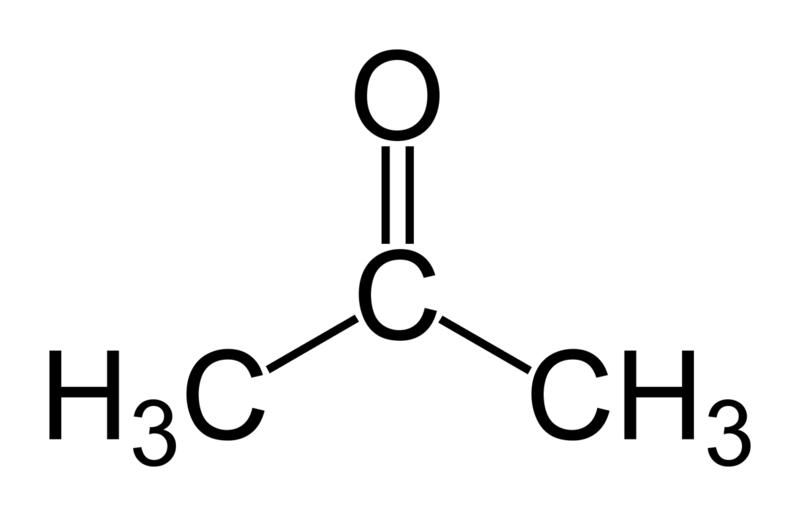
In general, ketones are named using IUPAC nomenclature by changing the suffix -e of the parent alkane to -one. For common ketones, some traditional names such as acetone and benzophenone predominate, and these are considered retained IUPAC names [2], although some introductory chemistry texts use names such as propanone.
Oxo is the formal IUPAC nomenclature for a ketone functional group. However, other prefixes are also used by various books and journals. For some common chemicals (mainly in biochemistry), keto or oxy is the term used to describe the ketone (also known as alkanone) functional group. Oxo also refers to a single oxygen atom coordinated to a transition metal (a metal oxo).
Physical properties
[edit | edit source]A carbonyl group is polar. This makes ketones polar compounds. The carbonyl groups interact with water by hydrogen bonding, and ketones are soluble in water. It is a hydrogen-bond acceptor, but not a hydrogen-bond donator, and cannot hydrogen-bond to itself. This makes ketones more volatile than alcohols and carboxylic acids of similar molecular weight.
Acidity
[edit | edit source]The α-hydrogen of a ketone is far more acidic (pKa ≈ 20) than the hydrogen of a regular alkane (pKa ≈ 50). This is due to resonance stabilization of the enolate ion that is formed through dissociation. The relative acidity of the α-hydrogen is important in the enolization reactions of ketones and other carbonyl compounds.
Spectroscopic properties
[edit | edit source]Spectroscopy is an important means for identifying ketones. Ketones and aldehydes will display a significant peak in infrared spectroscopy, at around 1700 cm−1 (slightly higher or lower, depending on the chemical environment)
Synthesis
[edit | edit source]Several methods exist for the preparation of ketones in the laboratory:
- Ketones can be created by oxidation of secondary alcohols. The process requires a strong oxidizing agent such as potassium permanganate, potassium dichromate or other agents containing Cr(VI). The alcohol is oxidized by heating under reflux in acidified solution. For example propan-2-ol is oxidised to acetone:
- H3C-CH(OH)-CH3 → H3C-CO-CH3
- Two atoms of hydrogen are removed, leaving a single oxygen atom double-bonded to a carbon atom.
- Ketones are also prepared by Gem halide hydrolysis.
- Alkynes can be turned into enols through hydration in the presence of an acid and HgS04, and subsequent enol-keto tautomerization gives a ketone. This always produces a ketone, even with a terminal alkyne, and Sia2BH is needed to get an aldehyde from an alkyne
- Aromatic ketones can be prepared in the Friedel-Crafts reaction and the Fries rearrangement.
- In the Kornblum–DeLaMare rearrangement ketones are prepared from peroxides and base
- In the Ruzicka cyclization, cyclic ketones are prepared from dicarboxylic acids.
- In the Nef reaction, ketones form by hydrolysis of salts of secondary nitro compounds
Reactions
[edit | edit source]Ketones engage in many organic reactions:
- Nucleophilic addition. The reaction of a ketone with a nucleophile gives a tetrahedral carbonyl addition compound.
