Sepsis medical therapy
 From Wikidoc - Reading time: 41 min
From Wikidoc - Reading time: 41 min
|
Sepsis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Sepsis medical therapy On the Web |
|
American Roentgen Ray Society Images of Sepsis medical therapy |
|
Risk calculators and risk factors for Sepsis medical therapy |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Priyamvada Singh, M.B.B.S.Aditya Ganti M.B.B.S. [2]; Parth Vikram Singh, MBBS[3]
Overview
[edit | edit source]The goals for the treatment of sepsis per the Surviving Sepsis Campaign include screening for high-risk patients; taking bacterial cultures soon after the patient arrived at the hospital; starting patients on broad-spectrum intravenous antibiotic therapy before the results of the cultures are obtained; identifying the source of infection and taking steps to control it (e.g., abscess drainage); administering intravenous fluids to correct a loss or decrease in blood volume; and maintaining glycemic (blood sugar) control.[1][2] These and similar guidelines have been tested by a number of hospitals and have shown potential for decreasing hospital mortality due to sepsis.[3][4][5] In addition, hospital length of stay may be shortened.[5][6]
Electronic alerts in the electronic health record may[7] or may not[8] improve outcomes for sepsis patients.[9][10] Alerts may trigger intervention by a hospital rapid response team.
Medical Therapy
[edit | edit source]Adherence to bundles of care is associated with improved outcomes[11].

Fluid therapy
[edit | edit source]Guidelines
[edit | edit source]The 2012 Surviving Sepsis Campaign (SSC) recommendations proposed:[1][2]
- Fluid challenge of at least 30 ml/kg of crystalloid in patients with sepsis-induced tissue hypoperfusion with suspicion of hypovolemia.
- However, due to variable patient response to treatment, the SSC guidelines also state that “more rapid administration and greater amounts of fluid may be needed in patients with sepsis-induced tissue hypoperfusion.”
The 30 mL/kg initial bolus remains the current guideline standard. Recent large randomized trials (SMART, BaSICS) demonstrated that balanced crystalloids such as lactated Ringer’s are preferred over normal saline (0.9%), owing to better renal outcomes and survival. Furthermore, trials comparing fluid-liberal versus restrictive strategies (CLASSIC, CLOVERS) found no major mortality difference, highlighting the need for personalized, physiology-guided fluid resuscitation strategies such as those tested in the FRESH trial.[12]
The SSC guidelines also targeted a mean arterial pressure (MAP) of at least 65 mmHg and a central venous pressure (CVP) of
- At least 8 mmHg in the non mechanically-ventilated patient
- At least 12-15 mmHg in the mechanically-ventilated patient
In National Institute for Health and Care Excellence (NICE) guidelines, as proposed by the National Guideline Centre (UK), a systematic review of randomized controlled trials concluded that patients over 16 years with severe sepsis or septic shock requiring fluid resuscitation should receive
- Crystalloids containing sodium in the range 130–154 mmol/litre with a bolus of 500 ml over less than 15 minutes.”
- The patient should then be reassessed and, if fluid status has not improved, receive a second bolus.
- If the patient’s status does not improve following the second bolus, the guidelines recommend consultation of a specialist.
Evidence
[edit | edit source]| Rivers, 2001[13] | ProCESS, 2014[14] | ARISE, 2014[15] | ProMISe, 2015[16] | CLASSIC, 2022[17] | |
|---|---|---|---|---|---|
| Lactate, mean (treatment; control |
6.9; 7.7 | 4.8; 5.0 | 6.7; 6.6 | 7.0; 6.8 | 3.8; 3.9 (median) |
| Fluids in first 6 hours (treatment; control) |
4900; 3500 | 3300; 2800 | 2000; 1700 | 2000; 1800 (median) |
1000; 1700 (24 hours) |
| Mortality (treatment; control) |
44%; 57% (60 days) |
21%; 18% | 19%; 19% | 30%; 29% | 42%; 42% |
EGDT to guide fluid therapy
[edit | edit source]Regarding underlying evidence, a systematic review of randomized control trials concluded:[18]
- Strict adherence to early goal directed therapy of septic shock is not necessary.
- However, principles of early identification and fluid resuscitation, prompt administration of antibiotics, and adequate monitoring of patient parameters should be considered in treatment of patients with severe sepsis and septic shock.
A second systematic review of ProCESS, ProMISe, and ARISE randomized controlled trials, concluded:[19]
- Early goal directed therapy remains the gold standard for treatment of severe sepsis and septic shock given that EGDT has proven internal and external validity.
- While non-inferior outcomes may be able to be achieved using alternative treatment strategies, there is a lack of sufficient evidence backing other protocols.
A third meta-analysis concluded:[20]
- In patient populations with higher mortality, EGDT or lactate level and central venous pressure normalization are viable therapies.
- Newer evidence challenges the need for strict adherence to six hour bundle goals in populations with lower mortality.
Another meta-analysis by the recent trialists[21] of the three most recent trials[15][14][16] found that low mortality was achieved even in the control groups in all three studies which was based on the standard care at each institution. The amount of fluid administered in these three trials was 30 ml/kg prior to enrollment[19] and then ranged from 1.9-2.6 L in the first three hours prior to diagnosis of septic shock. Approximately 1.7-3.3 L of additional fluid was given once the septic shock protocol was initiated in both the control and EGDT groups. Total fluids at 9 hours averaged between 4.0 - 5.5 L.
Trials of other protocols to guide fluid therapy
[edit | edit source]| Score | Description |
|---|---|
| 0 | No mottling |
| 1 | "Small mottling area (coinsize) localized to the center of the knee" |
| 2 | "A mottling area that does not exceed the superior edge of the knee cap" |
| 3 | "A mottling area that does not exceed the middle thigh" |
| 4 | "A mottling area that does not go beyond the fold of the groin" |
| 5 | "An extremely severe mottling area that goes beyond the fold of the groin |
In the CLASSIC randomized controlled trial, after an initial 30 ml/kg fluid bolus, the goal of resuscitation was mean arterial pressure (MAP) of at least 65 mm Hg. Further fluids in the experimental group (fluid boluses of 250–500 mL every 30 minutes) were allow if[23]:
- Lactate level of at least 4 mmol/L
- "MAP below 50 mmHg in spite of the infusion of norepinephrine"
- "Mottling beyond the edge of the kneecap (mottling score[22] greater than 2)"
- "Oliguria, but only in the first 2 h after randomisation, defined as urinary output at most 0.1 mL/kg IBW in the last hour."
The trial found an insignificant trend towards benefit in the experimental group.
In the ANDROMEDA-SHOCK randomized controlled trial, after an initial 20 mL/kg or more over 60 minutes, the goal of resuscitation was mean arterial pressure (MAP) of at least 65 mm Hg (in patients with chronic hypertension, a higher goal might be used). Further fluids in the experimental group (500 mL of crystalloids every 30 minutes) were allowed if[24]:
- Capillary refill time (CRT) was greater than 3 seconds. The CRT was measured by "applying firm pressure to the ventral surface of the right index finger distal phalanx with a glass microscope slide. The pressure was increased until the skin was blank and then maintained for 10 seconds. The time for return of the normal skin color was registered with a chronometer". "The CRT was evaluated every 30 minutes, because of its faster rate of recovery, until normalization and then every hour during the intervention period". The intervention period was 8 hours.
- Central venous pressure safety was not elevated
- The patient became fluid unresponsive as measured by one of several methods[25]
The trial found an insignificant trend towards benefit in the experimental group.
In the small, pilot Echo vs EGDT randomized controlled trial at Intermountain Healthcare, after a median of 35 mL/kg of fluids, the goal of resuscitation was mean arterial pressure (MAP) of at least 65 mm Hg. Further fluids in the experimental group (1 L of crystalloids every hour) were allowed if[26]
- CVP less than 8–12 mm Hg or, if no central line, shock index ≥ 1
- IVC collapsibility present. Collapsibility was defined by "the maximum diameter was < 5 mm or if vena cava collapsibility index (VCCI), defined as the difference in diameter during a respiratory cycle divided by the maximum diameter, was > 50%"
No differences were found in the results; however, 70% of patients has reached their lactate clearance goal after initial resuscitation and before the trial protocol started.
In the small RIFTS randomized controlled trial that compared ≤ 60 mL/kg of IV fluid) or usual care for the first 72 hours of care found no less fluids administered in the intervention without a difference in mortality≤ 60 mL/kg of IV fluid) or usual care for the first 72 hours of care≤ 60 mL/kg of IV fluid) or usual care for the first 72 hours of care[27] .
Timing of fluid therapy
[edit | edit source]Two cohort studies examined the impact of the timing of fluid therapy for severe sepsis and septic shock. Taken together, the cohorts suggest fluids should be started as fast as possible, but the total amount to be infused and the infusion rate are not clear.
- Regarding administration of 30 ml/kg of fluids, reduced time to start of administration is associated with increased survival; however, there is not a clear relationship between mortality and time of completion of fluid bolus as this was not measured in this study[28].
- Completing a 3 hour bundle for sepsis care and rapidly administering antibiotics showed a lower risk-associated in-hospital mortality. However, there was insufficient evidence in this study to conclude that rapid completion of the initial fluid bolus affected mortality[29].
Speed of fluid therapy
[edit | edit source]Regarding the speed fluid administration, only two trials revealed an explicit protocol[14][30]. These showed the fluids being administered in 500-1,000mL boluses every 30 minutes.
