Human Genome Project
 From Wikipedia - Reading time: 33 min
From Wikipedia - Reading time: 33 min
| Human Genome Project | |
|---|---|
| HGP | |
 Logo of the Human Genome Project | |
| Project type | International scientific research project |
| Funding agency | U.S. National Institutes of Health (NIH) and others |
| Objective | Mapping and sequencing the human genome |
| Location | Primarily in the United States, the United Kingdom, Japan, France, Germany, and China |
| Participants | At least 20 institutions, companies, and laboratories |
| Duration | 1990 – 2003 |
The Human Genome Project (HGP) was an international scientific research project with the goal of determining the base pairs that make up human DNA, and of identifying, mapping and sequencing all of the genes of the human genome from both a physical and a functional standpoint. It started in 1990 and was completed in 2003.[1] It was the world's largest collaborative biological project.[2] Planning for the project began in 1984 by the US government, and it officially launched in 1990. It was declared complete on 14 April 2003, and included about 92% of the genome.[3] Level "complete genome" was achieved in May 2021, with only 0.3% of the bases covered by potential issues.[4][5] The final gapless assembly was finished in January 2022.[6]
Funding came from the US government through the National Institutes of Health (NIH) as well as numerous other groups from around the world. A parallel project was conducted outside the government by the Celera Corporation, or Celera Genomics, which was formally launched in 1998. Most of the government-sponsored sequencing was performed in twenty universities and research centres in the United States, the United Kingdom, Japan, France, Germany, and China,[7] working in the International Human Genome Sequencing Consortium (IHGSC).
The Human Genome Project originally aimed to map the complete set of nucleotides contained in a human haploid reference genome, of which there are more than three billion. The genome of any given individual is unique; mapping the human genome involved sequencing samples collected from a small number of individuals and then assembling the sequenced fragments to get a complete sequence for each of the 23 human chromosome pairs (22 pairs of autosomes and a pair of sex chromosomes, known as allosomes). Therefore, the finished human genome is a mosaic, not representing any one individual. Much of the project's utility comes from the fact that the vast majority of the human genome is the same in all humans.
History
[edit]The Human Genome Project was a 13-year-long publicly funded project initiated in 1990 with the objective of determining the DNA sequence of the entire euchromatic human genome within 13 years.[8][9] The idea that sets of inherited genes predicted the concept of mapping a disease gene to a chromosomal region originated in the work of Ronald A. Fisher, whose work is also credited with later initiating the project.[10][11] In 1977, Walter Gilbert, Frederick Sanger, and Paul Berg invented these methods of sequencing DNA.[12][13]
In May 1985, Robert Sinsheimer organized a workshop at the University of California, Santa Cruz, to discuss the feasibility of building a systematic reference genome using gene sequencing technologies.[14] Gilbert wrote the first plan for what he called The Human Genome Institute on the plane ride home from the workshop.[15] In March 1986, the Santa Fe Workshop was organized by Charles DeLisi and David Smith of the Department of Energy's Office of Health and Environmental Research (OHER).[16] At the same time Renato Dulbecco, President of the Salk Institute for Biological Studies, first proposed the concept of whole genome sequencing in an essay in Science.[17] The published work, titled "A Turning Point in Cancer Research: Sequencing the Human Genome", was shortened from the original proposal of using the sequence to understand the genetic basis of breast cancer.[18] James Watson, one of the discoverers of the double helix shape of DNA in the 1950s, followed two months later with a workshop held at the Cold Spring Harbor Laboratory. Thus the idea for obtaining a reference sequence had three independent origins: Sinsheimer, Dulbecco and DeLisi. Ultimately it was the actions by DeLisi that launched the project.[19][20][21][22]
The fact that the Santa Fe Workshop was motivated and supported by a federal agency opened a path, albeit a difficult and tortuous one,[23] for converting the idea into public policy in the United States. In a memo to the Assistant Secretary for Energy Research Alvin Trivelpiece, then-Director of the OHER Charles DeLisi outlined a broad plan for the project.[24] This started a long and complex chain of events that led to the approved reprogramming of funds that enabled the OHER to launch the project in 1986, and to recommend the first line item for the HGP, which was in President Reagan's 1988 budget submission,[23] and ultimately approved by Congress. Of particular importance in congressional approval was the advocacy of New Mexico Senator Pete Domenici, whom DeLisi had befriended.[25] Domenici chaired the Senate Committee on Energy and Natural Resources, as well as the Budget Committee, both of which were key in the DOE budget process. Congress added a comparable amount to the NIH budget, thereby beginning official funding by both agencies.[citation needed]
Trivelpiece sought and obtained the approval of DeLisi's proposal from Deputy Secretary William Flynn Martin. This chart[26] was used by Trivelpiece in the spring of 1986 to brief Martin and Under Secretary Joseph Salgado regarding his intention to reprogram $4 million to initiate the project with the approval of John S. Herrington.[citation needed] This reprogramming was followed by a line item budget of $13 million in the Reagan administration's 1987 budget submission to Congress.[16] It subsequently passed both Houses. The project was planned to be completed within 15 years.[27]
In 1990 the two major funding agencies, DOE and the National Institutes of Health, developed a memorandum of understanding to coordinate plans and set the clock for the initiation of the Project to 1990.[28] At that time, David J. Galas was Director of the renamed "Office of Biological and Environmental Research" in the US Department of Energy's Office of Science and James Watson headed the NIH Genome Program. In 1993, Aristides Patrinos succeeded Galas and Francis Collins succeeded Watson, assuming the role of overall Project Head as Director of the NIH National Center for Human Genome Research (which would later become the National Human Genome Research Institute). A working draft of the genome was announced in 2000 and the papers describing it were published in February 2001. A more complete draft was published in 2003, and genome "finishing" work continued for more than a decade after that.[citation needed]