- the reaction with the anion of a terminal alkyne gives a hydroxyalkyne
- the reaction with ammonia or a primary amine gives an imine + water
- the reaction with secondary amine gives an enamine + water
- the reaction with a Grignard reagent gives a magnesium alkoxide and after aqueous workup a tertiary alcohol
- the reaction with an organolithium reagent also gives a tertiary alcohol
- the reaction with an alcohol, an acid or base gives a hemiketal + water and further reaction with an alcohol gives the ketal + water. This is a carbonyl-protecting reaction.
- reaction of RCOR' with sodium amide results in cleavage with formation of the amide RCONH2 and the alkane R'H, a reaction called the Haller-Bauer reaction (1909) [3]
- Electrophilic addition, reaction with an electrophile gives a resonance stabilized cation.
- the reaction with phosphonium ylides in the Wittig reaction gives alkenes
- reaction with water gives geminal diols
- reaction with thiols gives a thioacetal
- reaction with hydrazine or derivatives of hydrazine gives hydrazones
- reaction with a metal hydride gives a metal alkoxide salt and then with water an alcohol
- reaction of an enol with halogens to α-haloketone
- a reaction at an α-carbon is the reaction of a ketone with heavy water to give a deuterated ketone-d.
- fragmentation in photochemical Norrish reaction
- reaction with halogens and base of methyl ketones in the Haloform reaction
- reaction of 1,4-aminodiketones to oxazoles by dehydration in the Robinson-Gabriel synthesis
- reaction of aryl alkyl ketones with sulfur and an amine to amides in the Willgerodt reaction
Biochemistry
[edit | edit source]Acetone, acetoacetate and beta-hydroxybutyrate are ketones (or ketone bodies) generated from carbohydrates, fatty acids and amino acids in humans and most vertebrates. Ketones are elevated in blood after fasting including a night of sleep, and in both blood and urine in starvation, hypoglycemia due to causes other than hyperinsulinism, various inborn errors of metabolism, and ketoacidosis (usually due to diabetes mellitus). Although ketoacidosis is characteristic of decompensated or untreated type 1 diabetes, ketosis or even ketoacidosis can occur in type 2 diabetes in some circumstances as well. Acetoacetate and beta-hydroxybutyrate are an important fuel for many tissues, especially during fasting and starvation. The brain, in particular, relies heavily on ketone bodies as a substrate for lipid synthesis and for energy during times of reduced food intake. At the NIH, Dr. Richard Veech refers to ketones as "magic" in their ability to increase metobolic efficiency, while decreasing production of free radicals, the damaging byproducts of normal metabolism. His work has shown that ketone bodies may treat neurological diseases such as Alzheimer's and Parkinson's disease,[4] and the heart and brain operate 25% more efficiently using ketones as a source of energy.[5]
Applications
[edit | edit source]Ketones are often used in perfumes and paints to stabilize the other ingredients so that they don't degrade as quickly over time. Other uses are as solvents and intermediates in chemical industry. Examples of ketones are acetone, acetophenone, and methyl ethyl ketone.
See also
[edit | edit source]References
[edit | edit source]- ↑ The position of the carbonyl group is usually denoted by a number; in propanone there can only be one position. While propanone or 2-propanone is how the molecule should be named according to systematic nomenclature, the name "acetone" is retained in official IUPAC nomenclature
- ↑ List of retained IUPAC names retained IUPAC names Link
- ↑ http://nagoyaren.homeip.net/chem/reactions/123.htm
- ↑ Y. Kashiwaya, T. Takeshima, N. Mori, K. Nakashima, K. Clarke and R. L. Veech (2000). "D-beta -Hydroxybutyrate protects neurons in models of Alzheimer's and Parkinson's disease". PNAS. 97 (10): 5440–5444. doi:10.1073/pnas.97.10.5440.
- ↑ Y. Kashiwaya, K. Sato, N. Tsuchiya, S. Thomas, D. A. Fell, R. L. Veech and J. V. Passonneau (1994). "Control of glucose utilization in working perfused rat heart". J. Biol. Chem. 269 (41): 25502–25514.
ar:كيتون bg:Кетон ca:Cetona cs:Ketony da:Keton de:Ketone et:Ketoonid eo:Ketono fa:کتون ko:케톤 it:Chetoni he:קטון lv:Ketoni lt:Ketonai hu:Keton mk:Кетон nl:Keton no:Keton sk:Ketón sr:Кетон fi:Ketoni sv:Keton
 KSF
KSF