Composition of fluids
[edit | edit source]Regarding type of fluids, a meta-analysis concluded "among the patients with sepsis, fluid resuscitation with crystalloids compared to starch resulted in reduced use of RRT; the same may be true for albumin versus starch."[31]
A more recent trial of patients with diverse diagnoses found no difference.[32]
Fluid administration in cardiac and renal disease
[edit | edit source]The 2016 Surviving Sepsis guidelines do not make specific reference to fluid resuscitation in the setting of cardiorenal dysfunction.[33]
A systematic review in 2017 found that conservative fluids after resuscitation may be beneficial[34]
- A retrospective review addressing the implementation of a sepsis bundle for sepsis patients and intermediate patients (lactate levels between 2 and 4 mmol/L) found that for a subgroup of patients (46% of total) with a known history of heart failure or chronic kidney disease (both defined by any previous diagnosis) had a statistically significant decrease in 30 day mortality from 12.5% to 8.7% after implementation of the bundle and subsequent increase in mean fluid totals from 1.4L to 1.7L.[35]
Excessive fluid therapy
[edit | edit source]Positive fluid balance may be associated with worse outcomes in most[36][37][38][39][40][41][42], but not all[43] studies. For example, a retrospective review of patient in Vasopressin in Septic Shock Trial (VASST) determined positive fluid balance initially at 12 hr and cumulatively at 4 days resulted in higher mortality.[41]
De-escalation and removal of fluids
[edit | edit source]Avoidance of fluid overload may be avoid be careful restriction of fluids after the initial 30 ml/kg bolus according to a randomized controlled trial.[23] Expert opinion suggests positive fluid balance should be addresses within three days of resuscitation. [44]
Lactate level
[edit | edit source]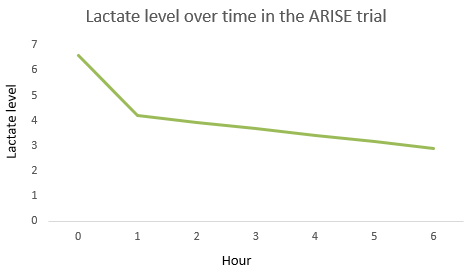
Lactate elevation seems due to aerobic glycolysis in skeletal muscle secondary to epinephrine stimulation[45].
The 2016 Surviving Sepsis Guidelines recommend:[46]
- "We suggest guiding resuscitation to normalize lactate in patients with elevated lactate levels as a marker of tissue hypoperfusion (weak recommendation, low quality of evidence)."
The guidelines by the Surviving Sepsis Campaign (SSC) do not specify how to response to slow lactate clearance. Two trails, both reporting reduced mortality from lactate-guided therapy, provide specific suggestions for responding to slow clearance:
- When the clearance falls by less than 10% over 1 hours, transfuse pack red blood cells or infuse the inotrope dobutamine is recommended by Jones[47]. Since publication of this trial, a subsequent trial showed that a hemoglobin of 7 g/dl is adequate[48]. In addition, subsequent guidelines by the SSC recommend norepinephrine as the first-choice vasopressor[46].
- When the clearance falls by less than 20% over 2 hours, either administer fluids, transfuse pack red blood cells, infuse the inotrope dobutamine, or increase administration of oxygen is recommended by Jansen[49]. However, since publication of this trial, subsequent guidelines by the SSC recommend norepinephrine as the first-choice vasopressor[46].
Evidence
[edit | edit source]Randomized controlled trials of monitoring lactate clearance to guide fluid resuscitation over the first 12 hours of resuscitation in sepsis and septic shock have been summarized in meta-analyses. A meta-analysis of four randomized controlled trials with 547 patients reported mortality benefit (RR=0.67, p=0.002) when lactate clearance strategies were used compared with early goal-directed therapy (EGDT) or usual care[50]. A meta-analysis of 4 trials with 448 patients showed a comparable mortality benefit (RR=0.64) with the use of lactate clearance strategies versus EGDT[20].
Hemodynamic monitoring
[edit | edit source]Regarding the monitoring of fluid administration, few randomized controlled trials used an explicit protocol[14][30]. These showed the fluids being administered in 500-1,000mL boluses every 30 minutes interspersed with more frequent patient reassessment than is currently required by both the Surviving Sepsis and CMS protocols. The patients in these studies were reevaluated at the time of each bolus administration (every 30 minutes.)
A meta-analysis of randomized controlled trials using early goal directed therapy found no significant benefit of the mandated use of central venous catheterization and central hemodynamic monitoring in all patients.[21]
Methods of assessing fluid responsiveness
[edit | edit source]Various methods are available and have been reviewed.[51][52][53][54]
Antibiotics
[edit | edit source]For every hour delay in the administration of appropriate antibiotic therapy there is an associated 4% rise in mortality.[55]
The Infectious Disease Society of America (IDSA) recommends that only patients with septic shock need antibiotics within one hour[56].
Timing of antibiotics
[edit | edit source]A systematic review found benefit from administration of antibiotics within one hour of diagnosis.[57] Several more recent observational studies have found benefits as well[58][59]. A recent cohort found benefit, "Each hour until initial antimicrobial administration was associated with a 8.0% increase in progression to septic shock".[60]
Early empiric antibiotics remain critical; delays are consistently associated with increased mortality. Avoidance of unnecessary anaerobic coverage (for example, routine use of piperacillin–tazobactam) is now emphasized, as excess coverage disrupts the microbiota and worsens outcomes. In addition, antimicrobial stewardship highlights de-escalation and shorter course durations tailored to infection site and clinical response.[61]
Administrating antibiotics before cultures will reduce the positivity rate of cultures. Two cohorts have found a decrease in positivity[62][63][63]; one looked at varying intervals and suggests harm starts as soon as 30 minutes after antibitoics[63].
Dissenting views
[edit | edit source]In 2019, a systematic review found no statistically significant difference in survival between projects that targeted antibiotics administered within 1 hour (7 studies) and 3 hours (8 studies)[64]. However, the studies did not reliably achieve rapid antibiotic administration. For example, among the studies targeting antibiotics within one hour, before project implementation, the rate of rapid administration was 55% and afterwards was 63%[65]. Thus, this study shows the difficulty in achieving effectiveness from administration within one hour rather than the efficacy when all patients receive antibiotics within one hour[66].
While an older systematic review found there was no significant mortality benefit from administering antibiotics within 3 hours of emergency department triage ( OR 1.16, 0.92 to 1.46, p = 0.21) or within 1 hour of shock recognition (OR 1.46, 0.89 to 2.40, p = 0.13) in severe sepsis and septic shock, several concerns exist:[67]
- The meta-analysis reported heterogeneity as insignificant (p = 0.09) with Cochran Q test. However, this is a significant result per the Cochrane collaboration's interpretation of the Cochran Q test.[68]
- The study grouped patients by time to administration of antimicrobial therapy, but did not consider time to administration of appropriate antimicrobial therapy- likely lessening the effect of early therapy.[69]
- One article[70] contributed the majority of the sample size for the 1h group- with said sample including patients with higher sepsis severity scores, higher rates of septic shock, and higher rates of mortality- leading to a potential underestimation of the impact of early antibiotic use.[71]
The two largest studies in the meta-analysis both found positive correlations between delays in antibiotics and mortality.[70][72]
Methods to improve speed of antibiotic administration
[edit | edit source]Regarding the improvement of speed to antibiotic administration, a controlled case series demonstrated a shorter time interval between ordering an antibiotic and its administration by using a hospital rapid response team (RRT) (median time in group without RRT activation was 157 minutes vs group with RRT activation of 54 minutes; p < .01). The RRT in this case series included a pharmacist. [73]
Antibiotic selection
[edit | edit source]The use of appropriate antibiotics, defined by most researchers as having in vitro activity against an isolated pathogen, in patients with bacteremia and/or severe sepsis or septic shock is associated with decreased mortality[74][75][76]. Lee (OR 2.26, 95% CI 1.10-5.13, p=0.04) and Nygard (OR 2.17, 95% CI 1.10-4.27, p=0.027) found that inappropriate antibiotics were associated with increased 28-day mortality[76][77]. Garnacho reported that adequate antibiotics are protective against mortality (OR 0.40, 95% CI 0.24-0.65, p<0.001)[77]. Observing a population of sixteen patients with urosepsis, Flaherty had similar conclusions, citing 18% overall mortality versus 25% among four patients who did not receive adequate antibiotic therapy[74].
Existing literature demonstrates that emergency department (ED) physicians prescribe appropriate initial antibiotic therapy 82-90% of the time[74][78], when treating patients with severe sepsis or septic shock. Other studies demonstrated similar rates of 80-81% among non-ED physicians[79][80]. Capp, et al. posited that inappropriate antibiotics were more likely to be prescribed in cases of gram-negative organisms[78]. Similarly, Flaherty, et al. found that antibiotic coverage was most appropriately chosen in patients presenting with pneumonia (97%); treatment failure was more common among the subgroup of patients with urinary tract infections, likely due to multidrug resistance[74].
Delivery method
[edit | edit source]Continuous infusion may be more effective.[81]
Pressors
[edit | edit source]Pressors that have been used are:
- Dopamine
- Epinephrine - Alpha and beta-agonist. Transiently raises the lactate level from baseline of 4.1 to 4.9[82].
- Norepinephrine - Alpha and beta-agonist
- Vasopressin
Evidence
[edit | edit source]In a randomized controlled trial comparing dopamine and norepinephrine, efficacy was similar but less adverse effect with norepinephrine[83] The change in lactate levels was similar in the two groups[83].
Pressors titrated to MAP of 80 to 85 mm Hg, as compared with 65 to 70 mm Hg does not change affect mortality among patients who already received a minimum 30 mL/kg within 6 hours prior to the start of catecholamines[84].
Norepinephrine remains the recommended first-line vasopressor. In older adults, permissive hypotension with a mean arterial pressure target of 60–65 mmHg (as studied in the 65 Trial) has been shown to be safe. When norepinephrine requirements escalate, vasopressin is commonly added; further clarification of its role is anticipated from ongoing clinical trials (e.g., NCT06217562).[85]
Giving pressors too early may be harmful[86].
Steroids
[edit | edit source]Corticosteroids may reduce mortality among patients with septic shock according to a meta-analysis[87] that includes the APROCCHSS[88] and ADRENAL[89] randomized controlled trials.
Corticosteroids may reduce mortality among patients with septic shock according to a systematic review by the Cochrane Collaboration.[90] However, a second systematic review published the same year found insignificant benefit from steroids[91].