The $3 billion project was formally founded in 1990 by the US Department of Energy and the National Institutes of Health, and was expected to take 15 years.[29] In addition to the United States, the international consortium comprised geneticists in the United Kingdom, France, Australia, China, and a myriad of other spontaneous relationships.[30] The project ended up costing less than expected, at about $2.7 billion (equivalent to about $5 billion in 2021).[7][31][32] Most of the genome was mapped over a two-year span.[33]
Two technologies enabled the project: gene mapping and DNA sequencing. The gene mapping technique of restriction fragment length polymorphism (RFLP) arose from the search for the location of the breast cancer gene by Mark Skolnick of the University of Utah,[34] which began in 1974.[35] Seeing a linkage marker for the gene, in collaboration with David Botstein, Ray White and Ron Davis conceived of a way to construct a genetic linkage map of the human genome. This enabled scientists to launch the larger human genome effort.[36]
Because of widespread international cooperation and advances in the field of genomics (especially in sequence analysis), as well as parallel advances in computing technology, a 'rough draft' of the genome was finished in 2000 (announced jointly by US President Bill Clinton and British Prime Minister Tony Blair on 26 June 2000).[37][38] This first available rough draft assembly of the genome was completed by the Genome Bioinformatics Group at the University of California, Santa Cruz, primarily led by then-graduate student Jim Kent and his advisor David Haussler.[39] Ongoing sequencing led to the announcement of the essentially complete genome on 14 April 2003, two years earlier than planned.[40][41] In May 2006, another milestone was passed on the way to completion of the project when the sequence of the very last chromosome was published in Nature.[42]
The various institutions, companies, and laboratories which participated in the Human Genome Project are listed below, according to the NIH:[7]
| No. | Nation | Name | Affiliation |
|---|---|---|---|
| 1 | United States | The Whitehead Institute/MIT Center for Genome Research | Massachusetts Institute of Technology |
| 2 | United Kingdom | The Wellcome Trust Sanger Institute | Wellcome Trust |
| 3 | United States | Washington University School of Medicine Genome Sequencing Center | Washington University in St. Louis |
| 4 | United States | United States DOE Joint Genome Institute | United States Department of Energy |
| 5 | United States | Baylor College of Medicine Human Genome Sequencing Center | Baylor College of Medicine |
| 6 | Japan | RIKEN Genomic Sciences Center | Riken |
| 7 | France | Genoscope and CNRS UMR-8030 | French Alternative Energies and Atomic Energy Commission |
| 8 | United States | GTC Sequencing Center | Genome Therapeutics Corporation, whose sequencing division is acquired by ABI |
| 9 | Germany | Department of Genome Analysis | Fritz Lipmann Institute, name changed from Institute of Molecular Biotechnology |
| 10 | China | Beijing Genomics Institute/Human Genome Center | Chinese Academy of Sciences |
| 11 | United States | Multimegabase Sequencing Center | Institute for Systems Biology |
| 12 | United States | Stanford Genome Technology Center | Stanford University |
| 13 | United States | Stanford Human Genome Center and Department of Genetics | Stanford University School of Medicine |
| 14 | United States | University of Washington Genome Center | University of Washington |
| 15 | Japan | Department of Molecular Biology | Keio University School of Medicine |
| 16 | United States | University of Texas Southwestern Medical Center at Dallas | University of Texas |
| 17 | United States | University of Oklahoma's Advanced Center for Genome Technology | Dept. of Chemistry and Biochemistry, University of Oklahoma |
| 18 | Germany | Max Planck Institute for Molecular Genetics | Max Planck Society |
| 19 | United States | Lita Annenberg Hazen Genome Center | Cold Spring Harbor Laboratory |
| 20 | Germany | GBF/German Research Centre for Biotechnology | Reorganized and renamed to Helmholtz Centre for Infection Research |
State of completion
[edit]Notably the project was not able to sequence all of the DNA found in human cells; rather, the aim was to sequence only euchromatic regions of the nuclear genome, which make up 92.1% of the human genome. The remaining 7.9% exists in scattered heterochromatic regions such as those found in centromeres and telomeres. These regions by their nature are generally more difficult to sequence and so were not included as part of the project's original plans.[43]
The Human Genome Project (HGP) was declared complete in April 2003. An initial rough draft of the human genome was available in June 2000 and by February 2001 a working draft had been completed and published followed by the final sequencing mapping of the human genome on 14 April 2003. Although this was reported to cover 99% of the euchromatic human genome with 99.99% accuracy, a major quality assessment of the human genome sequence was published on 27 May 2004, indicating over 92% of sampling exceeded 99.99% accuracy which was within the intended goal.[44]
In March 2009, the Genome Reference Consortium (GRC) released a more accurate version of the human genome, but that still left more than 300 gaps,[45] while 160 such gaps remained in 2015.[46]
Though in May 2020 the GRC reported 79 "unresolved" gaps,[47] accounting for as much as 5% of the human genome,[48] months later, the application of new long-range sequencing techniques and a hydatidiform mole-derived cell line in which both copies of each chromosome are identical led to the first telomere-to-telomere, truly complete sequence of a human chromosome, the X chromosome.[49] Similarly, an end-to-end complete sequence of human autosomal chromosome 8 followed several months later.[50]
In April 2022, the Telomere-to-Telomere (T2T) consortium published a complete sequence of the non-Y chromosomes, highlighting the 8% of the human genome that the HGP had not sequenced.[51][52][53][54] The T2T consortium then used this newly completed genome sequence[55] as a reference to identify over 2 million additional genomic variants.[56] In August 2023, Rhie et al. reported the successful sequencing of the previously missing regions of the Y chromosome, achieving the full sequencing of all 24 human chromosomes.[57][58]
Applications and proposed benefits
