Bis(Triethoxysilylpropyl)Tetrasulfide
 From Handwiki
From Handwiki 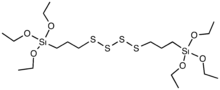
| |
| Names | |
|---|---|
| Preferred IUPAC name
[Tetrasulfanediyldi(propane-3,1-diyl)]bis(triethoxysilane) | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL |
|
| ChemSpider |
|
| EC Number |
|
PubChem CID
|
|
| UNII |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C18H42O6S4Si2 |
| Molar mass | 538.95 |
| Appearance | yellow syrup |
| Density | 1.08 g/cm3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Bis(triethoxysilylpropyl)tetrasulfide is an organosulfur compound with the formula S4[C3H6Si(OEt)3]2 (Et = C2H5). The molecule consists of two trialkoxysilyl propyl groups linked with a polysulfide. It is often sold as a mixture with the trisulfide. The compound is a colorless viscous liquid that is soluble in ordinary organic solvents such as toluene. Commercial samples often are yellowish. The compound is added to rubber compositions that contain silica filler.[1][2][3]
Synthesis and reactivity
The compound was first prepared by the reaction of 3-(triethoxysilyl)propyl chloride with sodium tetrasulfide:[4]
- Na2S4 + 2 ClC3H6Si(OEt)3 → S4[C3H6Si(OEt)3]2 + 2 NaCl
Bis(triethoxysilylpropyl)tetrasulfide is a bifunctional molecule in that it contains two kinds of reactive functional groups. The tetrasulfide group is a polysulfide, which means that it consists of a chain of sulfur atoms. S-S bonds are susceptible to reduction (to thiols), attachment to metals (e.g., for protection against corrosion), and vulcanization. The triethoxysilyl groups are susceptible to hydrolysis, resulting in cross-linking via sol-gel condensation. In the usual application of this chemical, the hydrolyzed siloxy groups attach to silica particles and the polysulfide groups link to the organic polymer.[5]
References
- ↑ Kohjiya, Shinzo; Ikeda, Yuko (2000). "Reinforcement of general-purpose grade rubbers by silica generated in situ". Rubber Chemistry and Technology 73 (3): 534–550. doi:10.5254/1.3547604.
- ↑ Wolff, Siegfried (1996). "Chemical aspects of rubber reinforcement by fillers". Rubber Chemistry and Technology 69 (3): 325–346. doi:10.5254/1.3538376.
- ↑ Vilmin, F.; Bottero, I.; Travert, A.; Malicki, N.; Gaboriaud, F.; Trivella, A.; Thibault-Starzyk, F. (2014). "Reactivity of Bis[3-(triethoxysilyl)propyl] Tetrasulfide (TESPT) Silane Coupling Agent over Hydrated Silica: Operando IR Spectroscopy and Chemometrics Study". The Journal of Physical Chemistry C 118 (8): 4056–4071. doi:10.1021/jp408600h.
- ↑ Thurn, Friedrich; Meyer-Simon, Eugen; Michel, Rudolf "Verfahren zur Herstellung von Organosiliziumverbindungen (Continuous manufacture of bis[3-(triethoxysilyl)propyl] tetrasulfide)" Ger. Offen. (1973), DE 2212239 A1 19731004.
- ↑ Choi, S.-S.; Kim, I.-S.; Woo, C.-S. (2007). "Influence of TESPT Content on Crosslink Types and Rheological Behaviors of Natural rubber compounds reinforced with Silica". Journal of Applied Polymer Science 106 (4): 2753–2758. doi:10.1002/app.25744.
 |
Categories: [Sulfur compounds] [Corrosion inhibitors] [Ethoxides] [Organosilicon compounds]
↧ Download as ZWI file | Last modified: 03/11/2024 21:49:02 | 1 views
☰ Source: https://handwiki.org/wiki/Chemistry:Bis(triethoxysilylpropyl)tetrasulfide | License: CC BY-SA 3.0

 KSF
KSF