The subsequent HYPRESS randomized controlled trial of patients with severe sepsis showed no benefit.[92]
Recent meta-analyses suggest that hydrocortisone combined with fludrocortisone shortens shock duration and may provide superior outcomes compared with hydrocortisone alone (Bosch et al. 2023). Nevertheless, due to heterogeneity in patient response, individualized use is recommended rather than universal application.[93]
Non-randomized studies reach conflicting conclusions with benefit found in the Intermountain cohort[5] and harm found in the Surviving Sepsis cohort[94].
The benefit of steroids may be confined to patients who have the SRS 1 variant (immunosuppressed) of the transcriptomic sepsis response signatures (SRS)[95].
The somewhat results of the Adjunctive Corticosteroid Treatment in Critically Ill Patients With Septic Shock (ADRENAL) and ADRENAL and Activated Protein C and Corticosteroids for Human Septic Shock (APROCCHSS) randomized controlled trials has been extensively debated[96].
Mineralocorticoid
[edit | edit source]Fludrocortisone was not effective in the COIITSS randomized controlled trial as a add on treatment to hydrocortisone[97].
Role of relative adrenal insufficiency
[edit | edit source]Patients who had a low response to at corticotropin stimulation test were not more likely to respond to a combination of hydrocortisone and fludrocortisone in the APROCCHSS trial[88].
Transfusion
[edit | edit source]In septic shock, leukoreduced erythrocyte transfusion is associated with similar clinical outcomes (90 day mortality and ischemic events) among patients who are administered erythrocyte transfusion at a hemoglobin threshold of 7 g/dL compared to those who receive it at a higher threshold of 9 g/dL.[48]
Protocolized therapy
[edit | edit source]Several protocols have been recommended and studied. Compliance with bundles of care is associated with reduced mortality.[5]
Early Goal Directed Therapy (EGDT)
[edit | edit source]Early Goal Directed Therapy (EGDT), developed at Henry Ford Hospital by E. Rivers, MD, is a systematic approach to resuscitation that has been validated in the treatment of severe sepsis and septic shock.[13] It is meant to be started in the Emergency Department. The theory is that one should use a step-wise approach, having the patient meet physiologic goals, to optimize cardiac preload, afterload, and contractility, thus optimizing oxygen delivery to the tissues.
Although initial studies reported benefit from EGDT,[47][13][98] the more recent ProCESS[14] and ARISE[15] trials failed to demonstrate any benefit. However, the outcomes in the control groups of these trials were much more favorable than in the earlier trials. The extent of protocol-based care in the 'usual care' of the control groups is not known.
In Early Goal Directed Therapy:
- Fluids are administered until the central venous pressure (CVP), as measured by a central venous catheter, reaches 8-12 cm of water (or 10-15 cm of water in mechanically ventilated patients).
- If the mean arterial pressure is less than 65 mmHg or greater than 90 mmHg, vasopressors or vasodilators are given as needed to reach the goal.
- The central venous saturation (ScvO2), i.e. the oxygen saturation of venous blood as it returns to the heart as measured at the superior vena cava, is optimized. If the ScvO2 is less than 70%, blood is given to reach a hemoglobin of 10 g/dl and then inotropes are added until the ScvO2 is optimized. Elective intubation may be performed to reduce oxygen demand if the ScvO2 remains low despite optimization of hemodynamics.
- Urine output is also monitored, with a goal of 0.5 mL/kg/h. In the original trial, mortality was cut from 46.5% in the control group to 30.5% in the intervention group. The Surviving Sepsis Campaign guidelines recommends EGDT for the initial resuscitation of the septic patient with a level B strength of evidence.
GENESIS Project
[edit | edit source]The protocol per the GENESIS Project is:[98]
- Measure serum lactate
- Obtain blood cultures and administer broad-spectrum antibiotic within 3 hours of emergency department admission
- If hypotensive or serum lactate 4 mmol/L:
- Administer 20 mL/kg of crystalloid
- If needed, add vasopressors to keep mean arterial pressure >65 mm Hg
- If needed, aim for central venous pressure 8 mm Hg
- If needed, aim for central venous oxygen saturation 70%
Standard treatment of infants with suspected sepsis consists of supportive care, maintaining fluid status with intravenous fluids, and the combination of a beta-lactam antibiotic (such as ampicillin) with an aminoglycoside such as gentamicin.
2018 Surviving Sepsis Campaign update
[edit | edit source]A major change with the update is that the 3 hour and 6 hour bundles were combined into one “Hour-1 Bundle.”[99] Per the Surviving Sepsis Campaign[100]:
The hour-1 bundle should be viewed as a quality improvement opportunity moving toward an ideal state. For critically ill patients with sepsis or septic shock, time is of the essence. Although the starting time for the Hour-1 bundle is recognition of sepsis, both sepsis and septic shock should be viewed as medical emergencies requiring rapid diagnosis and immediate intervention.The hour-1 bundle encourages clinicians to act as quickly as possible to obtain blood cultures, administer broad spectrum antibiotics, start appropriate fluid resuscitation, measure lactate, and begin vasopressors if clinically indicated. Ideally these interventions would all begin in the first hour from sepsis recognition but may not necessarily be completed in the first hour. Minimizing the time to treatment acknowledges the urgency that exists for patients with sepsis and septic shock.
*Note that the description of the hour-1 bundle above is the most current description, having passed all approval points effective October 10, 2019.
Hour-1 Bundle Pocket Card and Infographic
[edit | edit source]SSC Hour-1 Bundle of Care Elements:
- Measure lactate level*
- Obtain blood cultures before administering antibiotics.
- Administer broad-spectrum antibiotics.
- Begin rapid administration of 30mL/kg crystalloid for hypotension or lactate level ≥ 4 mmol/L.
- Apply vasopressors if hypotensive during or after fluid resuscitation to maintain MAP ≥ 65 mm Hg.
* Remeasure lactate if initial lactate is elevated (> 2 mmol/L).
2016 Surviving Sepsis Campaign: International Guidelines for Management of Severe Sepsis and Septic Shock (DO NOT EDIT)
[edit | edit source]2016 Bundle.[33]
[edit | edit source]| TO BE COMPLETED WITHIN 3 HOURS: |
|
| TO BE COMPLETED WITHIN 6 HOURS: |
|
| TABLE 1
DOCUMENT REASSESSMENT OF VOLUME STATUS AND TISSUE PERFUSION WITH: EITHER: • Repeat focused exam (after initial fluid resuscitation) including vital signs, cardiopulmonary, capillary refill, pulse, and skin findings. OR TWO OF THE FOLLOWING: • Measure CVP • Measure ScvO2 • Bedside cardiovascular ultrasound • Dynamic assessment of fluid responsiveness with passive leg raise or fluid challenge Of note, the 6-hour bundle has been updated from 2012; the 3-hour SSC bundle is not affected. |
Initial Resuscitation
[edit | edit source]Initial Resuscitation
[edit | edit source]1. Sepsis and septic shock are medical emergencies, and we recommend that treatment and resuscitation begin immediately (BPS).
2. We recommend that, in the resuscitation from sepsis-induced hypoperfusion, at least 30 mL/kg of IV crystalloid fluid be given within the first 3 hours (strong recommendation, low quality of evidence).
3. We recommend that, following initial fluid resuscitation, additional fluids be guided by frequent reassessment of hemodynamic status (BPS).
Remarks: Reassessment should include a thorough clinical examination and evaluation of available physiologic variables (heart rate, blood pressure, arterial oxygen saturation, respiratory rate, temperature, urine output, and others, as available) as well as other noninvasive or invasive monitoring, as available.
4. We recommend further hemodynamic assessment (such as assessing cardiac function) to determine the type of shock if the clinical examination does not lead to a clear diagnosis (BPS).
5. We suggest that dynamic over static variables be used to predict fluid responsiveness, where available.
6. We recommend an initial target mean arterial pressure of 65 mm Hg in patients with septic shock requiring vasopressors (strong recommendation, moderate quality of evidence).
7. We suggest guiding resuscitation to normalize lactate in patients with elevated lactate levels as a marker of tissue hypoperfusion (weak recommendation, low quality of evidence).
Screening for Sepsis and Performance Improvement
[edit | edit source]1. We recommend that hospitals and hospital systems have a performance improvement program for sepsis, including sepsis screening for acutely ill, high risk patients (BPS).
In a retrospective, quasi experimental study conducted in the Emergency Department, an EHR-based triage sepsis alert system and standardized protocol was implemented in an effort to identify adult patients with suspected sepsis, severe sepsis, or septic shock and improve patient outcomes. Using SIRS criteria in conjunction with physician approval, this triage system led to a significant reduction in the time to fluids and time to antibiotics, although no difference in mortality was found. [101]
Diagnosis
[edit | edit source]1. We recommend that appropriate routine microbiologic cultures (including blood) be obtained before starting antimicrobial therapy in patients with suspected sepsis or septic shock if doing so results in no substantial delay in the start of antimicrobials (BPS).
Remarks: Appropriate routine microbiologic cultures always include at least two sets of blood cultures (aerobic and anaerobic).
Antimicrobial Therapy
[edit | edit source]1. We recommend that administration of IV antimicrobials should be initiated as soon as possible after recognition and within one hour for both sepsis and septic shock (strong recommendation, moderate quality of evidence).
2. We recommend empiric broad-spectrum therapy with one or more antimicrobials for patients presenting with sepsis or septic shock to cover all likely pathogens (including bacterial and potentially fungal or viral coverage) (strong recommendation, moderate quality of evidence).
3. We recommend that empiric antimicrobial therapy be narrowed once pathogen identification and sensitivities are established and/or adequate clinical improvement is noted (BPS).
4. We recommend against sustained systemic antimicrobial prophylaxis in patients with severe inflammatory states of noninfectious origin (e.g., severe pancreatitis, burn injury) (BPS).
5. We recommend that dosing strategies of antimicrobials be optimized based on accepted pharmacokinetic/pharmacodynamic principles and specific drug properties in patients with sepsis or septic shock (BPS).
6. We suggest empiric combination therapy (using at least two antibiotics of different antimicrobial classes) aimed at the most likely bacterial pathogen(s) for the initial management of septic shock (weak recommendation, low quality of evidence.