[edit]The sequencing of the human genome holds benefits for many fields, from molecular medicine to human evolution. The Human Genome Project, through its sequencing of the DNA, can help researchers understand diseases including: genotyping of specific viruses to direct appropriate treatment; identification of mutations linked to different forms of cancer; the design of medication and more accurate prediction of their effects; advancement in forensic applied sciences; biofuels and other energy applications; agriculture, animal husbandry, bioprocessing; risk assessment; bioarcheology, anthropology and evolution. The sequence of the DNA is stored in databases available to anyone on the Internet. The US National Center for Biotechnology Information (and sister organizations in Europe and Japan) house the gene sequence in a database known as GenBank, along with sequences of known and hypothetical genes and proteins. Other organizations, such as the UCSC Genome Browser at the University of California, Santa Cruz,[59] and Ensembl[60] present additional data and annotation and powerful tools for visualizing and searching it. Computer programs have been developed to analyze the data because the data itself is difficult to interpret without such programs. Generally speaking, advances in genome sequencing technology have followed Moore's Law, a concept from computer science which states that integrated circuits can increase in complexity at an exponential rate.[61] This means that the speeds at which whole genomes can be sequenced can increase at a similar rate, as was seen during the development of the Human Genome Project. By 2023, the speed record for sequencing a genome was around five hours; more often, however, it takes weeks.[33]
Techniques and analysis
[edit]The process of identifying the boundaries between genes and other features in a raw DNA sequence is called genome annotation and is in the domain of bioinformatics. While expert biologists make the best annotators, their work proceeds slowly, and computer programs are increasingly used to meet the high-throughput demands of genome sequencing projects. Beginning in 2008, a new technology known as RNA-seq was introduced that allowed scientists to directly sequence the messenger RNA in cells. This replaced previous methods of annotation, which relied on the inherent properties of the DNA sequence, with direct measurement, which was much more accurate. Today, annotation of the human genome and other genomes relies primarily on deep sequencing of the transcripts in every human tissue using RNA-seq. These experiments have revealed that over 90% of genes contain at least one and usually several alternative splice variants, in which the exons are combined in different ways to produce 2 or more gene products from the same locus.[62]
The genome published by the HGP does not represent the sequence of every individual's genome. It is the combined mosaic of a small number of anonymous donors, of African, European, and East Asian ancestry. The HGP genome is a scaffold for future work in identifying differences among individuals.[citation needed] Subsequent projects sequenced the genomes of multiple distinct ethnic groups, though as of 2019 there is still only one "reference genome".[63]
Findings
[edit]Key findings of the draft (2001) and complete (2004) genome sequences include:
- There are approximately 22,300[64] protein-coding genes in human beings, the same range as in other mammals.
- The human genome has significantly more segmental duplications (nearly identical, repeated sections of DNA) than had been previously suspected.[65][66][67]
- At the time when the draft sequence was published, fewer than 7% of protein families appeared to be vertebrate specific.[68]
Accomplishments
[edit]
The human genome has approximately 3.1 billion base pairs.[69] The Human Genome Project was started in 1990 with the goal of sequencing and identifying all base pairs in the human genetic instruction set, finding the genetic roots of disease and then developing treatments. It is considered a megaproject.
The genome was broken into smaller pieces; approximately 150,000 base pairs in length.[70] These pieces were then ligated into a type of vector known as "bacterial artificial chromosomes", or BACs, which are derived from bacterial chromosomes which have been genetically engineered. The vectors containing the genes can be inserted into bacteria where they are copied by the bacterial DNA replication machinery. Each of these pieces was then sequenced separately as a small "shotgun" project and then assembled. The larger, 150,000 base pairs go together to create chromosomes. This is known as the "hierarchical shotgun" approach, because the genome is first broken into relatively large chunks, which are then mapped to chromosomes before being selected for sequencing.[71][72]
Funding came from the US government through the National Institutes of Health in the United States, and a UK charity organization, the Wellcome Trust, as well as numerous other groups from around the world. The funding supported a number of large sequencing centers including those at Whitehead Institute, the Wellcome Sanger Institute (then called The Sanger Centre) based at the Wellcome Genome Campus, Washington University in St. Louis, and Baylor College of Medicine.[29][73]
The UN Educational, Scientific and Cultural Organization (UNESCO) served as an important channel for the involvement of developing countries in the Human Genome Project.[74]
Public versus private approaches
[edit]In 1998 a similar, privately funded quest was launched by the American researcher Craig Venter, and his firm Celera Genomics. Venter was a scientist at the NIH during the early 1990s when the project was initiated. The $300 million Celera effort was intended to proceed at a faster pace and at a fraction of the cost of the roughly $3 billion publicly funded project. While the Celera project focused its efforts on production sequencing and assembly of the human genome, the public HGP also funded mapping and sequencing of the worm, fly, and yeast genomes, funding of databases, development of new technologies, supporting bioinformatics and ethics programs, as well as polishing and assessment of the genome assembly.[75] Both the Celera and public approaches spent roughly $250 million on the production sequencing effort.[76] For sequence assembly, Celera made use of publicly available maps at GenBank, which Celera was capable of generating, but the availability of which was "beneficial" to the privately-funded project.[65]
Celera used a technique called whole genome shotgun sequencing, employing pairwise end sequencing,[77] which had been used to sequence bacterial genomes of up to six million base pairs in length, but not for anything nearly as large as the three billion base pair human genome.
Celera initially announced that it would seek patent protection on "only 200–300" genes, but later amended this to seeking "intellectual property protection" on "fully-characterized important structures" amounting to 100–300 targets. The firm eventually filed preliminary ("place-holder") patent applications on 6,500 whole or partial genes. Celera also promised to publish their findings in accordance with the terms of the 1996 "Bermuda Statement", by releasing new data annually (the HGP released its new data daily), although, unlike the publicly funded project, they would not permit free redistribution or scientific use of the data. The publicly funded competitors were compelled to release the first draft of the human genome before Celera for this reason. On 7 July 2000, the UCSC Genome Bioinformatics Group released the first working draft on the web. The scientific community downloaded about 500 GB of information from the UCSC genome server in the first 24 hours of free and unrestricted access.[78]
In March 2000 President Clinton, along with Prime Minister Tony Blair in a dual statement, urged that all researchers who wished to research the sequence should have "unencumbered access" to the genome sequence.[79] The statement sent Celera's stock plummeting and dragged down the biotechnology-heavy Nasdaq. The biotechnology sector lost about $50 billion in market capitalization in two days.[citation needed]
Although the working draft was announced in June 2000, it was not until February 2001 that Celera and the HGP scientists published details of their drafts. Special issues of Nature (which published the publicly funded project's scientific paper)[65] described the methods used to produce the draft sequence and offered analysis of the sequence. These drafts covered about 83% of the genome (90% of the euchromatic regions with 150,000 gaps and the order and orientation of many segments not yet established). In February 2001, at the time of the joint publications, press releases announced that the project had been completed by both groups. Improved drafts were announced in 2003 and 2005, filling in to approximately 92% of the sequence currently.[citation needed]
Genome donors
[edit]In the International Human Genome Sequencing Consortium (IHGSC) public-sector HGP, researchers collected blood (female) or sperm (male) samples from a large number of donors. Only a few of many collected samples were processed as DNA resources. Thus the donor identities were protected so neither donors nor scientists could know whose DNA was sequenced. DNA clones taken from many different libraries were used in the overall project, with most of those libraries being created by Pieter J. de Jong. Much of the sequence (>70%) of the reference genome produced by the public HGP came from a single anonymous male donor from Buffalo, New York, (code name RP11; the "RP" refers to Roswell Park Comprehensive Cancer Center).[80][81]
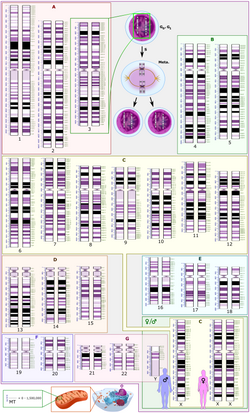
HGP scientists used white blood cells from the blood of two male and two female donors (randomly selected from 20 of each) – each donor yielding a separate DNA library. One of these libraries (RP11) was used considerably more than others, because of quality considerations. One minor technical issue is that male samples contain just over half as much DNA from the sex chromosomes (one X chromosome and one Y chromosome) compared to female samples (which contain two X chromosomes). The other 22 chromosomes (the autosomes) are the same for both sexes.
Although the main sequencing phase of the HGP has been completed, studies of DNA variation continued in the International HapMap Project, whose goal was to identify patterns of single-nucleotide polymorphism (SNP) groups (called haplotypes, or "haps"). The DNA samples for the HapMap came from a total of 270 individuals; Yoruba people in Ibadan, Nigeria; Japanese people in Tokyo; Han Chinese in Beijing; and the French Centre d'Etude du Polymorphisme Humain (CEPH) resource, which consisted of residents of the United States having ancestry from Western and Northern Europe.
In the Celera Genomics private-sector project, DNA from five different individuals was used for sequencing. The lead scientist of Celera Genomics at that time, Craig Venter, later acknowledged (in a public letter to the journal Science) that his DNA was one of 21 samples in the pool, five of which were selected for use.[82][83]
Developments
[edit]With the sequence in hand the next step was to identify the genetic variants that increase the risk for common diseases like cancer and diabetes.[28][70]
It is anticipated that detailed knowledge of the human genome will offer new avenues for advances in medicine and biotechnology. Clear practical results of the project emerged even before the work was finished. For example, a number of companies, such as Myriad Genetics, started offering easy ways to administer genetic tests that can show predisposition to a variety of illnesses, including breast cancer, hemostasis disorders, cystic fibrosis, liver diseases, and many others. Also, the etiologies for cancers, Alzheimer's disease and other areas of clinical interest are considered likely to benefit from genome information and possibly may lead in the long term to significant advances in their management.[84][85]
There are also many tangible benefits for biologists. For example a researcher investigating a certain form of cancer may have narrowed down their search to a particular gene. By visiting the human genome database on the internet, this researcher can examine what other scientists have written about this gene, including (potentially) the three-dimensional structure of its product, its functions, its evolutionary relationships to other human genes, or to genes in mice, yeast, or fruit flies, possible detrimental mutations, interactions with other genes, body tissues in which this gene is activated, and diseases associated with this gene or other datatypes. Further, a deeper understanding of the disease processes at the level of molecular biology may determine new therapeutic procedures. Given the established importance of DNA in molecular biology and its central role in determining the fundamental operation of cellular processes, it is likely that expanded knowledge in this area will facilitate medical advances in numerous areas of clinical interest that may not have been possible without them.[86]
Analysis of similarities between DNA sequences from different organisms is also opening new avenues in the study of evolution. In many cases, evolutionary questions can now be framed in terms of molecular biology; indeed, many major evolutionary milestones (the emergence of the ribosome and organelles, the development of embryos with body plans, the vertebrate immune system) can be related to the molecular level. Many questions about the similarities and differences between humans and their closest relatives (the primates, and indeed the other mammals) are expected to be illuminated by the data in this project.[84][87]
The project inspired and paved the way for genomic work in other fields, such as agriculture. For example by studying the genetic composition of Tritium aestivum, the world's most commonly used bread wheat, great insight has been gained into the ways that domestication has impacted the evolution of the plant.[88] It is being investigated which loci are most susceptible to manipulation, and how this plays out in evolutionary terms. Genetic sequencing has allowed these questions to be addressed for the first time, as specific loci can be compared in wild and domesticated strains of the plant. This will allow for advances in genetic modification in the future which could yield healthier and disease-resistant wheat crops, among other things.