Remarks: Readers should review Table 6 for definitions of empiric, targeted/definitive, broad-spectrum, combination, and multidrug therapy before reading this section.
7. We suggest that combination therapy not be routinely used for ongoing treatment of most other serious infections, including bacteremia and sepsis without shock (weak recommendation, low quality of evidence).
Remarks: This does not preclude the use of multidrug therapy to broaden antimicrobial activity.
8. We recommend against combination therapy for the routine treatment of neutropenic sepsis/bacteremia (strong recommendation, moderate quality of evidence).
Remarks: This does not preclude the use of multidrug therapy to broaden antimicrobial activity.
9. If combination therapy is used for septic shock, we recommend de-escalation with discontinuation of combination therapy within the first few days in response to clinical improvement and/or evidence of infection resolution. This applies to both targeted (for culture-positive infections) and empiric (for culture-negative infections) combination therapy (BPS).
10. We suggest that an antimicrobial treatment duration of 7 to 10 days is adequate for most serious infections associated with sepsis and septic shock (weak recommendation, low quality of evidence).
11. We suggest that longer courses are appropriate in patients who have a slow clinical response, undrainable foci of infection, bacteremia with Staphylococcus aureus, some fungal and viral infections, or immunologic deficiencies, including neutropenia (weak recommendation, low quality of evidence).
12. We suggest that shorter courses are appropriate in some patients, particularly those with rapid clinical resolution following effective source control of intra-abdominal or urinary sepsis and those with anatomically uncomplicated pyelonephritis (weak recommendation, low quality of evidence).
13. We recommend daily assessment for de-escalation of antimicrobial therapy in patients with sepsis and septic shock (BPS).
14. We suggest that measurement of procalcitonin levels can be used to support shortening the duration of antimicrobial therapy in sepsis patients (weak recommendation, low quality of evidence).
15. We suggest that procalcitonin levels can be used to support the discontinuation of empiric antibiotics in patients who initially appeared to have sepsis, but subsequently have limited clinical evidence of infection (weak recommendation, low quality of evidence).
Source Control
[edit | edit source]1. We recommend that a specific anatomic diagnosis of infection requiring emergent source control should be identified or excluded as rapidly as possible in patients with sepsis or septic shock, and that any required source control intervention should be implemented as soon as medically and logistically practical after the diagnosis is made (BPS).
2. We recommend prompt removal of intravascular access devices that are a possible source of sepsis or septic shock after other vascular access has been established (BPS).
3. Timely surgical or procedural intervention is strongly linked with improved outcomes, and effective multidisciplinary coordination (surgery, interventional radiology, intensive care) is often required for optimal source control.
Hemodynamic Support and Adjunctive Therapy
[edit | edit source]Fluid Therapy (of Severe Sepsis)
[edit | edit source]1. We recommend that a fluid challenge technique be applied where fluid administration is continued as long as hemodynamic factors continue to improve (BPS).
2. We recommend crystalloids as the fluid of choice for initial resuscitation and subsequent intravascular volume replacement in patients with sepsis and septic shock (strong recommendation, moderate quality of evidence).
3. We suggest using either balanced crystalloids or saline for fluid resuscitation of patients with sepsis or septic shock (weak recommendation, low quality of evidence).
4. We suggest using albumin in addition to crystalloids for initial resuscitation and subsequent intravascular volume replacement in patients with sepsis and septic shock, when patients require substantial amounts of crystalloids (weak recommendation, low quality of evidence).
5. We recommend against using hydroxyethyl starches for intravascular volume replacement in patients with sepsis or septic shock (strong recommendation, high quality of evidence).
6. We suggest using crystalloids over gelatins when resuscitating patients with sepsis or septic shock (weak recommendation, low quality of evidence).
Vasoactive Medications
[edit | edit source]1. We recommend norepinephrine as the first-choice vasopressor (strong recommendation, moderate quality of evidence).
2. We suggest adding either vasopressin (up to 0.03 U/min) (weak recommendation, moderate quality of evidence) or epinephrine (weak recommendation, low quality of evidence) to norepinephrine with the intent of raising mean arterial pressure to target, or adding vasopressin (up to 0.03 U/min) (weak recommendation, moderate quality of evidence) to decrease norepinephrine dosage.
3. We suggest using dopamine as an alternative vasopressor agent to norepinephrine only in highly selected patients (e.g., patients with low risk of tachyarrhythmias and absolute or relative bradycardia) (weak recommendation, low quality of evidence).
4. We recommend against using low-dose dopamine for renal protection (strong recommendation, high quality of evidence).
5. We suggest using dobutamine in patients who show evidence of persistent hypoperfusion despite adequate fluid loading and the use of vasopressor agents (weak recommendation, low quality of evidence).
Remarks: If initiated, dosing should be titrated to an end point reflecting perfusion, and the agent reduced or discontinued in the face of worsening hypotension or arrhythmias.
6. We suggest that all patients requiring vasopressors have an arterial catheter placed as soon as practical if resources are available (weak recommendation, very low quality of evidence).
Corticosteroids
[edit | edit source]1. We suggest against using IV hydrocortisone to treat septic shock patients if adequate fluid resuscitation and vasopressor therapy are able to restore hemodynamic stability. If this is not achievable, we suggest IV hydrocortisone at a dose of 200 mg per day (weak recommendation, low quality of evidence).
Other Supportive Therapy of Severe Sepsis
[edit | edit source]Blood Products
[edit | edit source]1. We recommend that RBC transfusion occur only when hemoglobin concentration decreases to < 7.0g/dL in adults in the absence of extenuating circumstances, such as myocardial ischemia, severe hypoxemia, or acute hemorrhage (strong recommendation, high quality of evidence).
2. We recommend against the use of erythropoietin for treatment of anemia associated with sepsis (strong recommendation, moderate quality of evidence).
3. We suggest against the use of fresh frozen plasma to correct clotting abnormalities in the absence of bleeding or planned invasive procedures (weak recommendation, very low quality of evidence).
4. We suggest prophylactic platelet transfusion when counts are < 10,000/mm3 (10×109/L) in the absence of apparent bleeding and when counts are < 20,000/mm3 (20×109/L) if the patient has a significant risk of bleeding. Higher platelet counts (≥ 50,000/mm3 [50 x 109/L]) are advised for active bleeding, surgery, or invasive procedures (weak recommendation, very low quality of evidence).
Immunoglobulins
[edit | edit source]1. We suggest against the use of IV immunoglobulins in patients with sepsis or septic shock (weak recommendation, low quality of evidence).
Blood Purification
[edit | edit source]1. We make no recommendation regarding the use of blood purification techniques.
Anticoagulants
[edit | edit source]1. We recommend against the use of antithrombin for the treatment of sepsis and septic shock (strong recommendation, moderate quality of evidence). 2. We make no recommendation regarding the use of thrombomodulin or heparin for the treatment of sepsis or septic shock.
Mechanical Ventilation
[edit | edit source]1. We recommend using a target tidal volume of 6 mL/kg predicted body weight compared with 12 mL/kg in adult patients with sepsis-induced acute respiratory distress syndrome (ARDS) (strong recommendation, high quality of evidence).
2. We recommend using an upper limit goal for plateau pressures of 30 cm H2O over higher plateau pressures in adult patients with sepsis-induced severe ARDS (strong recommendation, moderate quality of evidence).
3. We suggest using higher positive end-expiratory pressure (PEEP) over lower PEEP in adult patients with sepsis-induced moderate to severe ARDS (weak recommendation, moderate quality of evidence).
4. We suggest using recruitment maneuvers in adult patients with sepsis-induced, severe ARDS (weak recommendation, moderate quality of evidence).
5. We recommend using prone over supine position in adult patients with sepsis-induced ARDS and a PaO2/FIO2 ratio < 150 (strong recommendation, moderate quality of evidence).
6. We recommend against using high-frequency oscillatory ventilation in adult patients with sepsis-induced ARDS (strong recommendation, moderate quality of evidence).
7. We make no recommendation regarding the use of noninvasive ventilation for patients with sepsis-induced ARDS.
8. We suggest using neuromuscular blocking agents for ≤ 48 hours in adult patients with sepsis-induced ARDS and a PaO2/FIO2 ratio < 150 mm Hg (weak recommendation, moderate quality of evidence).
9. We recommend a conservative fluid strategy for patients with established sepsis-induced ARDS who do not have evidence of tissue hypoperfusion (strong recommendation, moderate quality of evidence).
10. We recommend against the use of ß-2 agonists for the treatment of patients with sepsis-induced ARDS without bronchospasm (strong recommendation, moderate quality of evidence).
11. We recommend against the routine use of the pulmonary artery catheter for patients with sepsis-induced ARDS (strong recommendation, high quality of evidence).
12. We suggest using lower tidal volumes over higher tidal volumes in adult patients with sepsis-induced respiratory failure without ARDS (weak recommendation, low quality of evidence).
13. We recommend that mechanically ventilated sepsis patients be maintained with the head of the bed elevated between 30 and 45 degrees to limit aspiration risk and to prevent the development of ventilator-associated pneumonia (strong recommendation, low quality of evidence).
14. We recommend using spontaneous breathing trials in mechanically ventilated patients with sepsis who are ready for weaning (strong recommendation, high quality of evidence).
15. We recommend using a weaning protocol in mechanically ventilated patients with sepsis-induced respiratory failure who can tolerate weaning (strong recommendation, moderate quality of evidence).
Sedation and Analgesia
[edit | edit source]1. We recommend that continuous or intermittent sedation be minimized in mechanically ventilated sepsis patients, targeting specific titration end points (BPS).
Glucose Control
[edit | edit source]1. We recommend a protocolized approach to blood glucose management in ICU patients with sepsis, commencing insulin dosing when two consecutive blood glucose levels are > 180 mg/dL. This approach should target an upper blood glucose level ≤
180 mg/dL rather than an upper target blood glucose level ≤ 110 mg/dL (strong recommendation, high quality of evidence).
2. We recommend that blood glucose values be monitored every 1 to 2 hours until glucose values and insulin infusion rates are stable, then every 4 hours thereafter in patients receiving insulin infusions (BPS).