Ethical, legal, and social issues
[edit]At the onset of the Human Genome Project, several ethical, legal, and social concerns were raised in regard to how increased knowledge of the human genome could be used to discriminate against people. One of the main concerns of most individuals was the fear that both employers and health insurance companies would refuse to hire individuals or refuse to provide insurance to people because of a health concern indicated by someone's genes.[89] In 1996, the United States passed the Health Insurance Portability and Accountability Act (HIPAA), which protects against the unauthorized and non-consensual release of individually identifiable health information to any entity not actively engaged in the provision of healthcare services to a patient.[90]
Along with identifying all of the approximately 20,000–25,000 genes in the human genome (estimated at between 80,000 and 140,000 at the start of the project), the Human Genome Project also sought to address the ethical, legal, and social issues that were created by the onset of the project.[91] For that, the Ethical, Legal, and Social Implications (ELSI) program was founded in 1990. Five percent of the annual budget was allocated to address the ELSI arising from the project.[29][92] This budget started at approximately $1.57 million in the year 1990, but increased to approximately $18 million in the year 2014.[93]
While the project may offer significant benefits to medicine and scientific research, some authors have emphasized the need to address the potential social consequences of mapping the human genome. Historian of science Hans-Jörg Rheinberger wrote that "the prospect of 'molecularizing' diseases and their possible cure will have a profound impact on what patients expect from medical help, and on a new generation of doctors' perception of illness."[94]
In July 2024, an investigation by Undark Magazine[95] and co-published with STAT News[96] revealed for the first time several ethical lapses by the scientists spearheading the Human Genome Project. Chief among these was the use of roughly 75 percent of a single donor's DNA in the construction of the reference genome, despite informed consent forms, provided to each of the 20 anonymous donors participating, that indicated no more than 10 percent of any one donor's DNA would be used. About 10 percent of the reference genome belonged to one of the project's lead scientists, Pieter De Jong.[95]
See also
[edit]- 1000 Genomes Project – International research effort on genetic variation
- 100,000 Genomes Project – UK Government project that is sequencing whole genomes from National Health Service patients
- Earth BioGenome Project – Initiative aimed at sequencing the genomes of all named eukaryotic species
- Chimpanzee genome project – Effort to determine the DNA sequence of the chimpanzee genome
- Human Cytome Project – Single-cell biology and biochemistry
- ENCODE – Research consortium investigating functional elements in human and model organism DNA
- Genographic Project – Citizen science project
- HUGO Gene Nomenclature Committee – Committee for human gene name standards
- Human Brain Project – Scientific research project
- Human Connectome Project – Research project
- Human Epigenome Project
- Human Microbiome Project – Former research initiative
- Human proteome project – Scientific project coordinated by the Human Proteome Organization
- Human Variome Project
- List of biological databases
- Neanderthal genome project – Effort to sequence the Neanderthal genome
- Physiome – Wholistic physiological dynamics of an organism
- Wellcome Sanger Institute – British genomics research institute
References
[edit]- ^ Harshit Singh (2024). Cracking the Code of Life (Television Show). Public Broadcasting Service.
- ^ "Economic Impact of the Human Genome Project – Battelle" (PDF). Archived from the original (PDF) on 23 December 2012. Retrieved 1 August 2013.
- ^ "Human Genome Project Completion: Frequently Asked Questions". National Human Genome Research Institute (NHGRI).
- ^ "CHM13 T2T v1.1 – Genome – Assembly – NCBI". www.ncbi.nlm.nih.gov. Retrieved 16 June 2021.
- ^ "Genome List – Genome – NCBI". www.ncbi.nlm.nih.gov. Retrieved 16 June 2021.
- ^ "T2T-CHM13v2.0 – Genome – Assembly – NCBI". www.ncbi.nlm.nih.gov. Retrieved 14 June 2022.
- ^ a b c "Human Genome Project Completion: Frequently Asked Questions". genome.gov.
- ^ "Human Genome Project: Sequencing the Human Genome | Learn Science at Scitable". Nature. Retrieved 25 January 2016.
- ^ "History of the Human Genome Project". web.ornl.gov.
- ^ Maloy, S.; Hughes, K. (2013). Brenner's Encyclopedia of Genetics. Elsevier Science. p. 2-PA48. ISBN 978-0-08-096156-9. Retrieved 30 January 2025.
- ^ Edwards, A. W. F. (2013). Maloy, Stanley R.; Hughes, Kelly (eds.). Brenner's Encyclopedia of Genetics (2nd ed.). London: Academic Press, Elsevier Science. pp. 48–49. ISBN 978-0-08-096156-9.
- ^ Maxam AM, Gilbert W (February 1977). "A new method for sequencing DNA". Proc Natl Acad Sci U S A. 74 (2): 560–4. doi:10.1073/pnas.74.2.560. PMC 392330. PMID 265521.
- ^ Sanger F, Nicklen S, Coulson AR (December 1977). "DNA sequencing with chain-terminating inhibitors". Proc Natl Acad Sci U S A. 74 (12): 5463–7. doi:10.1073/pnas.74.12.5463. PMC 431765. PMID 271968.
- ^ Sinsheimer RL (November 1989). "The Santa Cruz Workshop – May 1985 and". Genomics. 5 (4): 954–956. doi:10.1016/0888-7543(89)90142-0. PMID 2591974.
- ^ Cook-Deegan, R.M. (1994). The Gene Wars: Science, Politics, and the Human Genome. W.W. Norton & Company. ISBN 978-0-393-03572-8. Retrieved 24 December 2024.
- ^ a b DeLisi C (October 2008). "Meetings that changed the world: Santa Fe 1986: Human genome baby-steps". Nature. 455 (7215): 876–877. Bibcode:2008Natur.455..876D. doi:10.1038/455876a. PMID 18923499. S2CID 41637733.
- ^ Dulbecco R (March 1986). "A turning point in cancer research: sequencing the human genome". Science. 231 (4742): 1055–1056. Bibcode:1986Sci...231.1055D. doi:10.1126/science.3945817. PMID 3945817.
- ^ Cook-Deegan, Robert M. (1994). The Gene Wars: Science, Politics, and the Human Genome. New York: W.W. Norton. p. 108.
- ^ "President Clinton Awards the Presidential Citizens Medals". clintonwhitehouse5.archives.gov.
- ^ "File:Plaque commemorating the Human Genome Project, outside Charles DeLisi's former office at DOE.png – Wikimedia Commons".
- ^ Bevatron's Encyclopedia of Inventions: a compendium of technological leaps, ground break discoveries and scientific breakthroughs that changed the world. The Human Genome Project, Charles DeLisi, pp. 360–362.
- ^ Origins of the Human Genome Project: A Political History – Bob Cook-Deegan
- ^ a b Gene Wars, Op.Cit. p. 102.
- ^ "Search". georgetown.edu.
- ^ "President Clinton Awards the Presidential Citizens Medals". nara.gov. Archived from the original on 1 August 2012. Retrieved 6 August 2014.
- ^ "Archived copy". Archived from the original on 3 March 2016. Retrieved 19 August 2013.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ DeLisi C (1988). "The Human Genome Project". American Scientist. 76 (5): 488. Bibcode:1988AmSci..76..488D.
- ^ a b "About the Human Genome Project: What is the Human Genome Project". The Human Genome Management Information System (HGMIS). 18 July 2011. Archived from the original on 2 September 2011. Retrieved 2 September 2011.
- ^ a b c Human Genome Information Archive. "About the Human Genome Project". US Department of Energy & Human Genome Project program. Archived from the original on 2 September 2011. Retrieved 1 August 2013.
- ^ Collins F, Galas D (1 October 1993). "A New Five-Year Plan for the United States: Human Genome Program". National Human Genome Research Institute. Retrieved 1 August 2013.
- ^ "Life on Earth to have its DNA analysed in the name of conservation". Nature. 563 (7730): 155–156. November 2018. Bibcode:2018Natur.563..155.. doi:10.1038/d41586-018-07323-y. PMID 30401859.
- ^ Lewin HA, Robinson GE, Kress WJ, Baker WJ, Coddington J, Crandall KA, et al. (April 2018). "Earth BioGenome Project: Sequencing life for the future of life". Proceedings of the National Academy of Sciences of the United States of America. 115 (17): 4325–4333. Bibcode:2018PNAS..115.4325L. doi:10.1073/pnas.1720115115. PMC 5924910. PMID 29686065.
- ^ a b Siegel, Vivian (14 April 2023). "The Human Genome Project Turns 20: Here's How It Altered the World". MIT Department of Biology. Retrieved 30 January 2025.
- ^ Cook-Deegan, Robert M. (1994). The Gene Wars: Science, Politics, and the Human Genome. New York: W.W. Norton. pp. 95–96.
- ^ Bishop, Jerry E.; Waldholz, Michael (1990). Genome: The Story of the Most Astonishing Scientific Adventure of Our Time – the Attempt to Map All the Genes in the Human Body. New York: Simon and Schuster. p. 54.
- ^ Bishop, Jerry E.; Waldholz, Michael (1990). Genome: The Story of the Most Astonishing Scientific Adventure of Our Time – the Attempt to Map All the Genes in the Human Body. New York: Simon and Schuster. p. 201.
- ^ "White House Press Release". Retrieved 22 July 2006.
- ^ "Scientists Complete Rough Draft of Human Genome".
- ^ Gitschier, Jane (31 January 2013). "Life, the Universe, and Everything: An Interview with David Haussler". PLOS Genetics. 9 (1): e1003282. doi:10.1371/journal.pgen.1003282. ISSN 1553-7390. PMC 3561096. PMID 23382705.
- ^ Noble I (14 April 2003). "Human genome finally complete". BBC News. Retrieved 22 July 2006.
- ^ Kolata G (15 April 2013). "Human Genome, Then and Now". The New York Times. Retrieved 24 April 2014.
- ^ "Human genome completed (again)". Nature News. Archived from the original on 11 October 2007. Retrieved 12 February 2025.
- ^ "The Human Genome Project FAQ". Genoscope. Centre National de Séquençage. 19 October 2013. Archived from the original on 22 July 2015. Retrieved 12 February 2015.
- ^ Schmutz J, Wheeler J, Grimwood J, Dickson M, Yang J, Caoile C, et al. (May 2004). "Quality assessment of the human genome sequence". Nature. 429 (6990): 365–368. Bibcode:2004Natur.429..365S. doi:10.1038/nature02390. PMID 15164052.
- ^ Dolgin E (December 2009). "Human genomics: The genome finishers". Nature. 462 (7275): 843–845. doi:10.1038/462843a. PMID 20016572.
- ^ Chaisson MJ, Huddleston J, Dennis MY, Sudmant PH, Malig M, Hormozdiari F, Antonacci F, Surti U, Sandstrom R, Boitano M, Landolin JM, Stamatoyannopoulos JA, Hunkapiller MW, Korlach J, Eichler EE (January 2015). "Resolving the complexity of the human genome using single-molecule sequencing". Nature. 517 (7536): 608–611. Bibcode:2015Natur.517..608C. doi:10.1038/nature13907. PMC 4317254. PMID 25383537.
- ^ "Human Genome Issues". Genome Reference Consortium. Retrieved 29 June 2019.
- ^ The (near) complete sequence of a human genome, 6 October 2020
- ^ Miga, Karen H.; Koren, Sergey; Rhie, Arang; Vollger, Mitchell R.; Gershman, Ariel; Bzikadze, Andrey; Brooks, Shelise; Howe, Edmund; Porubsky, David; Logsdon, Glennis A.; Schneider, Valerie A. (September 2020). "Telomere-to-telomere assembly of a complete human X chromosome". Nature. 585 (7823): 79–84. Bibcode:2020Natur.585...79M. doi:10.1038/s41586-020-2547-7. ISSN 1476-4687. PMC 7484160. PMID 32663838.
- ^ Logsdon, Glennis A.; Vollger, Mitchell R.; Hsieh, PingHsun; Mao, Yafei; Liskovykh, Mikhail A.; Koren, Sergey; Nurk, Sergey; Mercuri, Ludovica; Dishuck, Philip C.; Rhie, Arang; de Lima, Leonardo G. (May 2021). "The structure, function and evolution of a complete human chromosome 8". Nature. 593 (7857): 101–107. Bibcode:2021Natur.593..101L. doi:10.1038/s41586-021-03420-7. ISSN 1476-4687. PMC 8099727. PMID 33828295.
- ^ Wrighton, Katharine (February 2021). "Filling in the gaps telomere to telomere". Nature Milestones: Genomic Sequencing: S21.
- ^ Reardon, Sara (4 June 2021). "A complete human genome sequence is close: how scientists filled in the gaps". Nature. 594 (7862): 158–159. Bibcode:2021Natur.594..158R. doi:10.1038/d41586-021-01506-w. PMID 34089035. S2CID 235346408.
- ^ "GitHub – marbl/CHM13-issues: CHM13 human reference genome issue tracking". GitHub. Retrieved 26 July 2021.
- ^ Rhie; et al. (1 December 2022). "The complete sequence of a human Y chromosome". bioRxiv. doi:10.1101/2022.12.01.518724. S2CID 254181409.
- ^ Special Issue: Completing the human genome, Science, vol. 376, no. 6588 (1 April 2022).
- ^ "The human genome sequence is now complete". Genome.gov. 6 April 2022. Retrieved 30 January 2025.