3. We recommend that glucose levels obtained with point-of-care testing of capillary blood be interpreted with caution because such measurements may not accurately estimate arterial blood or plasma glucose values (BPS).
4. We suggest the use of arterial blood rather than capillary blood for point-of-care testing using glucose meters if patients have arterial catheters (weak recommendation, low quality of evidence).
Renal Replacement Therapy
[edit | edit source]1. We suggest that either continuous or intermittent renal replacement therapy (RRT) be used in patients with sepsis and acute kidney injury (weak recommendation, moderate quality of evidence)
2. We suggest using continuous therapies to facilitate management of fluid balance in hemodynamically unstable septic patients (weak recommendation, very low quality of evidence).
3. We suggest against the use of RRT in patients with sepsis and acute kidney injury for increase in creatinine or oliguria without other definitive indications for dialysis (weak recommendation, low quality of evidence).
4. We recommend pharmacologic prophylaxis (unfractionated heparin [UFH] or low-molecular-weight heparin [LMWH]) against venous thromboembolism (VTE) in the absence of contraindications to the use of these agents (strong recommendation, moderate quality of evidence).
5. We recommend LMWH rather than UFH for VTE prophylaxis in the absence of contraindications to the use of LMWH (strong recommendation, moderate quality of evidence).
6. We suggest combination pharmacologic VTE prophylaxis and mechanical prophylaxis, whenever possible (weak recommendation, low quality of evidence).
7. We suggest mechanical VTE prophylaxis when pharmacologic VTE is contraindicated (weak recommendation, low quality of evidence).
Bicarbonate Therapy
[edit | edit source]1. We suggest against the use of sodium bicarbonate therapy to improve hemodynamics or to reduce vasopressor requirements in patients with hypoperfusion-induced lactic acidemia with pH ≥ 7.15 (weak recommendation, moderate quality of evidence).
Stress Ulcer Prophylaxis
[edit | edit source]1. We recommend that stress ulcer prophylaxis be given to patients with sepsis or septic shock who have risk factors for gastrointestinal (GI) bleeding (strong recommendation, low quality of evidence).
2. We suggest using either proton pump inhibitors or histamine-2 receptor antagonists when stress ulcer prophylaxis is indicated (weak recommendation, low quality of evidence).
3. We recommend against stress ulcer prophylaxis in patients without risk factors for GI bleeding (BPS).
Nutrition
[edit | edit source]1. We recommend against the administration of early parenteral nutrition alone or parenteral nutrition in combination with enteral feedings (but rather initiate early enteral nutrition) in critically ill patients with sepsis or septic shock who can be fed enterally (strong recommendation, moderate quality of evidence).
2. We recommend against the administration of parenteral nutrition alone or in combination with enteral feeds (but rather to initiate IV glucose and advance enteral feeds as tolerated) over the first 7 days in critically ill patients with sepsis or septic shock for whom early enteral feeding is not feasible (strong recommendation, moderate quality of evidence).
3. We suggest the early initiation of enteral feeding rather than a complete fast or only IV glucose in critically ill patients with sepsis or septic shock who can be fed enterally (weak recommendation, low quality of evidence).
4. We suggest either early trophic/hypocaloric or early full enteral feeding in critically ill patients with sepsis or septic shock; if trophic/hypocaloric feeding is the initial strategy, then feeds should be advanced according to patient tolerance (weak recommendation, moderate quality of evidence).
5. We recommend against the use of omega-3 fatty acids as an immune supplement in critically ill patients with sepsis or septic shock (strong recommendation, low quality of evidence).
6. We suggest against routinely monitoring gastric residual volumes in critically ill patients with sepsis or septic shock (weak recommendation, low quality of evidence). However, we suggest measurement of gastric residuals in patients with feeding intolerance or who are considered to be at high risk of aspiration (weak recommendation, very low quality of evidence).
Remarks: Ths recommendation refers to nonsurgical critically ill patients with sepsis or septic shock.
7. We suggest the use of prokinetic agents in critically ill patients with sepsis or septic shock and feeding intolerance (weak recommendation, low quality of evidence).
8. We suggest placement of post-pyloric feeding tubes in critically ill patients with sepsis or septic shock with feeding intolerance or who are considered to be at high risk of aspiration (weak recommendation, low quality of evidence).
9. We recommend against the use of IV selenium to treat sepsis and septic shock (strong recommendation, moderate quality of evidenence).
10. We suggest against the use of arginine to treat sepsis and septic shock (weak recommendation, low quality of evidence).
11. We recommend against the use of glutamine to treat sepsis and septic shock (strong recommendation, moderate quality of evidence).
12. We make no recommendation about the use of carnitine for sepsis and septic shock.
Setting Goals of Care
[edit | edit source]1. We recommend that goals of care and prognosis be discussed with patients and families (BPS).
2. We recommend that goals of care be incorporated into treatment and end-of-life care planning, utilizing palliative care principles where appropriate (strong recommendation, moderate quality of evidence).
3. We suggest that goals of care be addressed as early as feasible, but no later than within 72 hours of ICU admission (weak recommendation, low quality of evidence).
References
[edit | edit source]- ↑ 1.0 1.1 Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM; et al. (2013). "Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012". Crit Care Med. 41 (2): 580–637. doi:10.1097/CCM.0b013e31827e83af. PMID 23353941.
- ↑ 2.0 2.1 Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM; et al. (2013). "Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012". Intensive Care Med. 39 (2): 165–228. doi:10.1007/s00134-012-2769-8. PMID 23361625.
- ↑ Rhodes A, Phillips G, Beale R, Cecconi M, Chiche JD, De Backer D; et al. (2015). "The Surviving Sepsis Campaign bundles and outcome: results from the International Multicentre Prevalence Study on Sepsis (the IMPreSS study)". Intensive Care Med. 41 (9): 1620–8. doi:10.1007/s00134-015-3906-y. PMID 26109396.
- ↑ Levy MM, Rhodes A, Phillips GS, Townsend SR, Schorr CA, Beale R; et al. (2015). "Surviving Sepsis Campaign: association between performance metrics and outcomes in a 7.5-year study". Crit Care Med. 43 (1): 3–12. doi:10.1097/CCM.0000000000000723. PMID 25275252.
- ↑ 5.0 5.1 5.2 5.3 Miller RR, Dong L, Nelson NC, Brown SM, Kuttler KG, Probst DR; et al. (2013). "Multicenter implementation of a severe sepsis and septic shock treatment bundle". Am J Respir Crit Care Med. 188 (1): 77–82. doi:10.1164/rccm.201212-2199OC. PMC 3735248. PMID 23631750.
- ↑ Leisman D, Wie B, Doerfler M, Bianculli A, Frances Ward M, Akerman M; et al. (2016). "Association of Fluid Resuscitation Initiation Within 30 Minutes of Severe Sepsis and Septic Shock Recognition With Reduced Mortality and Length of Stay". Ann Emerg Med. doi:10.1016/j.annemergmed.2016.02.044. PMID 27085369.
- ↑ Narayanan N, Gross AK, Pintens M, Fee C, MacDougall C (2016). "Effect of an electronic medical record alert for severe sepsis among ED patients". Am J Emerg Med. 34 (2): 185–8. doi:10.1016/j.ajem.2015.10.005. PMID 26573784.
- ↑ Semler MW, Weavind L, Hooper MH, Rice TW, Gowda SS, Nadas A; et al. (2015). "An Electronic Tool for the Evaluation and Treatment of Sepsis in the ICU: A Randomized Controlled Trial". Crit Care Med. 43 (8): 1595–602. doi:10.1097/CCM.0000000000001020. PMC 4506222. PMID 25867906.
- ↑ GitHub Contributors. Sepsis alerts to improve diagnosis: a living systematic review. GitHub. Available at https://github.com/openMetaAnalysis/Sepsis-alerts-to-improve-diagnosis/blob/master/README.md. Accessed March 15, 2017.
- ↑ Harrison AM, Gajic O, Pickering BW, Herasevich V (2016). "Development and Implementation of Sepsis Alert Systems". Clin Chest Med. 37 (2): 219–29. doi:10.1016/j.ccm.2016.01.004. PMC 4884325. PMID 27229639.
- ↑ Townsend SR, Phillips GS, Duseja R, Tefera L, Cruikshank D, Dickerson R; et al. (2021). "Effects of Compliance With the Early Management Bundle (SEP-1) on Mortality Changes Among Medicare Beneficiaries With Sepsis: A Propensity Score Matched Cohort Study". Chest. doi:10.1016/j.chest.2021.07.2167. PMID 34364867 Check
|pmid=value (help). - ↑ Meyer NJ, Prescott HC (December 2024). "Sepsis and Septic Shock". N Engl J Med. 391 (22): 2133–2146. doi:10.1056/NEJMra2403213. PMID 39774315 Check
|pmid=value (help). - ↑ 13.0 13.1 13.2 13.3 Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B; et al. (2001). "Early goal-directed therapy in the treatment of severe sepsis and septic shock". N Engl J Med. 345 (19): 1368–77. doi:10.1056/NEJMoa010307. PMID 11794169. Review in: ACP J Club. 2002 May-Jun;136(3):90
- ↑ 14.0 14.1 14.2 14.3 14.4 14.5 ProCESS Investigators. Yealy DM, Kellum JA, Huang DT, Barnato AE, Weissfeld LA; et al. (2014). "A randomized trial of protocol-based care for early septic shock". N Engl J Med. 370 (18): 1683–93. doi:10.1056/NEJMoa1401602. PMC 4101700. PMID 24635773. Review in: Ann Intern Med. 2014 Jun 17;160(12):JC9
- ↑ 15.0 15.1 15.2 15.3 ARISE Investigators. ANZICS Clinical Trials Group. Peake SL, Delaney A, Bailey M, Bellomo R; et al. (2014). "Goal-directed resuscitation for patients with early septic shock". N Engl J Med. 371 (16): 1496–506. doi:10.1056/NEJMoa1404380. PMID 25272316. Review in: Ann Intern Med. 2015 Mar 17;162(6):JC4
- ↑ 16.0 16.1 16.2 Mouncey PR, Osborn TM, Power GS, Harrison DA, Sadique MZ, Grieve RD; et al. (2015). "Trial of early, goal-directed resuscitation for septic shock". N Engl J Med. 372 (14): 1301–11. doi:10.1056/NEJMoa1500896. PMID 25776532. Review in: Ann Intern Med. 2015 Aug 18;163(4):JC10
- ↑ 17.0 17.1 Meyhoff TS, Hjortrup PB, Wetterslev J, Sivapalan P, Laake JH, Cronhjort M; et al. (2022). "Restriction of Intravenous Fluid in ICU Patients with Septic Shock". N Engl J Med. 386 (26): 2459–2470. doi:10.1056/NEJMoa2202707. PMID 35709019 Check
|pmid=value (help). - ↑ Rusconi AM, Bossi I, Lampard JG, Szava-Kovats M, Bellone A, Lang E (2015). "Early goal-directed therapy vs usual care in the treatment of severe sepsis and septic shock: a systematic review and meta-analysis". Intern Emerg Med. 10 (6): 731–43. doi:10.1007/s11739-015-1248-y. PMID 25982917.
- ↑ 19.0 19.1 Nguyen HB, Jaehne AK, Jayaprakash N, Semler MW, Hegab S, Yataco AC; et al. (2016). "Early goal-directed therapy in severe sepsis and septic shock: insights and comparisons to ProCESS, ProMISe, and ARISE". Crit Care. 20 (1): 160. doi:10.1186/s13054-016-1288-3. PMC 4929762. PMID 27364620.
- ↑ 20.0 20.1 Simpson SQ, Gaines M, Hussein Y, Badgett RG (2016). "Early goal-directed therapy for severe sepsis and septic shock: A living systematic review". J Crit Care. 36: 43–48. doi:10.1016/j.jcrc.2016.06.017. PMID 27546746.
- ↑ 21.0 21.1 Angus DC, Barnato AE, Bell D, Bellomo R, Chong CR, Coats TJ; et al. (2015). "A systematic review and meta-analysis of early goal-directed therapy for septic shock: the ARISE, ProCESS and ProMISe Investigators". Intensive Care Med. 41 (9): 1549–60. doi:10.1007/s00134-015-3822-1. PMID 25952825.
- ↑ 22.0 22.1 Ait-Oufella H, Lemoinne S, Boelle PY, Galbois A, Baudel JL, Lemant J; et al. (2011). "Mottling score predicts survival in septic shock". Intensive Care Med. 37 (5): 801–7. doi:10.1007/s00134-011-2163-y. PMID 21373821.
- ↑ 23.0 23.1 Hjortrup PB, Haase N, Bundgaard H, Thomsen SL, Winding R, Pettilä V; et al. (2016). "Restricting volumes of resuscitation fluid in adults with septic shock after initial management: the CLASSIC randomised, parallel-group, multicentre feasibility trial". Intensive Care Med. 42 (11): 1695–1705. doi:10.1007/s00134-016-4500-7. PMID 27686349.
- ↑ Hernández G, Ospina-Tascón GA, Damiani LP, Estenssoro E, Dubin A, Hurtado J; et al. (2019). "Effect of a Resuscitation Strategy Targeting Peripheral Perfusion Status vs Serum Lactate Levels on 28-Day Mortality Among Patients With Septic Shock: The ANDROMEDA-SHOCK Randomized Clinical Trial". JAMA. doi:10.1001/jama.2019.0071. PMID 30772908.
- ↑ Monnet X, Marik PE, Teboul JL (2016). "Prediction of fluid responsiveness: an update". Ann Intensive Care. 6 (1): 111. doi:10.1186/s13613-016-0216-7. PMC 5114218. PMID 27858374.
- ↑ Lanspa MJ, Burk RE, Wilson EL, Hirshberg EL, Grissom CK, Brown SM (2018). "Echocardiogram-guided resuscitation versus early goal-directed therapy in the treatment of septic shock: a randomized, controlled, feasibility trial". J Intensive Care. 6: 50. doi:10.1186/s40560-018-0319-3. PMC 6090604. PMID 30123511.
- ↑ Corl KA, Prodromou M, Merchant RC, Gareen I, Marks S, Banerjee D; et al. (2019). "The Restrictive IV Fluid Trial in Severe Sepsis and Septic Shock (RIFTS): A Randomized Pilot Study". Crit Care Med. doi:10.1097/CCM.0000000000003779. PMID 30985449.
- ↑ Pruinelli L, Westra BL, Yadav P, Hoff A, Steinbach M, Kumar V; et al. (2018). "Delay Within the 3-Hour Surviving Sepsis Campaign Guideline on Mortality for Patients With Severe Sepsis and Septic Shock". Crit Care Med. 46 (4): 500–505. doi:10.1097/CCM.0000000000002949. PMC 5851815. PMID 29298189.
- ↑ Seymour CW, Gesten F, Prescott HC, Friedrich ME, Iwashyna TJ, Phillips GS; et al. (2017). "Time to Treatment and Mortality during Mandated Emergency Care for Sepsis". N Engl J Med. 376 (23): 2235–2244. doi:10.1056/NEJMoa1703058. PMC 5538258. PMID 28528569.
- ↑ 30.0 30.1 Kuan WS, Ibrahim I, Leong BS, Jain S, Lu Q, Cheung YB; et al. (2016). "Emergency Department Management of Sepsis Patients: A Randomized, Goal-Oriented, Noninvasive Sepsis Trial". Ann Emerg Med. 67 (3): 367–378.e3. doi:10.1016/j.annemergmed.2015.09.010. PMID 26475246.
- ↑ Rochwerg B, Alhazzani W, Gibson A, Ribic CM, Sindi A, Heels-Ansdell D; et al. (2015). "Fluid type and the use of renal replacement therapy in sepsis: a systematic review and network meta-analysis". Intensive Care Med. 41 (9): 1561–71. doi:10.1007/s00134-015-3794-1. PMID 25904181.
- ↑ Young P, Bailey M, Beasley R, Henderson S, Mackle D, McArthur C; et al. (2015). "Effect of a Buffered Crystalloid Solution vs Saline on Acute Kidney Injury Among Patients in the Intensive Care Unit: The SPLIT Randomized Clinical Trial". JAMA. 314 (16): 1701–10. doi:10.1001/jama.2015.12334. PMID 26444692.
- ↑ 33.0 33.1 Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R; et al. (2017). "Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016". Intensive Care Med. doi:10.1007/s00134-017-4683-6. PMID 28101605.
- ↑ Silversides JA, Major E, Ferguson AJ, Mann EE, McAuley DF, Marshall JC; et al. (2017). "Conservative fluid management or deresuscitation for patients with sepsis or acute respiratory distress syndrome following the resuscitation phase of critical illness: a systematic review and meta-analysis". Intensive Care Med. 43 (2): 155–170. doi:10.1007/s00134-016-4573-3. PMID 27734109. </. Two previous studies suggest possible benefits of including this subset of patients in sepsis protocol bundles administration without significant harm:
- In one open-labeled randomized controlled trial, results for a subgroup of patients (11% of total) with a diagnosis of underlying fluid overload state (defined as a history of heart failure, ejection fraction less than 50%, or end-stage renal disease) showed that the intervention group received clinically significant more IV fluids than the control group (difference = 975 mls, 95% CI: -450 ml to +1725 ml) and attained shorter hospital lengths of stay (difference -4.5 days, 95% CI -9.5-2.5 days).<ref name="pmid26475246">Kuan WS, Ibrahim I, Leong BS, Jain S, Lu Q, Cheung YB; et al. (2016). "Emergency Department Management of Sepsis Patients: A Randomized, Goal-Oriented, Noninvasive Sepsis Trial". Ann Emerg Med. 67 (3): 367–378.e3. doi:10.1016/j.annemergmed.2015.09.010. PMID 26475246.
- ↑ Liu VX, Morehouse JW, Marelich GP, Soule J, Russell T, Skeath M; et al. (2016). "Multicenter Implementation of a Treatment Bundle for Patients with Sepsis and Intermediate Lactate Values". Am J Respir Crit Care Med. 193 (11): 1264–70. doi:10.1164/rccm.201507-1489OC. PMC 4910898. PMID 26695114.
- ↑ Neyra JA, Li X, Canepa-Escaro F, Adams-Huet B, Toto RD, Yee J; et al. (2016). "Cumulative Fluid Balance and Mortality in Septic Patients With or Without Acute Kidney Injury and Chronic Kidney Disease". Crit Care Med. 44 (10): 1891–900. doi:10.1097/CCM.0000000000001835. PMID 27352125.
- ↑ Brotfain E, Koyfman L, Toledano R, Borer A, Fucs L, Galante O; et al. (2016). "Positive fluid balance as a major predictor of clinical outcome of patients with sepsis/septic shock after discharge from intensive care unit". Am J Emerg Med. doi:10.1016/j.ajem.2016.07.058. PMID 27553826.
- ↑ Mitchell KH, Carlbom D, Caldwell E, Leary PJ, Himmelfarb J, Hough CL (2015). "Volume Overload: Prevalence, Risk Factors, and Functional Outcome in Survivors of Septic Shock". Ann Am Thorac Soc. 12 (12): 1837–44. doi:10.1513/AnnalsATS.201504-187OC. PMC 4722831. PMID 26394090.
- ↑ Acheampong A, Vincent JL (2015). "A positive fluid balance is an independent prognostic factor in patients with sepsis". Crit Care. 19: 251. doi:10.1186/s13054-015-0970-1. PMC 4479078. PMID 26073560.
- ↑ de Oliveira FS, Freitas FG, Ferreira EM, de Castro I, Bafi AT, de Azevedo LC; et al. (2015). "Positive fluid balance as a prognostic factor for mortality and acute kidney injury in severe sepsis and septic shock". J Crit Care. 30 (1): 97–101. doi:10.1016/j.jcrc.2014.09.002. PMID 25269788.