- ^ Rhie, Arang; Nurk, Sergey; Cechova, Monika; Hoyt, Savannah J.; Taylor, Dylan J.; Altemose, Nicolas; Hook, Paul W.; Koren, Sergey; Rautiainen, Mikko; Alexandrov, Ivan A.; Allen, Jamie; Asri, Mobin; Bzikadze, Andrey V.; Chen, Nae-Chyun; Chin, Chen-Shan (23 August 2023). "The complete sequence of a human Y chromosome". Nature. 621 (7978): 344–354. Bibcode:2023Natur.621..344R. doi:10.1038/s41586-023-06457-y. ISSN 0028-0836. PMC 10752217. PMID 37612512. S2CID 254181409.
- ^ 'Most troublesome chromosome' gets new respect after full sequence (Report). 23 August 2023. doi:10.1126/science.adk4669.
- ^ "UCSC Genome Browser Home". genome.ucsc.edu.
- ^ "Ensembl Genome Browser". ensembl.org.
- ^ Mardis ER (March 2008). "The impact of next-generation sequencing technology on genetics". Trends in Genetics. 24 (3): 133–141. doi:10.1016/j.tig.2007.12.007. PMC 2680276. PMID 18262675.
- ^ Liu Y, Gonzàlez-Porta M, Santos S, Brazma A, Marioni JC, Aebersold R, et al. (August 2017). "Impact of Alternative Splicing on the Human Proteome". Cell Reports. 20 (5): 1229–1241. doi:10.1016/j.celrep.2017.07.025. PMC 5554779. PMID 28768205.
- ^ Ballouz S, Dobin A, Gillis JA (August 2019). "Is it time to change the reference genome?". Genome Biology. 20 (1): 159. doi:10.1186/s13059-019-1774-4. PMC 6688217. PMID 31399121.
- ^ Pertea M, Salzberg SL (2010). "Between a chicken and a grape: estimating the number of human genes". Genome Biology. 11 (5): 206. doi:10.1186/gb-2010-11-5-206. PMC 2898077. PMID 20441615.
- ^ a b c Venter JC, Adams MD, Myers EW, Li PW, Mural RJ, Sutton GG, et al. (February 2001). "The sequence of the human genome". Science. 291 (5507): 1304–1351. Bibcode:2001Sci...291.1304V. doi:10.1126/science.1058040. PMID 11181995.
- ^ Lander ES, Linton LM, Birren B, Nusbaum C, Zody MC, Baldwin J, et al. (International Human Genome Sequencing Consortium (IHGSC)) (October 2004). "Finishing the euchromatic sequence of the human genome". Nature. 431 (7011): 931–945. Bibcode:2004Natur.431..931H. doi:10.1038/nature03001. PMID 15496913.
- ^ Spencer G (20 December 2004). "International Human Genome Sequencing Consortium Describes Finished Human Genome Sequence". NIH Nes Release. National Institutes of Health.
- ^ Bryant JA (2007). Design and information in biology: From molecules to systems. WIT Press. p. 108. ISBN 978-1-85312-853-0.
...brought to light about 1200 protein families. Only 94 protein families, or 7%, appear to be vertebrate specific
- ^ Piovesan, A.; Pelleri, M. C.; Antonaros, F.; Strippoli, P.; Caracausi, M.; Vitale, L. (2019). "On the length, weight and GC content of the human genome". BMC Research Notes. 12 (1): 106. doi:10.1186/s13104-019-4137-z. PMC 6391780. PMID 30813969.
- ^ a b Wellcome Sanger Institute. "The Human Genome Project: a new reality". Wellcome Trust Sanger Institute, Genome Research Limited. Archived from the original on 1 August 2013. Retrieved 1 August 2013.
- ^ "Celera: A Unique Approach to Genome Sequencing". ocf.berkeley.edu. Biocomputing. 2006. Retrieved 1 August 2013.
- ^ Davidson College (2002). "Sequencing Whole Genomes: Hierarchical Shotgun Sequencing v. Shotgun Sequencing". bio.davidson.edu. Department of Biology, Davidson College. Retrieved 1 August 2013.
- ^ Human Genome Project Information Archive (2013). "U.S. & International HGP Research Sites". US Department of Energy & Human Genome Project. Retrieved 1 August 2013.
- ^ Vizzini C (19 March 2015). "The Human Variome Project: Global Coordination in Data Sharing". Science & Diplomacy. 4 (1).
- ^ Sulston, John; Ferry, Georgina (2002). The Common Thread: A Story of Science, Politics, Ethics and the Human Genome. London: Bantam Press. p. 160. ISBN 0593-048016.
- ^ Wade, Nicholas (23 March 1999). "Who'll Sequence Human Genome First? It's Up to Phred". New York Times.
- ^ Roach JC, Boysen C, Wang K, Hood L (March 1995). "Pairwise end sequencing: a unified approach to genomic mapping and sequencing". Genomics. 26 (2): 345–353. doi:10.1016/0888-7543(95)80219-C. PMID 7601461.
- ^ Center for Biomolecular Science & Engineering. "The Human Genome Project Race". Center for Biomolecular Science and Engineering. Retrieved 1 August 2013.
- ^ Gillis, Justin (15 March 2000). "Clinton, Blair Urge Open Access to Gene Data". Washington Post.
- ^ Osoegawa K, Mammoser AG, Wu C, Frengen E, Zeng C, Catanese JJ, de Jong PJ (March 2001). "A bacterial artificial chromosome library for sequencing the complete human genome". Genome Research. 11 (3): 483–496. doi:10.1101/gr.169601. PMC 311044. PMID 11230172.
- ^ Tuzun E, Sharp AJ, Bailey JA, Kaul R, Morrison VA, Pertz LM, et al. (July 2005). "Fine-scale structural variation of the human genome". Nature Genetics. 37 (7): 727–732. doi:10.1038/ng1562. PMID 15895083. S2CID 14162962.
- ^ Kennedy D (August 2002). "Not wicked, perhaps, but tacky". Science. 297 (5585): 1237. doi:10.1126/science.297.5585.1237. PMID 12193755.
- ^ Venter JC (February 2003). "A part of the human genome sequence". Science. 299 (5610): 1183–1184. doi:10.1126/science.299.5610.1183. PMID 12595674. S2CID 5188811.