- ↑ 41.0 41.1 Boyd JH, Forbes J, Nakada TA, Walley KR, Russell JA (2011). "Fluid resuscitation in septic shock: a positive fluid balance and elevated central venous pressure are associated with increased mortality". Crit Care Med. 39 (2): 259–65. doi:10.1097/CCM.0b013e3181feeb15. PMID 20975548.
- ↑ Wang N, Jiang L, Zhu B, Wen Y, Xi XM, Beijing Acute Kidney Injury Trial (BAKIT) Workgroup (2015). "Fluid balance and mortality in critically ill patients with acute kidney injury: a multicenter prospective epidemiological study". Crit Care. 19: 371. doi:10.1186/s13054-015-1085-4. PMC 4619072. PMID 26494153.
- ↑ Cronhjort M, Hjortrup PB, Holst LB, Joelsson-Alm E, Mårtensson J, Svensen C; et al. (2016). "Association between fluid balance and mortality in patients with septic shock: a post hoc analysis of the TRISS trial". Acta Anaesthesiol Scand. 60 (7): 925–33. doi:10.1111/aas.12723. PMID 27030514.
- ↑ Benes J (2016). "Cumulative Fluid Balance: The Dark Side of the Fluid". Crit Care Med. 44 (10): 1945–6. doi:10.1097/CCM.0000000000001919. PMID 27635489.
- ↑ James JH, Luchette FA, McCarter FD, Fischer JE (1999). "is an unreliable indicator of tissue hypoxia in injury or sepsis". Lancet. 354 (9177): 505–8. doi:10.1016/S0140-6736(98)91132-1. PMID 10465191.
- ↑ 46.0 46.1 46.2 Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R; et al. (2017). "Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016". Crit Care Med. 45 (3): 486–552. doi:10.1097/CCM.0000000000002255. PMID 28098591.
- ↑ 47.0 47.1 Jones AE, Shapiro NI, Trzeciak S, Arnold RC, Claremont HA, Kline JA; et al. (2010). "Lactate clearance vs central venous oxygen saturation as goals of early sepsis therapy: a randomized clinical trial". JAMA. 303 (8): 739–46. doi:10.1001/jama.2010.158. PMC 2918907. PMID 20179283.
- ↑ 48.0 48.1 Holst LB, Haase N, Wetterslev J, Wernerman J, Guttormsen AB, Karlsson S; et al. (2014). "Lower versus higher hemoglobin threshold for transfusion in septic shock". N Engl J Med. 371 (15): 1381–91. doi:10.1056/NEJMoa1406617. PMID 25270275.
- ↑ Jansen TC, van Bommel J, Schoonderbeek FJ, Sleeswijk Visser SJ, van der Klooster JM, Lima AP; et al. (2010). "Early lactate-guided therapy in intensive care unit patients: a multicenter, open-label, randomized controlled trial". Am J Respir Crit Care Med. 182 (6): 752–61. doi:10.1164/rccm.200912-1918OC. PMID 20463176.
- ↑ Gu WJ, Zhang Z, Bakker J (2015). "Early lactate clearance-guided therapy in patients with sepsis: a meta-analysis with trial sequential analysis of randomized controlled trials". Intensive Care Med. 41 (10): 1862–3. doi:10.1007/s00134-015-3955-2. PMID 26154408.
- ↑ Teboul JL, Saugel B, Cecconi M, De Backer D, Hofer CK, Monnet X; et al. (2016). "Less invasive hemodynamic monitoring in critically ill patients". Intensive Care Med. 42 (9): 1350–9. doi:10.1007/s00134-016-4375-7. PMID 27155605.
- ↑ Stens J, Oeben J, Van Dusseldorp AA, Boer C (2016). "Non-invasive measurements of pulse pressure variation and stroke volume variation in anesthetized patients using the Nexfin blood pressure monitor". J Clin Monit Comput. 30 (5): 587–94. doi:10.1007/s10877-015-9759-7. PMC 5023739. PMID 26318314.
- ↑ Bentzer P, Griesdale DE, Boyd J, MacLean K, Sirounis D, Ayas NT (2016). "Will This Hemodynamically Unstable Patient Respond to a Bolus of Intravenous Fluids?". JAMA. 316 (12): 1298–309. doi:10.1001/jama.2016.12310. PMID 27673307.
- ↑ Porhomayon J, Zadeii G, Congello S, Nader ND (2012). "Applications of minimally invasive cardiac output monitors". Int J Emerg Med. 5: 18. doi:10.1186/1865-1380-5-18. PMC 3353182. PMID 22531454.
- ↑ Seymour, Christopher W.; Gesten, Foster; Prescott, Hallie C.; Friedrich, Marcus E.; Iwashyna, Theodore J.; Phillips, Gary S.; Lemeshow, Stanley; Osborn, Tiffany; Terry, Kathleen M.; Levy, Mitchell M. (2017). "Time to Treatment and Mortality during Mandated Emergency Care for Sepsis". New England Journal of Medicine. doi:10.1056/NEJMoa1703058. ISSN 0028-4793.
- ↑ Rhee C, Chiotos K, Cosgrove SE, Heil EL, Kadri SS, Kalil AC; et al. (2020). "Infectious Diseases Society of America Position Paper: Recommended Revisions to the National Severe Sepsis and Septic Shock Early Management Bundle (SEP-1) Sepsis Quality Measure". Clin Infect Dis. doi:10.1093/cid/ciaa059. PMID 32374861 Check
|pmid=value (help). - ↑ Sherwin R, Winters ME, Vilke GM, Wardi G (2017). "Does Early and Appropriate Antibiotic Administration Improve Mortality in Emergency Department Patients with Severe Sepsis or Septic Shock?". J Emerg Med. doi:10.1016/j.jemermed.2016.12.009. PMID 28916120.
- ↑ Liu VX, Fielding-Singh V, Greene JD, Baker JM, Iwashyna TJ, Bhattacharya J; et al. (2017). "The Timing of Early Antibiotics and Hospital Mortality in Sepsis". Am J Respir Crit Care Med. 196 (7): 856–863. doi:10.1164/rccm.201609-1848OC. PMC 5649973. PMID 28345952.
- ↑ Pruinelli L, Westra BL, Yadav P, et al.Delay Within the 3-Hour Surviving Sepsis Campaign Guideline on Mortality for Patients With Severe Sepsis and Septic Shock*. Critical Care Medicine. 2018;46(4):500–505. doi: 10.1097/CCM.0000000000002949.
- ↑ Whiles BB, Deis AS, Simpson SQ (2017). "Increased Time to Initial Antimicrobial Administration Is Associated With Progression to Septic Shock in Severe Sepsis Patients". Crit Care Med. doi:10.1097/CCM.0000000000002262. PMID 28169944.
- ↑ Meyer NJ, Prescott HC (December 2024). "Sepsis and Septic Shock". N Engl J Med. 391 (22): 2133–2146. doi:10.1056/NEJMra2403213. PMID 39774315 Check
|pmid=value (help). - ↑ Scheer CS, Fuchs C, Gründling M, Vollmer M, Bast J, Bohnert JA; et al. (2019). "Impact of antibiotic administration on blood culture positivity at the beginning of sepsis: a prospective clinical cohort study". Clin Microbiol Infect. 25 (3): 326–331. doi:10.1016/j.cmi.2018.05.016. PMID 29879482.
- ↑ 63.0 63.1 63.2 Cheng MP, Stenstrom R, Paquette K, Stabler SN, Akhter M, Davidson AC; et al. (2019). "Blood Culture Results Before and After Antimicrobial Administration in Patients With Severe Manifestations of Sepsis: A Diagnostic Study". Ann Intern Med. 171 (8): 547–554. doi:10.7326/M19-1696. PMID 31525774.
- ↑ Pepper DJ, Sun J, Cui X, Welsh J, Natanson C, Eichacker PQ (2019). "Antibiotic- and Fluid-Focused Bundles Potentially Improve Sepsis Management, but High-Quality Evidence Is Lacking for the Specificity Required in the Centers for Medicare and Medicaid Service's Sepsis Bundle (SEP-1)". Crit Care Med. 47 (10): 1290–1300. doi:10.1097/CCM.0000000000003892. PMID 31369426.
- ↑ Ferreras Amez JM, Arribas Entrala B, Sarrat Torres MA, García Noain A, Caudevilla Martínez A, Colás Oros C; et al. (2017). "[Before-after study of the effect of implementing a sepsis code for emergency departments in the community of Aragon]". Emergencias. 29 (3): 154–160. PMID 28825234.
- ↑ Haynes B (1999). "Can it work? Does it work? Is it worth it? The testing of healthcareinterventions is evolving". BMJ. 319 (7211): 652–3. doi:10.1136/bmj.319.7211.652. PMC 1116525. PMID 10480802.
- ↑ Sterling SA, Miller WR, Pryor J, Puskarich MA, Jones AE (2015). "The Impact of Timing of Antibiotics on Outcomes in Severe Sepsis and Septic Shock: A Systematic Review and Meta-Analysis". Crit Care Med. 43 (9): 1907–15. doi:10.1097/CCM.0000000000001142. PMC 4597314. PMID 26121073. Review in: Evid Based Med. 2015 Dec;20(6):214-5
- ↑ Identifying and measuring heterogeneity. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from http://www.cochrane-handbook.org
- ↑ Kumar A (2016). "Systematic Bias in Meta-Analyses of Time to Antimicrobial in Sepsis Studies". Crit Care Med. 44 (4): e234–5. doi:10.1097/CCM.0000000000001512. PMID 26974458.
- ↑ 70.0 70.1 Ferrer R, Martin-Loeches I, Phillips G, Osborn TM, Townsend S, Dellinger RP; et al. (2014). "Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: results from a guideline-based performance improvement program". Crit Care Med. 42 (8): 1749–55. doi:10.1097/CCM.0000000000000330. PMID 24717459.
- ↑ Shirakura Y, Kuriyama A (2016). "Timing of Antibiotic Administration in Sepsis and Septic Shock: The Impact That a Meta-Analysis Does Not Depict". Crit Care Med. 44 (10): e1004. doi:10.1097/CCM.0000000000001891. PMID 27635498.