- ^ a b Naidoo N, Pawitan Y, Soong R, Cooper DN, Ku CS (October 2011). "Human genetics and genomics a decade after the release of the draft sequence of the human genome". Human Genomics. 5 (6): 577–622. doi:10.1186/1479-7364-5-6-577. PMC 3525251. PMID 22155605.
- ^ Gonzaga-Jauregui C, Lupski JR, Gibbs RA (2012). "Human genome sequencing in health and disease". Annual Review of Medicine. 63 (1): 35–61. doi:10.1146/annurev-med-051010-162644. PMC 3656720. PMID 22248320.
- ^ Snyder M, Du J, Gerstein M (March 2010). "Personal genome sequencing: current approaches and challenges". Genes & Development. 24 (5): 423–31. doi:10.1101/gad.1864110. PMC 2827837. PMID 20194435.
- ^ Lander ES (February 2011). "Initial impact of the sequencing of the human genome" (PDF). Nature. 470 (7333): 187–97. Bibcode:2011Natur.470..187L. doi:10.1038/nature09792. hdl:1721.1/69154. PMID 21307931. S2CID 4344403.
- ^ Peng JH, Sun D, Nevo E (2011). "Domestication Evolution, Genetics And Genomics In Wheat". Molecular Breeding. 28 (3): 281–301. doi:10.1007/s11032-011-9608-4. S2CID 24886686.
- ^ Greely H (1992). The Code of Codes: Scientific and Social Issues in the Human Genome Project. Cambridge, Massachusetts: Harvard University Press. pp. 264–265. ISBN 978-0-674-13646-5.
- ^ US Department of Health and Human Services (26 August 2015). "Understanding Health Information Privacy".
- ^ Human Genome Information Archive. "Insights Learned from the Human DNA Sequence". US Department of Energy & Human Genome Project program. Archived from the original on 3 September 2011. Retrieved 20 February 2021.
- ^ "What were some of the ethical, legal, and social implications addressed by the Human Genome Project?". Genetics Home Reference. US National Library of Medicine. 2013. Retrieved 1 August 2013.
- ^ "ELSI Research Program Fact Sheet – National Human Genome Research Institute (NHGRI)". www.genome.gov. Retrieved 27 September 2016.
- ^ Rheinberger HJ (2000). Living and Working with the New Medical Technologies. Cambridge: Cambridge University Press. p. 20.
- ^ a b Magazine, Undark (9 July 2024). "Haunting the Human Genome Project: A Question of Consent". Undark Magazine. Retrieved 11 July 2024.
- ^ Undark, Ashley Smart- (9 July 2024). "5 takeaways from the Human Genome Project investigation". STAT. Retrieved 11 July 2024.
Further reading
[edit]- McElheny VK (2010). Drawing the Map of Life: Inside the Human Genome Project. Basic Books. ISBN 978-0-465-03260-0. 361 pages. Examines the intellectual origins, history, and motivations of the project to map the human genome; draws on interviews with key figures.
- Collins F (2006). The Language of God: A Scientist Presents Evidence for Belief. Free Press. ISBN 978-0-7432-8639-8. OCLC 65978711.
- Venter JC (18 October 2007). A Life Decoded: My Genome: My Life. New York City: Viking Adult. ISBN 978-0-670-06358-1. OCLC 165048736.
- Cook-Deegan R (1994). The Gene Wars: Science, Politics, and the Human Genome. New York: W W Norton. ISBN 978-0-393-03572-8.
- Lone Dog L (1999). "Whose genes are they? The Human Genome Diversity Project". Journal of Health & Social Policy. 10 (4): 51–66. doi:10.1300/J045v10n04_04. PMID 10538186.
External links
[edit]- National Human Genome Research Institute (NHGRI). NHGRI led the National Institutes of Health's contribution to the International Human Genome Project. This project, which had as its primary goal the sequencing of the three billion base pairs that make up the human genome, was successfully completed in April 2003.
- Human Genome News. Published from 1989 to 2002 by the US Department of Energy, this newsletter was a major communications method for coordination of the Human Genome Project. Complete online archives are available.
- The HGP information pages Department of Energy's portal to the international Human Genome Project, Microbial Genome Program, and Genomics:GTL systems biology for energy and environment
- yourgenome.org: The Sanger Institute public information pages has general and detailed primers on DNA, genes, and genomes, the Human Genome Project and science spotlights.
- Ensembl project, an automated annotation system and browser for the human genome
- UCSC genome browser, This site contains the reference sequence and working draft assemblies for a large collection of genomes. It also provides a portal to the ENCODE project.
- Nature magazine's human genome gateway, including the HGP's paper on the draft genome sequence
- Wellcome Trust Human Genome website A free resource allowing you to explore the human genome, your health and your future.
- Learning about the Human Genome. Part 1: Challenge to Science Educators. ERIC Digest.
- Learning about the Human Genome. Part 2: Resources for Science Educators. ERIC Digest.
- Patenting Life by Merrill Goozner
- Prepared Statement of Craig Venter of Celera Venter discusses Celera's progress in deciphering the human genome sequence and
relationship to healthcare and to the federally funded Human Genome Project.
- Cracking the Code of Life Companion website to 2-hour NOVA program documenting the race to decode the genome, including the entire program hosted in 16 parts in either QuickTime or RealPlayer format.
- Bioethics Research Library Numerous original documents at Georgetown University.
- David J. Galas
- Works by archive
- Works by Human Genome Project at Project Gutenberg
- Project Gutenberg hosts e-texts for Human Genome Project, titled Human Genome Project, Chromosome Number # (# denotes 01–22, X and Y). This information is the raw sequence, released in November 2002; access to entry pages with download links is available through Human Genome Project, Chromosome Number 01 for Chromosome 1 sequentially to Human Genome Project, Y Chromosome for the Y Chromosome. Note that this sequence might not be considered definitive because of ongoing revisions and refinements. In addition to the chromosome files, there is a supplementary information file dated March 2004 which contains additional sequence information.
- Works by or about Human Genome Project at the Internet Archive
 KSF
KSF