- ↑ Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S; et al. (2006). "Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock". Crit Care Med. 34 (6): 1589–96. doi:10.1097/01.CCM.0000217961.75225.E9. PMID 16625125.
- ↑ Sarani B, Brenner SR, Gabel B, Myers JS, Gibson G, Phillips J; et al. (2008). "Improving sepsis care through systems change: the impact of a medical emergency team". Jt Comm J Qual Patient Saf. 34 (3): 179–82, 125. PMID 18419048.
- ↑ 74.0 74.1 74.2 74.3 Flaherty SK, Weber RL, Chase M, Dugas AF, Graver AM, Salciccioli JD; et al. (2014). "Septic shock and adequacy of early empiric antibiotics in the emergency department". J Emerg Med. 47 (5): 601–7. doi:10.1016/j.jemermed.2014.06.037. PMC 4254037. PMID 25218723.
- ↑ Garnacho-Montero J, Gutiérrez-Pizarraya A, Escoresca-Ortega A, Fernández-Delgado E, López-Sánchez JM (2015). "Adequate antibiotic therapy prior to ICU admission in patients with severe sepsis and septic shock reduces hospital mortality". Crit Care. 19: 302. doi:10.1186/s13054-015-1000-z. PMC 4549859. PMID 26307060.
- ↑ 76.0 76.1 Lee CC, Lee CH, Chuang MC, Hong MY, Hsu HC, Ko WC (2012). "Impact of inappropriate empirical antibiotic therapy on outcome of bacteremic adults visiting the ED". Am J Emerg Med. 30 (8): 1447–56. doi:10.1016/j.ajem.2011.11.010. PMID 22205015.
- ↑ 77.0 77.1 Nygård ST, Langeland N, Flaatten HK, Fanebust R, Haugen O, Skrede S (2014). "Aetiology, antimicrobial therapy and outcome of patients with community acquired severe sepsis: a prospective study in a Norwegian university hospital". BMC Infect Dis. 14: 121. doi:10.1186/1471-2334-14-121. PMC 3975934. PMID 24588984.
- ↑ 78.0 78.1 Capp R, Chang Y, Brown DF (2011). "Effective antibiotic treatment prescribed by emergency physicians in patients admitted to the intensive care unit with severe sepsis or septic shock: where is the gap?". J Emerg Med. 41 (6): 573–80. doi:10.1016/j.jemermed.2010.10.024. PMID 21371846.
- ↑ Kumar A, Ellis P, Arabi Y, Roberts D, Light B, Parrillo JE; et al. (2009). "Initiation of inappropriate antimicrobial therapy results in a fivefold reduction of survival in human septic shock". Chest. 136 (5): 1237–1248. doi:10.1378/chest.09-0087. PMID 19696123.
- ↑ van der Velden LB, Tromp M, Bleeker-Rovers CP, Hulscher M, Kullberg BJ, Mouton JW; et al. (2012). "Non-adherence to antimicrobial treatment guidelines results in more broad-spectrum but not more appropriate therapy". Eur J Clin Microbiol Infect Dis. 31 (7): 1561–8. doi:10.1007/s10096-011-1478-5. PMC 3364419. PMID 22108843.
- ↑ Roberts JA, Abdul-Aziz MH, Davis JS, Dulhunty JM, Cotta MO, Myburgh J; et al. (2016). "Continuous versus Intermittent β-Lactam Infusion in Severe Sepsis. A Meta-analysis of Individual Patient Data from Randomized Trials". Am J Respir Crit Care Med. 194 (6): 681–91. doi:10.1164/rccm.201601-0024OC. PMID 26974879.
- ↑ Levy B, Perez P, Perny J, Thivilier C, Gerard A (2011). "Comparison of -dobutamine to epinephrine for hemodynamics, metabolism, and organ function variables in cardiogenic shock. A prospective, randomized pilot study". Crit Care Med. 39 (3): 450–5. doi:10.1097/CCM.0b013e3181ffe0eb. PMID 21037469.
- ↑ 83.0 83.1 De Backer D, Biston P, Devriendt J, Madl C, Chochrad D, Aldecoa C; et al. (2010). "Comparison of dopamine and in the treatment of shock". N Engl J Med. 362 (9): 779–89. doi:10.1056/NEJMoa0907118. PMID 20200382.
- ↑ Asfar P, Meziani F, Hamel JF, Grelon F, Megarbane B, Anguel N; et al. (2014). "High versus low blood-pressure target in patients with septic shock". N Engl J Med. 370 (17): 1583–93. doi:10.1056/NEJMoa1312173. PMID 24635770. Review in: Ann Intern Med. 2014 Jul 15;161(2):JC7
- ↑ Meyer NJ, Prescott HC (December 2024). "Sepsis and Septic Shock". N Engl J Med. 391 (22): 2133–2146. doi:10.1056/NEJMra2403213. PMID 39774315 Check
|pmid=value (help). - ↑ Subramanian S, Yilmaz M, Rehman A, Hubmayr RD, Afessa B, Gajic O (2008). "Liberal vs. conservative vasopressor use to maintain mean arterial blood pressure during resuscitation of septic shock: an observational study". Intensive Care Med. 34 (1): 157–62. doi:10.1007/s00134-007-0862-1. PMID 17924093.
- ↑ Duran BA, Badgett RG, Simpson SQ (2018). "Hydrocortisone plus fludrocortisone reduced mortality at 90 days in patients with septic shock". Ann Intern Med. 168 (12): JC68. doi:10.7326/ACPJC-2018-168-12-068. PMID 29913493.
- ↑ 88.0 88.1 Annane D, Renault A, Brun-Buisson C, Megarbane B, Quenot JP, Siami S; et al. (2018). "Hydrocortisone plus Fludrocortisone for Adults with Septic Shock". N Engl J Med. 378 (9): 809–818. doi:10.1056/NEJMoa1705716. PMID 29490185.
- ↑ Venkatesh B, Finfer S, Cohen J, Rajbhandari D, Arabi Y, Bellomo R; et al. (2018). "Adjunctive Glucocorticoid Therapy in Patients with Septic Shock". N Engl J Med. 378 (9): 797–808. doi:10.1056/NEJMoa1705835. PMID 29347874.
- ↑ Annane D, Bellissant E, Bollaert PE, Briegel J, Keh D, Kupfer Y (2015). "Corticosteroids for treating sepsis". Cochrane Database Syst Rev (12): CD002243. doi:10.1002/14651858.CD002243.pub3. PMID 26633262.
- ↑ Volbeda M, Wetterslev J, Gluud C, Zijlstra JG, van der Horst IC, Keus F (2015). "Glucocorticosteroids for sepsis: systematic review with meta-analysis and trial sequential analysis". Intensive Care Med. 41 (7): 1220–34. doi:10.1007/s00134-015-3899-6. PMC 4483251. PMID 26100123.
- ↑ Keh D, Trips E, Marx G, Wirtz SP, Abduljawwad E, Bercker S; et al. (2016). "Effect of Hydrocortisone on Development of Shock Among Patients With Severe Sepsis: The HYPRESS Randomized Clinical Trial". JAMA. 316 (17): 1775–1785. doi:10.1001/jama.2016.14799. PMID 27695824.
- ↑ Meyer NJ, Prescott HC (December 2024). "Sepsis and Septic Shock". N Engl J Med. 391 (22): 2133–2146. doi:10.1056/NEJMra2403213. PMID 39774315 Check
|pmid=value (help). - ↑ Casserly B, Gerlach H, Phillips GS, Lemeshow S, Marshall JC, Osborn TM; et al. (2012). "Low-dose steroids in adult septic shock: results of the Surviving Sepsis Campaign". Intensive Care Med. 38 (12): 1946–54. doi:10.1007/s00134-012-2720-z. PMID 23064466.
- ↑ Antcliffe DB, Burnham KL, Al-Beidh F, Santhakumaran S, Brett SJ, Hinds CJ; et al. (2018). "Transcriptomic Signatures in Sepsis and a Differential Response to Steroids: From the VANISH Randomized Trial". Am J Respir Crit Care Med. doi:10.1164/rccm.201807-1419OC. PMID 30365341.
- ↑ Venkatesh B, Cohen J (2019). "The authors reply". Crit Care Med. 47 (12): e1035–e1036. doi:10.1097/CCM.0000000000004014. PMID 31738260.
- ↑ COIITSS Study Investigators. Annane D, Cariou A, Maxime V, Azoulay E, D'honneur G; et al. (2010). "Corticosteroid treatment and intensive insulin therapy for septic shock in adults: a randomized controlled trial". JAMA. 303 (4): 341–8. doi:10.1001/jama.2010.2. PMID 20103758. Review in: Ann Intern Med. 2010 May 18;152(10):JC5-5
- ↑ 98.0 98.1 Cannon CM, Holthaus CV, Zubrow MT, Posa P, Gunaga S, Kella V; et al. (2012). "The GENESIS Project (GENeralized Early Sepsis Intervention Strategies): A Multicenter Quality Improvement Collaborative". J Intensive Care Med. doi:10.1177/0885066612453025. PMID 22902347.
- ↑ Levy MM, Evans LE, Rhodes A (2018). "The Surviving Sepsis Campaign Bundle: 2018 update". Intensive Care Med. 44 (6): 925–928. doi:10.1007/s00134-018-5085-0. PMID 29675566.
- ↑ Adult Patients. Available at https://www.sccm.org/SurvivingSepsisCampaign/Guidelines/Adult-Patients
- ↑ Hayden GE, Tuuri RE, Scott R, Losek JD, Blackshaw AM, Schoenling AJ; et al. (2016). "Triage sepsis alert and sepsis protocol lower times to fluids and antibiotics in the ED". Am J Emerg Med. 34 (1): 1–9. doi:10.1016/j.ajem.2015.08.039. PMC 4905767. PMID 26386734.
index.php?title=Category:Causes of death index.php?title=Category:Disease index.php?title=Category:Emergency medicine index.php?title=Category:Intensive care medicine index.php?title=Category:Infectious Disease Project index.php?title=Category:Emergency mdicine index.php?title=Category:Up-To-Date index.php?title=Category:Infectious disease
 KSF
KSF